tpj12302-sup-0003-legends
advertisement

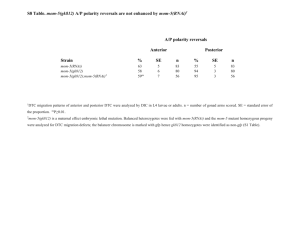
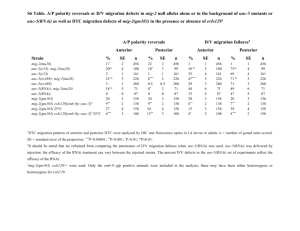
SUPPORTING LEGENDS Table S1. Accession numbers. Nicotiana benthamiana and tobacco are allotetraploid species. PES, BOP1, and WDR12 are single genes in Arabidopsis and in each diploid genome of N. benthamiana. Based on TIGR Gene Index (http://compbio.dfci.harvard.edu/tgi/), there are two nonoverlapping Expressed Sequence Transcripts (ESTs) of PES and WDR12 in tobacco, indicating that tobacco PES and WDR12 may be single genes. There are five ESTs for tobacco BOP1, and comparison of the sequences suggests that there are at least two BOP1 genes in each diploid genome of tobacco. N. benthamiana PES and BOP1 genes used in this study are underlined. Table S2. PCR primers used in this study. Figure S1. VIGS constructs, phenotypes, and suppression of NbPES transcripts. (a) Schematic drawing of NbPES structure and three VIGS constructs (F, N, and C) that contain different NbPES cDNA fragments, as indicated by bars. (b) Phenotypes of VIGS plants. The NbPES VIGS plants showed growth arrest and abnormal leaf development, as compared to the control TRV at 20 days after infiltration. (c) Real-time quantitative RT-PCR analysis of NbPES transcript levels in TRV:NbPES(F), TRV:NbPES(N) and TRV:NbPES(C) lines. The β-tubulin mRNA level was used as the control. Each value represents the mean standard deviation (SD) of three replicates per experiment. Asterisks denote statistical significance as follows: *, P ≤ 0.05; **, P ≤ 0.01. 1 Figure S2. Root phenotypes and gene silencing in the AtPES RNAi Arabidopsis seedlings. (a) Retarded root growth and death of the aerial tissues caused by AtPES silencing. The AtPES RNAi seedlings were grown on MS medium, and then transferred to (+)DEX medium for vertical growth. Scale bars = 9 mm. (b) Root length was measured in seedlings shown in (a) 3, 7, and 10 days after transfer. Each data point represents the mean standard deviation (SD) (n = 20 seedlings). Asterisks denote statistical significance as follows: *, P ≤ 0.05; **, P ≤ 0.01. (c) Relative AtPES transcript levels in the RNAi lines. Real-time quantitative RT-PCR analysis was carried out on WT and two RNAi lines (#28 and #38) after 3 and 6 days of DEX treatment. Seedlings were grown for 7 days on MS media, and transferred to media containing either ethanol (-DEX) or 10 μM DEX for either 3 or 6 more days of growth. Quantification of transcript levels, relative to the (-)DEX sample (at 3 days), is shown. The UBC10 mRNA level was used as the control. Each value represents the mean standard deviation (SD) of three replicates per experiment. (d) Relative AtPES transcript levels in the AtPES RNAi-10 line based on real-time quantitative RT-PCR. Seedlings were grown for 7 days on MS media, and transferred to media containing either ethanol (-)DEX or 10 μM DEX for 6 more days of growth. Figure S3. GVG transcript levels and DEX concentration-dependent phenotypes of AtPES RNAi seedlings. (a) GVG transcript levels in different AtPES RNAi lines. Semiquantitative RT-PCR analysis was carried out on WT (Col-0) and nine independent RNAi lines (#28, #38, #3, 2 #10, #12, #41, #47, #5, and #9). Seedlings grown for 12 days on MS media were used for RT-PCR using GVG-specific primers with amplification cycles as indicated. The UBC10 mRNA level was used as a control. (b) DEX concentration-dependent phenotypes. Two RNAi lines (#10 and #28) were grown 12 days on media containing DEX at the final concentration of 0-30 μM. RNAi28 seedlings showed severe growth retardation in response to 0.1-30 μM DEX, while RNAi-10 seedlings showed a growth defect only at 30 μM DEX. (c) Phenotypes of the AtPES RNAi-10 and RNAi-28 seedlings grown in the dark for 4 days on media containing ethanol (-DEX) or 0.1 μM DEX. Figure S4. Relative NtPES transcript levels in NbPES RNAi BY-2 cell lines. (a) Real-time quantitative RT-PCR analysis was performed 2 days after either ethanol (DEX) or 10 μM DEX treatment. The β-tubulin mRNA level was used as the control. Each value represents the mean SD of three replicates per experiment. Asterisks denote statistical significance as follows: *, P ≤ 0.05; **, P ≤ 0.01. (b) Real-time quantitative RT-PCR analysis was performed after 10 μM DEX treatment for 1 day or after 5 μM DEX treatment for 4 days. Figure S5. Visualization of AtPES interactions with BOP1 and WDR12 using bimolecular fluorescence complementation (BiFC). (a) Structural organization of Arabidopsis BOP1 and WDR12 proteins. BOP1 contains the BOP1 domain and six WD40 repeats. WDR12 contains the Notchless (NLE)-like domain and seven WD40 repeats. aa, amino acids. (b) For BiFC, AtPES, BOP1, and WDR12 were co-expressed as either YFPN or YFPC 3 fusion proteins in N. benthamiana leaves using agroinfiltration. As a control, AtPES:YFPN and YFPC, and YFPN and AtPES:YFPC were co-expressed. Protoplasts, prepared from the infiltrated leaves, were observed for YFP fluorescence by confocal laser scanning microscopy. (c) Localization of the YFP signal in the nucleolus. DAPI staining indicates a nucleus. Figure S6. Time-course analyses of protein translation. The AtPES RNAi seedlings (#10 and #28) were treated with either ethanol for 4 days (-) or DEX for 2, 3, and 4 days. After 35S-methionine labeling of the seedlings, total protein extracts were separated by SDS-PAGE, and the gel was Coomassie-stained briefly to show the total cellular proteins. After destaining, the gel was dried and analyzed with a phosphorimager. Protein molecular weight markers (MW) are shown. The rbcL protein band was marked with an asterisk. Appendix S1. Supporting methods and references. The experimental procedures used in this study are described in detail. 4