exer9c
advertisement

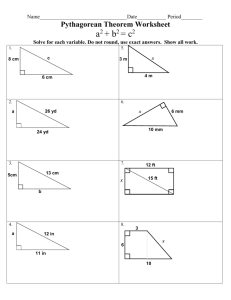
Biomolecular NMR spectroscopy Exercise 9 1. Consider homonuclear cross-relaxation that is governed by the rate constant ISNOE 2 02 4 c 40 2 rIS6 6 . 1 2 2 1 4 0 c Draw ISNOE as function of for few values of c, i.e. small (amino acid), medium (peptide) and large (protein) molecule. A small molecule has a short tc and a large one a large tc. Let’s plot function and identify the small, medium and large molecules from the curve. Most importantly we see that the function changes its sign. It could be that an intermediate size of a molecule gives no NOEs (no signal). Well this is interesting. Perhaps with adjustments of temperature that will affect tc we are able to tune for a peptide to give almost zero NOEs or slightly negative. Suppose that the peptide binds to a larger protein, then it will give NOEs because of the increase in correlation time. This can happen even if the peptide is in a large molar excess compared to the large protein. The signal is a weighted average and the weight is in favor of the molecular complex. 2. Consider also rotating frame homonuclear cross-relaxation rate constant ISROE 2 02 4 c 3 2 2 6 2 2 40 rIS 1 0 c for large molecules. An experiment (TOCSY) is designed to carry homonuclear correlation spectroscopy via scalar couplings but contains ISNOE contribution as an artifact due to rapid rf-pulsing. How could you remove the effect of ISROE? We see that ROE signal is a monotonous with c and positive whereas NOE is for large molecules negative. Perhaps we could insert windows into the TOCSY sequence that would allow NOE to build up just enough to cancel ROE. Such a sequence is called clean-TOCSY. 3. Short inter proton distances are obtained from a two (or higher) dimensional proton-proton correlation spectrum. The cross peak intensities ( I S) develop with time 1 a IS (t ) a SI (t ) exp ( )t1 exp( 2t ) 2 and the diagonal peaks ( I = S) develop with time a II (t ) a SS (t ) 1 exp ( )t1 exp( 2t ). 2 Imagine that you are setting up a three-dimensional proton-proton NOE spectrum that is edited by 13C frequency. The experiment will take three days because your sample is dilute and your lab hasn’t got the money to buy a cryo-probe. Under these circumstances you wish to maximize the cross peak intensity by setting the mixing time mix accordingly 1 but you are only able to monitor quickly the dependence of diagonal peak on mix from the one-dimensional spectrum. A one-dimensional experiment will come in a couple of minutes. How do you choose mix? Once a gain we need to plot the cross peak and diagonal peak intensities as a function of time. We are really interested in to optimize for the diagonal peaks that we don’t see from the one dimensional spectrum. However, now that we now the mutual time dependences of the cross and diagonal peaks we can of course read from the diagonal peak intensity how large the cross peak should be. As a rule of thumb you will find that when the diagonal peak has decreased to have of its initial intensity (mix = 0) when the cross peak has reached its maximum. 4. A long mixing time has its drawbacks i.e. spin-diffusion. Consider three protons in space so that H 1 – 4 Å – H2 – 4 Å – H3. The corresponding relaxation rate matrix is 1 12 0 R 12 2 23 0 3 23 The time dependence of I1z is 3 3 1 2 2 1 3 3 I 1z ( mix ) exp( R mix )1k I kz (0) E1k R1k mix R1k mix R1k mix I kz (0) 2 6 k 1 k 1 1 1 1 1 mix ( 12 12 2 ) mix 2 ( 13 2 1 12 2 2 2 12 2 ) mix 3 I 1z (0) 2 6 1 1 2 2 3 12 mix ( 1 2 ) 12 mix ( 12 122 ) 12 ( 1 2 ) 2 12 12 23 mix I 2 z (0) 2 6 1 1 2 3 12 23 mix ( 1 2 3 ) 12 23 mix I 3 z (0) 6 2 What is the meaning of 1mix and –12mix and the second-order term 1/21223mix2? We see from above (ex.3) that 1mix must present the falling intensity of the diagonal peaks. I1 is loosing its magnetization to the neighbors. The desired term is –12mix which will result the cross peak that allows us to identify that 1 and 2 are spatially close. The second-order term is in this respect a nuisance because it will give a peak between 1 and 3 that are not close by but the magnetization is relayed via 2. 5. Pulsed field gradients (PFG) are used to purge undesired coherences or to select desired coherence. PFGs can be treated by the product operator formalism. When the gradient with strength Gz is applied along z-axis the operator will cause an evolution during t the effect = GzzIzt depends on the position of the spin in the NMR tube. Clearly when the PFG-pulse is long spins will dephase, in a way wrap, however not in an arbitrary manner. We can rephase them by applying a focusing PFG that will unwrap the phase. Find out what will happen to Iy and Iz subject to chemical shift evolution during the echo sequences with 180x pulse between two gradients. 2 3 Useful formulas In the very fast limit 1/T2,obs = pA/T2A + pB/T2B In the moderately fast limit 1/T2,obs = pA/T2A + pB/T2B + pApB24(AB)2/k-1 G = RTc [22.96 + ln(Tc/)] No exchange k = 0 linewidth vo = 1/T2 Slow exchange k = vevo) where ve is the exchange linewidth Intermediate exchange k = fe2fo2)1/2/21/2 where fo and fe is the frequency difference of the two peaks without and with exchange. Coalescence k = fo Fast exchange k = fo2vevo) where ve is the exchange linewidth 4