Greenhouse Effect and Global Warming
advertisement

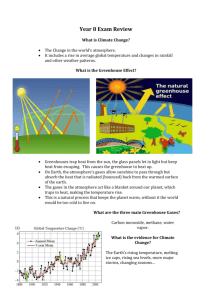
Climate Change Notes E2.3A Explain how carbon exists in different forms such as limestone (rock), carbon dioxide (gas), carbonic acid (water), and animals (life) with Earth systems and how those forms can be beneficial or harmful to humans. E2.2D Identify the main sources of energy to the climate system. E4.p2B Describe the difference between weather and climate. (prerequisite) E5.4A Explain the natural mechanism of the Greenhouse effect including comparisons of the major greenhouse gases (water vapor, carbon dioxide, methane, nitrous oxide, and ozone). Chapter 14.4; Chapter 27.3; Pgs. 375-377 Pg. 782 (compares atmosphere on Earth with atmosphere on Mars and Venus) The Greenhouse Effect- the heating process where gases in the atmosphere trap heat from the sun to continually warm the earth - About 70 percent of the sun’s energy that reaches the outer atmosphere enters (other 30 percent reflected by outer atmosphere) - 50 percent reaches the surface - 20 percent is absorbed by the atmosphere on its way in - Solar radiation strikes the earth’s surface, then gets re-radiated back into the atmosphere. - Gases like water vapor (H2O) and carbon dioxide (CO2) absorb this energy and then radiate it back to the earth’s surface - Without it, life (as we know it) would not exist on earth. EX: Sitting in your car with the windows rolled up on a hot, sunny day. Hotter inside the car than outside. Greenhouse Gases- atmospheric gases that absorb solar energy and trap heat to warm the earth. They allow solar radiation to come in but not get out. Water Vapor (H2O)- accounts for most of the greenhouse effect Carbon Dioxide (CO2)- produced naturally from volcanic eruptions, and also by humans; has increased by 30 percent since Industrial Revolution Methane (CH4)- principal component of natural gas; produced by bacteria and given off from cattle gas (burping); also put into atmosphere by the melting of permafrost at the poles Nitrous Oxide (N2O)- laughing gas; also used as a propellant; produced by bacteria in soils and the oceans; attacks the ozone (helps regulate the amount) Ozone (O3)- produced when UV rays react with atmospheric oxygen; * SIDE NOTES - Venus is very HOT because atmosphere contains 95 percent CO2 - Burning ONE molecule of Methane (natural gas) in the presence of oxygen produces ONE molecule of CO2 and 2 molecules of water vapor (H2O)- BOTH are greenhouse gases. - Fossil fuels: coal, oil, gasoline Anthropogenic (Human) Affects CO2- Burning fossil fuels (gasoline and oil) in cars and industry; deforestation (cutting down trees); respiration (breathing) CH4- Increased livestock; landfill emissions; septic emissions; gas pipeline emissions N2O- Increased use of fertilizers that release nitrous oxides CFCs- used in refrigeration systems and fire suppressants It’s a vicious cycle. As temperatures increase, relative humidity increases (warm air has capacity to hold more water) and more water vapor in the air means higher temperatures due to the greenhouse effect. Global Warming- a rise in average global temperatures - Gases that trap heat are called greenhouse gases - Recently, there has been an increase in the amount of CO2 gases, due to human activity, burning fossil fuels (factories, automobiles, etc.) - With an increase in greenhouse gases, more heat energy is being trapped inside the earth, and global temperatures continue to rise. - By 2010, yearly average temperatures could rise about 6 F. Radiation Balance and What’s Being Done http://asd-www.larc.nasa.gov/SCOOL/budget.gif - For life to continue to exist on earth, the amount of heat energy (radiation) must be in balance. If CO2 levels continue rise, too much heat would be trapped, which could severely affect plant and animal life and habitat. - Many countries have signed agreements to reduce the amount of CO2 that is being produced in their countries. E5.4B Describe natural mechanisms that could result in significant changes in climate (e.g. major volcanic eruptions, changes in sunlight received by the earth, and meteorite impacts). Pgs. 372-374 Volcanic Eruptions Emit CO2 (& water vapor) into the atmosphere, which is a heat-trapping greenhouse gas. Emit sulfuric gases, which turn into sulfuric aerosols. These aerosols absorb or trap terrestrial (earth) radiation and heat the earth. These sulfuric gases also combine with water to form acid rain which increases the weathering of limestone and puts more carbon dioxide into the air. Emit gases that react with CFCs and reduce the ozone levels The HAZE EFFECT- when gases and dust (mainly from volcanic eruptions) prevent solar radiation from reaching the earth and global temperature temporarily COOLS. Solar Radiation The stronger the intensity of solar radiation (more sunspots), the warmer the earth’s atmosphere will be. The weaker the intensity of solar radiation (less sunspots), the cooler the earth’s atmosphere will be. Axial Tilt and Orbital Change (Milankovitch Cycles) As earth’s axial tilt increases (higher angle), temperature range increases. As earth’s axial tilt decreases (lower angle), temperature range decreases. Changes every 40,000 years (between 21.5 and 24.5 degree tilt) As earth orbits closer to (narrower/elliptical orbit) the sun, temperature increases. As earth orbits further away from (wider/circular orbit) the sun, temperature decreases. Known as eccentricity Changes every 100,000 years When earth “wobbles” (for 26,000 years), like a top that is slowing down, it gets closer to and further away from the Sun. Known as precession Meteorite Impacts Meteorite- space object that collides with the Earth and survives impact. Dust from these collisions causes a haze effect which can affect climate Haze temporarily cools the climate (Can later warm climate as more heat is trapped) Dust provides more condensation nuclei (the beginning of clouds), which lead to more rain E5.4C Analyze the empirical relationship between the emissions of carbon dioxide, atmospheric carbon dioxide levels and the average global temperature over the past 150 years. Carbon Dioxide Emissions Since 1750, carbon dioxide emissions have increased by 87 ppm (31% increase) 315 billion tons of carbon has been released Nearly half of it has been released in the last 35 years (since 1970) 2003-2004, there was a 5.4 percent increase in used carbon BOTTOM LINE: Humans are putting more and more carbon dioxide into the atmosphere Atmospheric Carbon Dioxide Levels Before the start of the Industrial Revolution (1800s), atmospheric CO2 was at 280 ppm (parts per million). In the last 233 years, it has increased to 380 ppm. First 200 years, it increased from 280 to 330 ppm. In the last 33 years, it has increased from 330 to 380 ppm. Average Global Temperature Over the past 150 years average global temperature has risen anywhere from 0.5° to 1.5° C E5.4D Based on evidence of observable changes in recent history and climate change models, explain the consequences of warmer oceans (including the results of increased evaporation, shoreline and estuarine impacts, oceanic algae growth, and coral bleaching) and changing climatic zones (including the adaptive capacity of the atmosphere). Consequences of Warmer Oceans Increased evaporation and shoreline impacts Increased evaporation leads to more rain (stronger hurricanes) Can raise sea-levels (in conjunction with the melting of the polar ice caps) Floods will occur Shoreline environments are disrupted and certain organisms cannot adapt Highly populated coastal areas are at greater risk of damage and destruction Increased evaporation will also lead to higher salinity of ocean water Estuarine Impacts and algae growth Estuary- place where river meets the sea/ocean. Warmer ocean water heats the atmosphere above and more rain can produce more runoff. More runoff can add phosphates and nitrates to the ocean, which can cause an immense growth of algae. This prevents other aquatic life from inhabiting that area of water. Less runoff would decrease flushing, and allow predators and pathogens to penetrate the estuarine zone. Coral Bleaching Coral Reefs play a major role in the economies of Florida and Hawaii. Fisheries, recreation, tourism, coastal protection In the last 100 years, there has been a major decline in the health of coral reefs With warmer ocean temperatures, coral skeletons get weaker, they expel the algae they depend on to survive (get white, known as coral bleaching), and they do not grow as much. As these coral die, surrounding areas could lose millions to billions of dollars a year in economic dollars. Certain areas of the world receive more or less heat and more or less precipitation, which has a domino affect on the local ecosystem. E5.4e Based on evidence from historical climate research (e.g. fossils, varves, ice core data) and climate change models, explain how the current melting of polar ice caps can impact the climactic system. Polar ice caps reflect incoming solar radiation back into space due to their high albedo (ability to reflect light). Through this process, the global climate is kept cooler because the solar radiation is reflected versus being absorbed. With the increasing global temperatures, polar ice caps have begun melting which has allowed for more solar radiation to be absorbed. Hadley Scientists predict that sea level will rise 8 to 12 inches by the year 2100. Liquid water absorbs the Sun's energy rather than reflecting it into the atmosphere the way ice does. As the oceans warm and ice thins, more solar energy is absorbed by the water, creating positive feedbacks that lead to further melting. Such dynamics can change the temperature of ocean layers and impact ocean circulation by decreasing salinity (and therefore density). This disrupts the thermohaline current and effects the distribution of heat. This can alter regional climate and change marine habitats. Thermohaline circulation would weaken and slow down and temperatures (especially in the North Atlantic) would become cooler. Evidence from sedimentary rocks has shown that the last time it shut down was about 12,700 years ago during the Younger Dryas Event (ice age), causing temperatures to cool about 5 degrees on average. Scientists study the ratio of oxygen-16 and oxygen-18 in the calcium carbonate of fossils and how they vary in accordance to water temperature in order to study past climates. Data from ice core samples strongly suggests a relationship between rising atmospheric carbon dioxide levels and rising temperature. E5.4f Describe the geologic evidence that implies climates were significantly colder at times in the geologic record (e.g. geomorphology, striations, and fossils). Geomorphology- study of the form and structure of the earth. Scientific evidence exists that proves climatic conditions on the Earth were once much cooler. Glacial evidence is found in many locations on the Earth that now have very warm or temperate climates (like Africa). These pieces of evidence are: striations which indicate glacial movement over rocks (where they normally wouldn’t exist today) South Africa, India, South America- Has ancient glacial “scars”. Too warm of a climate to have glaciers today moraines that mark climate stability (glacial movement ceasing temporarily and then retreating) glacial erratics which show glacial boulders being transported long distances from their place of origin Another piece of geologic evidence is fossils found in locations where that organism couldn’t possibly exist indicating a different climate in the past. Because certain types of plants grow in particular climates, the discovery of plant fossils in areas that presently don’t support that type of plant provide evidence that a different (in this case, cooler) climate once existed there. EX: Wegener’s evidence for his theory of continental drift Spitsbergen- Norwegian Island has tropical plant fossils. Too cold of a climate today to have plant life. E5.4g Compare and contrast the heat-trapping mechanisms of the major greenhouse gases resulting from emissions (carbon dioxide, methane, nitrous oxide, and fluorocarbons) as well as their abundance and heat-trapping capacity. Natural Emissions Water Vapor- volcanic eruptions; evaporation of water; Most common/abundant greenhouse gas in the atmosphere (accounts for 60 to 70 percent of all greenhouse gases) As human activities produce more greenhouse gases in the atmosphere and temperature increases, so does the rate of evaporation, which will put more water vapor into the atmosphere. Artificial Emissions Carbon Dioxide- volcanic eruptions; decay of organic material; burning of fossil fuels (coal, oil, gasoline); burning of wood products; burning of waste; deforestation (release carbon into atmosphere and reduces the amount of atmospheric carbon dioxide that can be absorbed). We are releasing carbon dioxide faster than it can be absorbed and this is a problem. Scientists have analyzed air bubbles trapped in glaciers to compare atmospheric carbon dioxide from long ago to today’s current levels. The amount has increased by about 35 percent since 1750. Scientists predict CO2 levels will double or triple in the next 100 years if current trends don’t cease. CO2 can stay in the atmosphere for centuries. Methane- animal gas from digestion; decomposition of wastes (landfills); microorganisms breaking down organic matter (especially in damp soils like rice fields) Methane is 30 times more efficient at trapping heat than carbon dioxide Methane only stays in the atmosphere for about a century Atmospheric Methane has doubled since 1750 Nitrous Oxide- burning of fossils fuels and automobile exhaust; nitrogen fertilizers that release NO2 when breaking down the soil NO2 is 300 times more efficient at trapping heat than carbon dioxide NO2 stays in atmosphere for a century Atmospheric NO2 has increased by about 18 percent since 1750 Ozone- both naturally produced and artificially produced A component of smog- limited to industrialized regions Fluorocarbons (CFCs and HCFCs)- used in manufacturing processes; GREATEST heat-trapping capacity Several thousand times more efficient at trapping heat than carbon dioxide CFCs- aerosols, refrigerants Release chlorine into the upper atmosphere, which would destroy the protective ozone Ban on CFCs in developed countries, except for asthma inhalers Switched to use of HCFCs, but they still release chlorine into atmosphere. By 2030, these will no longer be used HFCs and PFCs do not destroy ozone, but are still powerful greenhouse gases Links Paleoclimate (climate from the past) http://www.ncdc.noaa.gov/paleo/ Carbon Cycle (E2.3A) Carbon Cycle Animation http://bcs.whfreeman.com/thelifewire/content/chp58/5802002.html Main sources of energy to the Climate System (E2.2D) http://www.solcomhouse.com/climatechange.htm E4.5A Power Point on Climate Change http://www.earth4567.com Info http://www.epa.gov/climatechange/ http://www.ecobridge.org/content/g_cse.htm http://www.esd.ornl.gov/projects/qen/transit.html Greenhouse Effect and Global Warming Animations http://www.epa.gov/climatechange/kids/global_warming_version2.html http://science.nationalgeographic.com/science/environment/global-warming/gw-overview-interactive.html http://encarta.msn.com/media_701765046_761578504_-1_1/Greenhouse_Effect.html http://www.bbc.co.uk/climate/evidence/greenhouse_effect_img.shtml GREAT Greenhouse Effect/Global Warming Simulation http://phet.colorado.edu/new/simulations/sims.php?sim=The_Greenhouse_Effect Global Warming Article (Time) http://www.time.com/time/magazine/article/0,9171,1176980,00.html Greenhouse Gases and Greenhouse Effect http://en.wikipedia.org/wiki/Greenhouse_gas Greenhouse Gases and Society http://www.umich.edu/~gs265/society/greenhouse.htm EPA on Greenhouse Gas Emissions http://yosemite.epa.gov/oar/globalwarming.nsf/content/emissions.html Greenhouse Effect Computer Lab Activity and Graphing Assignment http://teachertech.rice.edu/Participants/sboone/Lessons/Greenhouse/ Effects of Global Warming Info http://www.effectofglobalwarming.com/ E5.4B Volcanic Effects on Climate http://volcano.und.nodak.edu/vwdocs/frequent_questions/top_101/oldEffects.html http://www.cotf.edu/ete/modules/volcanoes/vclimate.html Explanation of Volcanic Haze Effect on Cooling Temperatures http://www.geology.sdsu.edu/how_volcanoes_work/climate_effects.html Info and Graph on Correlation Between Number of Sunspots and Temperature http://www.oar.noaa.gov/spotlite/archive/spot_sunclimate.html http://stars5.netfirms.com/suncli.htm Graph on Annual Number of Sunspots since 1700 http://www.ocs.orst.edu/ats210/sunspots.gif Image of Difference in Eccentricity http://www.geol.umd.edu/~jmerck/geol100/images/37/eccentricity.gif E5.4c CO2 emissions info and data http://cdiac.ornl.gov/trends/emis/meth_reg.htm CO2 emissions graphs http://www.eia.doe.gov/oiaf/archive/ieo01/images/figure_94.jpg http://www.guardian.co.uk/globalwarming/graphic/0,,397009,00.html Atmospheric CO2 concentration graphs http://www.omgfilms.com/spark1/images/stories/fruit/mauna_loa_carbon_dioxide.png http://www.aip.org/history/climate/images/maunaloa.jpg CO2 emissions AND Atmospheric CO2 concentration graph http://www.eia.doe.gov/oiaf/1605/ggccebro/chapter1.html Global Temperatures graphs http://cires.colorado.edu/~maurerj/scatterometry/global_temperatures.gif http://www.sightedmoon.com/wpcontent/uploads/2006/09/Average%20Global%20Temperature%201880-205.JPG http://alignment.hep.brandeis.edu/Hashemi/Climate/Global_Temp.gif Global Temperatures data table http://www.earth-policy.org/Indicators/Temp/Temp_data.htm#fig5 Atmospheric CO2 concentrations graph (at Mauna Loa, HI) http://www.grida.no/climate/vital/06.htm Atmospheric CO2 concentrations table (at Point Barrow, AK) http://www.fsl.noaa.gov/visitors/education/samii/SAMII_Act6_Table6.1d.jpg E5.4D Affects of Climate Change on Oceans http://www.usgcrp.gov/usgcrp/Library/nationalassessment/overviewcoastal.htm E5.4e Varves http://en.wikipedia.org/wiki/Varve Cryosphere and Ice Cap Animations http://www.nasa.gov/centers/goddard/earthandsun/climate_change.html Affects of Climate Change on Polar Ice (Sea Level Rise) http://www.usgcrp.gov/usgcrp/Library/nationalassessment/overviewcoastal.htm Affects of Melting Polar Ice on Regional Climate http://www.thewe.cc/weplanet/poles/thermohaline_circulation.htm http://www.windows.ucar.edu/tour/link=/earth/polar/icemelt_oceancirc.html E5.4g Greenhouse gases and their heat trapping capacity http://encarta.msn.com/encyclopedia_761578504/Greenhouse_Effect.html Good movie to watch: An Inconvenient Truth (Al Gore)