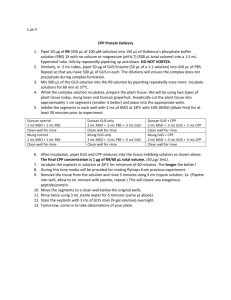
Linkage for mutants dot1, dot2, dot3, 116, dot4, 129B, 134
advertisement

Figure s5. Supplemental methods Genetic mapping and cloning Linkage for mutants dot1, dot2, dot3, 116, dot4, 129B, 134, 139, dot5, and 4385 was established to between nga168 and nga361, to between nga 106 and nga 139, nga249, nga139, nga1139, nga162, nga63, nga158, nga63, and to AtCTR1, respectively. DNA from 300-800 mutant plants was assayed with Cereon’s collection of insertion/deletion and single-nucleotide polymorphism markers (Jander et al., 2002) to finely map the mutations. The mutations not cloned were mapped to regions flanked by the following BACs: 116 (MBG8 and M1K19, 14 BACs), 129B (MQC12 and MXL8, 7 BACs), 134 (F26G16 and F17F8, 4 BACs), and 4385 (F17F15 and T32M21, 6 BACs). For cloned genes (dot1/At2g36120, dot2/At5g16780, dot3/At5g10250, dot4/At4g18750, dot5/At1g13290), mutations were finely mapped as above to a region spanned by a few genes (less than 10) and subsequently sequenced and/or transformed with transformation-competent artificial chromosomes (TACs) (Liu et al., 1995) and examined for complementation of mutant phenotypes. Vector construction dot1 The DOT1/At2g36120 coding sequence and 1.54 kb of upstream sequence were PCR-amplified from genomic DNA and sub-cloned into XbaI/EcoRI sites of the PBI101.1 binary vector (Clontech, Palo Alto, CA) and the DOT1/At2g36120 coding sequence was sub-cloned into BamHI/EcoR1 sites of the pJIM19 (KAN) binary vector downstream of the 35S CaMV promoter for complementation (PDOT1:DOT1; P35S:DOT1). 1.54 kb of upstream sequence was sub-cloned into XbaI/BamH1 sites of the PBI101.1 binary vector (Clontech) upstream of the GUS gene (PDOT1/At2g36120:GUS). dot2 The dot2/At5g16780 coding sequence (minus the stop codon) was PCRamplified and sub-cloned into BamHI/StuI sites of the pJIM19smGFP binary vector in frame and upstream of the smGFP gene (minus the start codon) (P35S:DOT2). 1 kb of DOT2/At5g16780 upstream sequence was sub-cloned into HindIII/BamHI sites of the pBI101.1 binary vector (Clontech) upstream of the GUS gene (PDOT2/At5g16780:GUS). dot3 The upstream sequence of DOT3/At5g10250 (661 bp of sequence to the next gene), the gene, and the predicted 3’ UTR were amplified from genomic DNA and subcloned into the SalI/BamHI sites of the pBI101.1 binary vector (Clonetech) (PDOT3 :DOT3), for complementation and for the DOT3 promoter:GUS fusion construct (PDOT3/At5g10250:GUS), as no expression was detected from fusion of 661 bp of upstream sequence to the GUS gene. The DOT3/At5g10250 genomic sequence was PCR amplified and subcloned into BamHI/KpnI sites of the pJIM19 (KAN) binary vector, generating P35S:DOT3. dot4 The DOT4/At4g18750 coding sequence was PCR-amplified and sub-cloned into BamHI/SacI sites of the pJIM19 (KAN) binary vector downstream of the 35S CaMV promoter (P35S:DOT4). 1.59 kb of upstream sequence was sub-cloned into XbaI/BamHI sites of the pBI101.1 binary vector (Clontech) upstream of the GUS gene (PDOT4/At4g18750:GUS), or replaced the 35S CaMV in the XbaI/BamHI site of the P35S:DOT4 construct upstream of the coding sequence (PDOT4:DOT4). dot5 The full DOT5/At1g13290 genomic sequence was PCR amplified from wild-type L. erecta plants and subcloned into XbaI/XhoI sites of the pJIM19 (KAN) binary vector (P35S:DOT5) for complementation. DOT5/At1g13290 upstream sequence was sub-cloned into SmaI/XbaI sites of the pBI101.1 binary vector (Clontech) upstream of the GUS gene (PDOT5/At1g13290:GUS). Constructs were introduced into wild-type and/or mutants by the same aforementioned method for TACs. Homozygous mutants were transformed, except for dot2, which is infertile and heterozygous mutants were used in this case. Transformants were selected on agar media 50g/mL kanamycin.