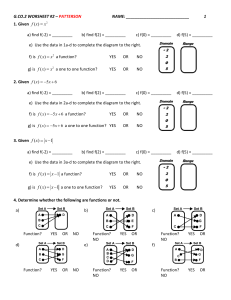
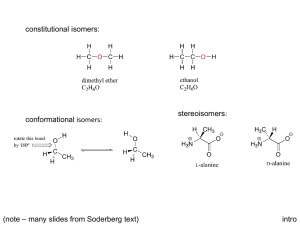
Organocatalysis, catalytic transformations with small organic
advertisement

Organocatalysis, catalytic transformations with small organic molecules, has found renewed interest in both academia and industry. Although it was already known for a century that organic compounds could catalyze asymmetric reactions, it is only since half a decade that the potential of organocatalysis is understood and is starting to be fully explored. In the treatment of diseases, one enantiomer of a medicine is usually more potent, and in the worst scenario, the opposite enantiomer can cause serious side effects or even death. Therefore, the demand from both pharmaceutical and chemical industry for new reliable asymmetric transformations of molecular skeletons is higher then ever. Nowadays, the construction of (highly) functionalized asymmetric skeletons still suffers from drawbacks, especially when there is a quaternary stereocenter involved in the target molecule. Organocatalysis gained popularity in a relatively short time span, because it has led to a large assortment of new asymmetric demanding transformations in the last five years. In some cases, organocatalysts meet the selectivity and efficiency levels of established metal catalyzed organic reactions. Since organocatalysis has a hidden potential, it could provide a solution for challenging alterations in the future. Moreover, organocatalysis can serve as a green alternative for transition metal catalyzed reactions. During this master research project in organocatalysis we found that the organocatalyst βisocupreidine (β-ICPD) effectively catalyzes a one-step asymmetric transformation of Morita-Baylis-Hillman carbonates. This transformation led to new molecular assemblies with vicinal quaternary and tertiary stereocenters. We observed high chemo-, diastereo-, and enantioselectivities with this reaction catalyzed by β-isocupreidine, in addition the chemical yields of these transformations are excellent. This one-step organocatalytic allylic alkylation is the first, out of six reactions reported, in which the reaction mechanism can not be only explained in terms of a conjugate addition and thereby leading to adjacent quaternary and tertiary stereocenters.