Unit 4 note A`s
advertisement

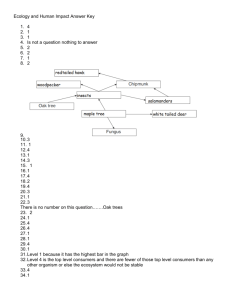
Unit 4 Carbon Chemistry Ch. 9 Carbon Chemistry Objectives: 1. State the relationship between the number of carbon atoms in, and the properties of, a hydrocarbon. 2. Describe the difference between the three types of unsaturated hydrocarbons. 3. Describe the different fossil fuels that are composed of hydrocarbons. 4. Distinguish between the different types of synthetic and natural polymers. 5. Understand the use and importance of synthetic materials, which came from natural resources, in today’s society. Vocabulary Organic solid Network solid Hydrocarbon Saturated hydrocarbon Alkane Unsaturated hydrocarbon Alkene Alkyne Aromatic hydrocarbons Fossil fuels Polymer Monomer Glucose Carbohydrates Cellulose Nucleic acids Amino acid Protein 9.1 Carbon Compounds Read p. 262-269 and make notes by answering the following questions. Describe the three types of fossil fuels. - Coal – began as plants in swamps about 300 MYA; pressure changed the plants to a mixture of mostly aromatic hydrocarbons with a high ration of carbon to hydrogen; they produce more soot than other fossil fuels; used to produce electricity and causes acid rain - Natural gas – formed from marine organisms; composed of small alkane chains; used in heating, cooking, and some electrical generation; always found with coal or petroleum - Petroleum – formed from marine organisms; usually a thick liquid composed mainly of long-branched alkanes and alkenes; once separated into its components, it’s used for gasoline, heating oil, and manufacturing of products (plastics) Describe a combustion reaction. - Hydrocarbon (fossil fuel) + oxygen gas = carbon dioxide and water What is the difference between complete combustion and incomplete combustion? - When not enough oxygen is present, the hydrocarbon does not break down fully, and carbon monoxide is formed rather than carbon dioxide, which is formed in complete combustion. How does burning fossil fuels add to the problem of acid rain? - Carbon dioxide dissolves in water and forms carbonic acid. Sulfur dioxide and nitrogen oxides, released from burning coal, form sulfuric acid and nitric acid, when dissolved in water. These acids damage metal, stone, and harm aquatic organisms. 9.3 Polymers Read p. 275-280 and make notes by answering the following questions. What is a synthetic polymer? Describe three examples. - They are developed by chemists in labs and factories - Rubber – made naturally but synthetic is cheaper, more readily available, and of better quality; made from petroleum; used in tires and adhesives - Nylon – made to replace silk; nylon is very strong, durable, and shiny; used in clothing, fishing line, carpets, ropes, outdoor coverings - Polyethylene – plastic; chains of an alkene; more carbons the harder the plastic is What is a natural polymer? Describe three examples. - Large molecules produced by organisms Starches – sugars (glucose, fructose, sucrose); produced in plants; formed from carbohydrates Cellulose – fiber (cotton and wood); formed from starches; cellulose gives strength to plant stems; most animals cannot digest it Nucleic Acids – DNA and RNA; found in all cells and used to store information