Copper cycle lab
advertisement

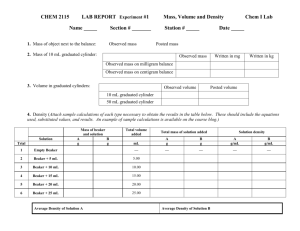
Chemistry 2015-2016 Copper Cycle Lab Procedure Name: Date: Per: Day 1: 1. Label your beaker with your period and group number 2. Weigh out 0.25 grams of solid copper. Record the precise mass in your lab notebook 3. In the fume hood, with the hood running, carefully measure and add 5 mL of nitric acid. 4. Record your observations. 5. Place your beaker in fume hood #1 over night. ---------------------------------------------------------END OF DAY ONE----------------------------------------------------Day 2: 1. Pick up your beaker out of the fume hood. Make sure you have your own beaker! 2. Add approximately 10mL of distilled water (dH2O). 3. Slowly add 5 mL of sodium hydroxide (NaOH) to the beaker. Be sure to look for any solid that begins to appear scattered throughout the liquid. 4. Record your observations. 5. Heat the beaker with a Bunsen burner on a double ring stand. Stir with a glass stir rod while heating. Continue heating until a solid appears (this may take 10 or more minutes). Always be careful when heating chemicals – they will burn exposed skin more quickly when hot! Be especially careful not to splash any of the chemicals on your skin while you are stirring the contents of the beaker. 6. Turn off the flame and allow the beaker to cool. While the beaker is cooling, record your observations. Move the beaker from the ring stand to fume hood #1 overnight. -----------------------------------------------------END OF DAY TWO--------------------------------------------------Day 3: 1. Pick up your beaker out of the fume hood. Make sure you have your own beaker! 2. Record your observations. 3. Carefully decant the liquid above the precipitate into a waste beaker as seen in the diagram. 4. Add about 7 mL of 6 M sulfuric acid (H2SO4) slowly while stirring. As you add the acid, observe any changes in the liquid and the solid. 5. Record your observations. 6. Measure an amount of zinc equal to the amount of copper you measured in day 1 and add the zinc to the beaker. Stir the solution until colorless. 7. Record your observations. 8. Filter the solution with a funnel and a carefully weighed piece of filter paper (weigh to the 0.01 g). Allow the filter paper and contents to dry overnight. You will weigh your final product the next day. In your notebook, create a data table that has 2 columns and 8 rows (plus titles). The first column should read, “what I did” and the second column should read, “what I observed.” Fill in the actions you will do in the first column (hint: look at when you’re recording observations to help you decide what to write). Throughout the lab, record your observations in this column. Chemistry 2015-2016 Copper Cycle Lab Analysis Name: Date: Per: Directions: Answer the following in your notebook. When possible, use complete sentences. 1. Write out the chemical formula for the following compounds and elements, including states of matter as used in this lab. a. Copper b. Nitric acid c. Sodium hydroxide d. Sulfuric acid e. Zinc 2. What is a possible product containing copper when the following reactants combine? Write out the name and chemical formula. a. Copper and nitric acid (day 1 – hint: look at the warm-up on this day) b. Copper nitrate and sodium hydroxide (day 2 step 3 – hint: this is a double replacement reaction) c. Copper oxide and Sulfuric acid (day 3 step 4 – hint: this is a double replacement reaction) d. Copper sulfate and zinc (day 3 step 6 – hint: this is a single replacement reaction) 3. Why can each reaction be called a conversion? 4. What was always present? 5. How was it converted? 6. What is the purpose of decanting (day 3 step 3)? 7. List two reactions that produced a precipitate and write out the reactants. 8. Which reaction produced heat? 9. What was evidence for a chemical reaction: a. In reaction 1 when you added nitric acid? b. In reaction 2 when you added sodium hydroxide? c. In reaction 3 when you added heat? d. In reaction 4 when you added sulfuric acid? e. In reaction 5 when you added zinc? 10. At the beginning of your reaction you started with a certain amount of pure copper, and you should have ended up with that same amount at the end of your experiment. Calculate your percent error for this experiment (initial mass is your theoretical, final mass is experimental). 11. What are some possible sources of error in your lab? Be specific!