COURSE INFORMATION FORM Course Code and Title KIM 117
advertisement

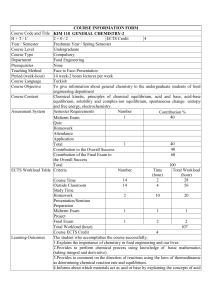
COURSE INFORMATION FORM Course Code and Title KIM 117 GENERAL CHEMISTRY-1 H+T/C 2+0/2 ECTS Credit 4 Year / Semester Freshman Year / Fall Semester Course Level Undergraduate Course Type Compulsory Department Food Engineering Prerequisites None Teaching Method Face to Face-Presentation Period (week-hour) 14 week-2 hours of lectures 2 hours of practical per week Course Language Turkish Course Objective To give information about general chemistry to the undergraduate students of food engineering department Course Content Measurement and properties of matter, atoms and atomic theory, chemical compounds, chemical reactions, introduction to the reactions of aqueous solutions, gases, thermo chemistry, liquids, solids and intermolecular forces. Assessment System Semester Requirements Number Contribution % 1 20 Midterm Exam Quiz Homework Attendance 14 20 Application 15 Total 40 Contribution to the Overall Success Contribution of the Final Exam to 60 The Overall Success Total 100 ECTS Workload Table Criteria Number Time Total Workload (hour) (hour) 14 2 28 Course Time Outside Classroom 14 5 70 Study Time Homework Presentation/Seminar Preparation Midterm Exam 1 1 1 Project Final Exam 1 2 2 Total Workload (hour) 101 Course ECTS Credit 4 Learning Outcomes The student who accomplishes the course successfully; 1.Explains the importance of chemistry in food engineering and our lives 2.Provides to perform chemical process using knowledge of basic mathematics (taking integral and derivative). 3.Defines atom, mole, Avogadro number and matter forming a basis of chemistry. 4.Informs about periodic table and properties. 5.Describes chemical reactions 6.Informs about solids, liquids and gases 7.Informs about thermo chemistry 8.Explains how we encounter with all this in formations in our current lives. Course Stream Course Sources Contribution of the Course to Department Learning Outcomes Week Education Modules Preliminary Documents Studies 1. Properties of Matter and Measurement Presentation Proposed Preparation Sources 2. Atoms and Atomic Theory Presentation Proposed Preparation Sources 3. Presentation Proposed Chemical compounds Preparation Sources 4. Presentation Proposed Chemical Reactions Preparation Sources 5. Introduction to Reactions in Aqueous Presentation Proposed Solutions Preparation Sources 6. Presentation Proposed Gases Preparation Sources 7. Electronic Structure of the Atom Presentation Proposed Preparation Sources 8. Mid-Term Exam 9. Thermochemistry Presentation Proposed Preparation Sources 10. Periodic Table and Some Properties of Atoms Presentation Proposed Preparation Sources 11. Basic Concepts of Chemical Bonding Presentation Proposed Preparation Sources 12. Theories of Chemical Bonding Presentation Proposed Preparation Sources 13. Solids, Liquids, and Intermolecular Forces Presentation Proposed Preparation Sources 14. Solutions and Chemical Properties Presentation Proposed Preparation Sources Presentation Proposed 15. Solutions and Chemical Properties Preparation Sources 1.Petrucci, R. H., Harwood, W.S. and Herring, F.G. 1993; General Chemistry 1, Palm Publishing. 2.Mortimer, C.E. 2004. Modern University Chemistry 1, Çağlayan Books. 3.Alpaydın, S. and Simsek, A. 2009, General Chemistry, the Nobel Publishing. Course Learning Outcomes Dep. Learn. Outcomes 1.Explains the importance of chemistry in food engineering and our 1, 4 lives. 2.Provides to perform chemical process using knowledge of basic 2, 11 mathematics (taking integral and derivative). 3.Defines atom, mole, Avogadro number and matter forming a basis 2 of chemistry. 4.Informs about periodic table and properties. 2, 11 5.Describes chemical reactions 1, 2, 11 6.Informs about solids, liquids and gases 1, 11, 12 7.Informs about thermo chemistry 10, 11, 12 8.Explains how we encounter with all this in formations in our 6 current lives. Course Coordinator(s) Assist. Prof. Cemalettin BALTACI, Assist. Prof. Sevim Beyza ÖZTÜRK SARIKAYA, Assist. Prof. Ali GÜNDOĞDU