Interaction of Nanoparticles with Microbial Biofilm in Water Treatment
advertisement

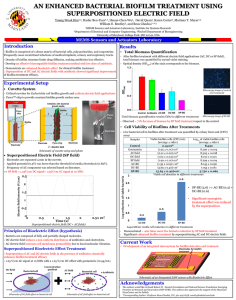
FINAL TECHNICAL PAPER Interaction of Nanoparticles with Microbial Biofilm in Water Treatment Facility Processes Submitted To: The 2014 Summer NSF REU Program Sponsored By: The National Science Foundation Grant ID No.: DUE-0756921 and EEC-1004623 College of Engineering and Applied Science University of Cincinnati Cincinnati, Ohio Prepared By: Victoria Sumner – Junior, Chemical Engineering, University of Cincinnati Stephanie Palmer – Pre-Junior, Chemical Engineering, University of Cincinnati Dorien Clark – Sophomore, Chemical Engineering, University of Cincinnati Report Reviewed By: Margaret J. Kupferle, PhD, PE REU Faculty Mentor Program Chair, Environmental Engineering and Associate Professor of Environmental Engineering George A. Sorial, PhD REU Faculty Co-Mentor Head, Department of Biomedical, Chemical and Environmental Engineering and Professor of Environmental Engineering Hengye Jing REU Graduate Research Assistant, Environmental Engineering August 1, 2014 Abstract Providing access to clean water is a growing international concern and is one of the Grand Challenges of Engineering recently put forth by the National Academy of Engineering (Committee of National Academy of Engineering, 2012). Water treatment processes are impacted by biofilms. Biofilms are any group of microorganisms in which cells stick to each other on a surface. When sufficient nutrients for growth are present, biofilm will grow and form a protective matrix that can harbor harmful bacteria and is hard to disinfect. This presents problems in water distribution pipes at water treatment facilities. Chlorination of water in the water treatment system is used to kill pathogens within the water and provide protection from bacterial contamination as the water is pumped through the distribution system. However, the use of chlorine can be harmful to people in the long run due to the formation of carcinogenic disinfection by-products. Alternative ways to decrease the use of chlorination and control the growth of biofilm in water treatment distribution systems by looking at its interaction with different piping materials and silver nanoparticles were studied. Pipe material impacted biofilm growth with copper encouraging the most growth and stainless steel the least. Silver nanoparticles sorbed to the biofilm quickly and were able to effectively kill biofilm on the coupon materials. This suggests they could be used in future applications to control biofilm, such as a coating inside pipelines in distribution systems. Key Words: biofilm, water distribution, silver nanoparticles, disinfection byproducts, adsorption 1 1. Introduction. Providing access to clean water is a growing international concern and is one of the Grand Challenges of Engineering recently put forth by the National Academy of Engineering (Committee of National Academy of Engineering, 2012). Water treatment processes are impacted by biofilms. Biofilms, also known as microbial aggregates, are microorganisms that accumulate on a solid–liquid interface and are encased in a matrix of highly hydrated extracellular polymeric substance (EPS). EPS mainly consists of polysaccharides, proteins, uronic acids, humic acids, DNA, and cell fragments (Späth, et al. 1998). Other forms of biofilm are plaque that forms on teeth, algae which are present on rocks in lakes and rivers, and can contain bacteria, archaea, protozoa, and fungi. It can form inside of medical equipment, such as prosthetics or knee joints; it can also infect and form mucus in the lungs causing cystic fibrosis. Biofilm grows within water treatment facilities as well as distribution pipes. Because of the negative effects of biofilm growth on certain surfaces, its removal has been a topic of study and research for quite some time. It is difficult to remove due to the formation of a protective layer that is composed of EPS and water channels that serve as a permeable barrier. (Vollmer et al., 2008; Sheng and Liu, 2011; Stewart and Franklin, 2008; SahleDemessie and Tadesse, 2011; Vu et al., 2009). Biofilm also has beneficial applications; it has been used successfully in water and wastewater treatment for well over a century (Cunningham, 2001-2010). If biofilm growth is consistently controlled and regulated in specific environments, then it can be used for positive applications such as these. As previous studies have shown, controlling where and how fast it grows is challenging. 2 The water treatment process starts with the inlet feed from a water source, like a river, flowing into holding reservoirs, where enough water for several days can be stored and some settling of large particles occurs. Next, chemical flocculation encourages more clumping of smaller particles for more solids to be removed in secondary sedimentation. Then sand filtration removes any remaining particulates that got through the secondary sedimentation process. Granular activated carbon adsorption and UV disinfection may be used to remove organics that remain in the water. Finally, a disinfectant such as chlorine is added through the clear wells to keep the water safe and kill any pathogens as it is distributed to the consumers via the distribution pipelines. Biofilm can grow in these pipes decreasing the quality of the drinking water that was just treated. Biofilm can protect and shelter pathogens such as parasites, bacterial pathogens and intestinal viruses, which can be released into water systems causing the deterioration of the quality of drinking water. Currently, the majority of drinking water treatment facilities, including Greater Cincinnati Water Works, add chlorine to drinking water to protect water from pathogens during distribution to consumers. This reduces the amount of acute or immediate health risk due to microbial contamination, but actually creates chronic or long term health risk to consumers due to formation of disinfection byproducts (DPB’s) when chlorine reacts with organic materials in the water. A major challenge that water utilities face is how to control and limit the risks from pathogens and disinfection byproducts to minimize health risks to the population (U.S. Environmental Protection Agency, 2013). Trihalomethanes, haloacetic acids, bromate and chlorite are the main DBP’s that are created in distribution pipelines due to the interaction of organic materials and chlorine. Some people who drink water containing DBP’s in excess of the maximum containment level (MCL) over many years could experience 3 liver, kidney, or central nervous system problems and increased risk of cancer (U.S. Environmental Protection Agency, 2013). Ways to decrease the need for chlorination to control microbial growth are of great interest to researchers. Choice of pipe materials and the impact of nanoparticles are of interest in this application. Different pipe materials have different roughness and surface charge properties that may impact biofilm growth. They also may release toxic or stimulating substances into the water that either kill or stimulate growth of biofilms. The coupon materials tested for inhibition or stimulation of the growth of Pseudomonas fluorescens biofilm in this study included copper, stainless steel, polyvinyl chloride (PVC) and polyethylene (PE). Nanoparticles occur in everyday life in products such as makeup, coatings of metal products and medical equipment. They are tiny particles that range from 1 to 100 nanometers in size. An increasingly common application is the use of silver nanoparticles for antimicrobial coatings, and many textiles, keyboards, wound dressings, and biomedical devices now contain silver nanoparticles that continuously release a low level of silver ions to provide protection against bacteria (Olderburg, 2014). Due to the increasing prevalence of Ag-NPs in consumer products, there is a large international effort underway to verify silver nanoparticle safety and to understand the mechanism of action for antibacterial effects. In this project, adsorption and antibacterial effects of silver nanoparticles (Ag-NPs) were studied to understand more about how Ag-NPs interact with Pseudomonas fluorescens biofilms. Adsorption is the physical adherence or bonding of ions and molecules onto the surface of another phase. It is important to know if nanoparticles adsorb to the biofilm or pass through the system. If they are adsorbed to the biofilm, they may kill the bacteria in the 4 biofilm given their reported antimicrobial effects. Some antibacterial effect studies indicate that Ag-NPs will be highly toxic to natural bacterial communities if/when they reach the environment. Studies have shown that Ag-NPs toxicity is size dependent at low concentrations. One study observed that silver nanoparticles of 1-10 nm were preferentially bound to cell membranes and were incorporated into bacteria, whereas larger nanoparticles were not, while others indicated that toxicity was shape dependent. Additionally, it has been shown that Ag-NPs caused pitting or corrosion on bacterial cell membranes, leading to increased permeability and cell death (Fabrega, et. al 2009). A potential use of nanoparticles in water systems is as a coating inside of the distribution pipes to reduce biofilm growth if they prove to effectively decrease biofilm growth in data results. Pseudomonas fluorescens, a gram-negative rod shaped type of bacteria, commonly found in decaying organic material such as leaves, soil, plants and water surfaces, was used in this study because it commonly occurs in water and grows quickly at room temperature. It was obtained from MicroBiologics, Inc. (St. Cloud, MN). It is gram-negative because its EPS mainly contains phosphoryl groups along with the polysaccharides. The negative charge may impact biofilm growth on different pipe materials with different surface charges and adsorption of nanoparticles to biofilm, depending on the charge of the particles. 5 The goals of the research were to: determine which materials will be best for preventing biofilm growth in pipelines in water distribution systems, understand the anti-bacterial effect of nanoparticles on biofilm and how they can be used to prevent biofilm growth in the environment, and learn and understand specific research methods and procedures with hands on experience. 2. Materials and Methods Biofilm Growth and Sampling: Biofilm reactors (see Figure 1) were used to grow Pseudomonas fluorescens while feeding it carbon and nitrate nutrient solutions. Carbon and nitrate nutrient solutions were pumped into the biofilm reactors at 1 mL/min. Nutrient solutions had a pH around 7.5 to 8. For the biofilm reactor, a 1000-mL glass container with a polyethylene lid that holds eight polypropylene coupon holders was used. A stirring vane at the bottom of the reactor provided continuous mixing. To simulate biofilm growth in pipelines, the biofilm was grown on four different types of coupons in one of the reactors. The two additional reactors containing polycarbonate coupons were used for kinetic sorption and antibacterial effect studies. 6 Figure 1. Basic Biofilm Reactor Set-up Pipe Materials Study: Biofilm growth on different coupon materials was observed by removing coupon samples weekly for testing. The coupon materials chosen for this study were: copper, stainless steel, polyvinyl chloride (PVC), and polyethylene (PE). Four separate holding vials (one for each coupon material) were prepared with 10 mL of carbon solution and 10 mL of nitrate solution so the biofilm on the coupon materials remained in the same chemical environment prior to staining. A Laser Scanning Microscope (LSM) (Zeiss LSM710, Carl Zeiss) at the U.S. EPA was used to measure numbers of dead and live cells from coupon samples in both 2D and 3D. Each coupon was placed in a Petri dish and staining solution, also known as the BacLight Live/Dead bacterial viability kit (Life Technologies Corporation), was used to stain the samples. The stained coupons were observed using the Laser Scanning Microscope and the thickness of biofilm on each material was recorded. Three sets of 7 pictures were taken of the current biofilm growth and decay from each week and the images were analyzed for live/dead pixels per standard surface area of 2120 µm x 2124 µm. Surface Zeta-Potential Analysis: The Zeta-Potential Analyzer was used to determine the overall surface electrical charge of the biofilm and the piping materials at the U.S. EPA. A 0.001 M potassium chloride solution was made for the electrolyte solution used in the analysis. The analyzer then was rinsed with deionized water and the pH meter was calibrated with solutions at 4, 7, and 10. The conductivity was calibrated at 1413 µs/cm and the conductivity constant was measured to be 1.109 cm. After putting the electrolyte solution in the container (450 mL), the first material, copper (see Figure 2), was analyzed. Before running the machine, the gage gap was checked to make sure that the pressure was around 300 mbar and the flow rate was around 100 mL/min. The flow check needed to have a slope that was linear with no spikes before running titration. The titration was run from a pH of 2 to 11 using potassium hydroxide and the titration was run from a pH of 12 to 2 using hydrochloric acid. These steps were repeated with stainless steel, polyvinyl chloride, and polyethylene. 8 Figure 2. Picture of a copper coupon materials being placed to be run on the Zeta-Potential Analyzer. Scanning Electron Microscope: A JEM7600F (JEOL USA Peabody, MA) Scanning Electron Microscope (SEM) was used to scan the surface of the biofilm on the materials. Christina Bennett-Stamper at the U.S. EPA took the images using the SEM. 1. 2. Figure 3. (1) Scanning Electron Microscope surface image of Pseudomonas fluorescens with citrate reduced nanoparticles magnified to 3,500, with a depth of 1 µm. (2) Scanning Electron Microscope surface image of Pseudomonas fluorescens with citrate reduced nanoparticles magnified to 8,500, with a depth of 1 µm. Silver Nanoparticle (Ag-NP) Synthesis and Purification: Sodium citrate (Na3C6H5O7) was used as a reducing agent to separate the silver in a silver nitrate (AgNO3) to synthesize the silver nanoparticles (AG-NPs). Specifically, a solution of 1x10^-3 M AgNO3 (99.99%) was mixed with a solution of 1x10-2 M Na3C6H5O7 ·H2O (99+%) in a volume ratio of 2:1, respectively. The mixture was heated at 70 .C for 4 hours in a water bath. The color of the solution changed from clear to a tan color. After the first hour, the solution had to be checked every hour for aggregation. A Tangential Flow Filtration system (see Figure 4) used to purify 9 the Ag-NP from ions in the matrix. The ions were separated as permeate into the waste container and the pH of the retentate Ag-NP solution was lowered to ~5. Figure 4. Tangential Flow Filtration Set-up: was used to purify the previously synthesized silver nanoparticles. Kinetic Sorption Study: For the kinetic sorption study, the silver nanoparticle solutions were prepared at 3 different pH levels (5, 7.5 and 9) for 7 different time intervals or 21 in total. Polycarbonate coupons from the second biofilm reactor were dropped into the pH-adjusted Ag-NP solution at time zero. The coupons were allowed to sit undisturbed in Ag-NP solution and at designated sampling times, a coupon was carefully removed with tweezers from a given sample and the remaining solution was analyzed using a UV-visible spectrometer. Absorbance was measured at 30, 60, 120, 180, 240, 300, and 360 minutes. Absorbance was 10 related to Ag-NP concentration using a standard curve and reported as a function of time for each material. Antibacterial Effect Study: The silver nanoparticle solution was diluted in half with deionized water in a 1 L volumetric flask and the diluted solution was placed in a clean empty biofilm reactor container. The biofilm coupon lid with polycarbonate coupons from one of the growth reactors was placed in the new biofilm reactor filled with nanoparticle solution. After designated time increments, coupon samples were removed and biofilm growth was observed at times of 30, 60, 120, 180, 240, 300, and 360 minutes using the Laser Scanning Microscope as described previously. 3. Results and Discussion Pipe Material Study: A Zeta-Potential Analyzer, Scanning Electron Microscope, and Laser Scanning Microscope were used to study the impact of pipe material on biofilm growth. Zeta-Potential Analysis: The overall surface charge can help determine how readily biofilm organisms will attach to the different materials. Data plotted in Figure 5 show that the zeta potential of polyvinyl chloride is more positive around the pH of 7.5 and all the other materials are less than zero. The pH of 7.5 was chosen as a reference point because it is the pH used to grow the biofilm. Speculations can be made that the negatively charged biofilm could have attracted to the positively charged PVC, which could explain the medium to high amount of biofilm growth on the coupons. 11 Copper Stainless Steel Polyvinyl Chloride Polyethylene Polycarbonate P. fluorescens Biofilm Figure 5. A graph of the surface charge or zeta-potential of copper, stainless steel, polyvinyl chloride, polyethylene, polycarbonate, and the P. fluorescens biofilm at different pH levels. Results for pH level 7.5 are of interest since that was the pH of the nutrient solution. Scanning Electron Microscope: Representative Scanning Electron Microscope photos of the bare surfaces of the piping materials used in this study are shown in Figure 6. These images were used to explore surface roughness and to help explain biofilm formation. Note the large crevasses on the surface of copper, which could be one reason why it had high biofilm growth. It also has an electrochemical structure that could have affected the biofilm decay. In general, the polymer materials were smoother than the metals. The rough coefficients found in the literature were all the same due to the way they were manufactured. 12 1. 2. 3. 4. Figure 6. Scanning Electron Microscope surface images of the copper coupon (1), stainless steel coupon (2), polyvinyl chloride (3), and polyethylene (4). Laser Scanning Microscope: The Laser Scanning Microscope was used to record the amount of live cells versus dead cells per coupon area each week. A sample image for Week 4 on copper is shown in Figure 7A. A 3D plot of the thickness of the biofilm grown is shown in Figure 7B. Figure 8 summarizes the data for the number of live cells for each week. In general, the amount of green or alive biofilm organisms declined each week. For copper, the growth was high for the first week, but declined each week. Speculations could be made that this was due to the toxicity of its ions killing the biofilm. For the stainless steel, there was not much growth at all. For PVC, there was medium to high growth, but was patchy as seen by the error bars. For PE, it had a decreasing trend like 13 copper, but the 5th week, there was a huge increase in biofilm growth. This could be due to its structural properties. A. B. Figure 7. A. 2D representation of biofilm growth on a copper coupon from week 4. B. 3D graph of the biofilm growth on a coupon showing its thickness. Figure 8. Graph shows a plot of the average biofilm growth on all of the material coupons over a five week period. Summary of Findings: Results of this study are summarized in Table 1. For copper, high biofilm growth was expected due to its rough surface, large crevasses, and corrosive 14 behavior. Stainless steel and PVC were expected to have low biofilm growth due to their smooth surface. PE was expected to have high growth of biofilm due to its ability to release nutrients like carbon for the biofilm to feed on. Copper and stainless steel proved to have the amount of biofilm growth that was expected due to properties of their surfaces, although the biofilm on the copper decreased each week. The biofilm growth could have decreased due to the toxicity of its ions being released. The PVC coupon however, had high biofilm growth, but was patchy. PVC had a positive surface charge at the pH used for the study, so this may have increased growth of the gram-negative bacteria used. There was a decreasing trend for the growth of biofilm on PE as well, until the 5th week. Speculations could be made that the growth was due to its ability to give off carbon as a nutrient for the biofilm to eat. These results could have been affected by the set-up of the experiment however all four materials were in the same biofilm reactor so the release of ions from copper or the carbon from the PE may have impacted the results. A future study separating coupon material by reactor would help discover if this was an effect. Table 1. 15 Nanoparticle Study: In addition to nanoparticle synthesis, two studies were conducted to explore nanoparticle interactions with biofilm, a kinetic sorption study to see how quickly they accumulate in biofilm and an antibacterial effect study to see if they can be used to control biofilm. Kinetic Sorption Study- Sorption happened when the biofilm was suspended in the silver nanoparticles solution. As seen in Figure 9, most of the sorption occurred in the first 30 minutes of contact. As time progressed, it reached equilibrium implying that the biofilm sorbed as much silver nanoparticles as it could. Figure 9. Graph shown is a plot of the concentration of the nanoparticles after they’ve interacted with the biofilm over time. 16 Anti-Bacterial Effect Study- The Ag-NP solution proved to kill the biofilm over time. A lot of biofilm was able to grow on the materials, but they all had a trend of decreased growth and having an increase of dead cells. A. B. Figure 10. A. This is a 3D graph shown in 2D of the live cells of the biofilm over 6 hour period of time. B. This is a 3D graph shown in 2D of the dead biofilm over a 6 hour period of time. 4. Conclusion Conclusions can be made about what effect different materials have on the growth of biofilm (piping material study), and about how biofilm interacts with silver nanoparticles (nanoparticle study) from the data we collected over the past 7 weeks. For the piping material study, it was found that stainless steel performed the best at preventing biofilm growth. However due to its high cost, stainless steel is uneconomical to use in a water distribution system but it may be useful in limited applications such as hospitals or in the food industry. Copper performed very poorly and recorded the highest biofilm growth over the 5 weeks of sampling. This could be partly because the large crevasses that are found on its surface from the Scanning Electron Microscope results; a rough surface structure allows the biofilm to attach to the surface of the material. The last two materials that were tested 17 gave unexpected results. From literature review, Polyvinyl Chloride (PVC) was expected to have a low growth of biofilm, due to a smooth surface structure, and Polyethylene (PE) to have a high growth of biofilm, due to nutrients on the surface of the material. In the beginning weeks, PVC recorded medium growth of biofilm on its surface, which may have been related to its positive surface charge at pH 7.5. The biofilm had a negative charge, which could explain why they were attracted to each other. For PE, the amount of biofilm on the surface grew dramatically in the later weeks of sampling. This could have happened because it takes time for the carbon chains on the surface of the coupon to break down, and provide nutrient for the biofilm. All of the data collected from the piping material study show that stainless steel would be very good for a water distribution system if the cost was not so high. PVC (which is currently used in pipelines), would be the next best option because it is so cost effective and can prevent biofilm well enough. The second major study conducted during the program was the nanoparticle study. This study was to show how biofilm interacts with silver nanoparticles. This study was comprised of two main parts: a kinetic sorption study and an antibacterial effect study. The size distribution of the nanoparticles was also examined and it was observed that pH affects the size of silver nanoparticles and that slight aggregation occurs within the first 30 minutes. The kinetic sorption study was performed to see how the biofilm sorbed and released the silver nanoparticles over time. From the data collected, it was observed that biofilm does, in fact, absorb silver nanoparticles with most of the uptake occurring in the first 30 minutes. There was some variance in the data possibly be due to coupon variance. The antibacterial effect study was conducted to visualize if the silver nanoparticles killed the biofilm once it was absorbed. The Laser Scanning Microscope was used to visualize the thickness of live and 18 dead biofilm cells over time. As time progressed, the thickness of biofilm cells decreased, which show that the nanoparticles had predicted the anti-bacterial effect. There was a decrease in live biofilm cells and a steady increase in dead biofilm cells over a time period of 6 hours. The data collected about the nanoparticles show that silver nanoparticles have an antibacterial effect on biofilm. This work can be used to study the application of nanoparticles in water distribution systems, such as coatings on pipes. 6. Acknowledgements We would like to thank to the National Science Foundation for funding our project via two grants (DUE-0756921 and EEC-1004623), the U.S. Environmental Protection Agency for allowing us to use their facilities and equipment, and Christina Bennett-Stamper at U.S. EPA for Scanning Electron Microscope photographs. 19 5. References. 1. Committee of National Academy of Engineering. (2012). “Provide access to clean water.” National Academy of Engineering, <http://www.engineeringchallenges.org/cms/8996/9142.aspx> (July 28, 2014). 2. Flemming, H-C., and Wingender, J. (2010). "The biofilm matrix." Nature Reviews|Microbiology, 8, 623-633. 3. Vollmer, W., Blanot, D., De Pedro, M.A. (2008). "FEMS Microbiol." Rev. 32, 149–167. 4. Späth, R., Flemming, H-C., and Wuertz, S. (1998). "Sorption Properties of Biofilms." Wat. Sci. Tech. 37(4-5). 207-210. 5. Strathmann, M., Leon-Morales, C., Flemming, H.-C. (2007). "Influence of biofilms on colloid mobility in the subsurface." In: Frimmel, F., Kammer, F., Flemming, H.-C. (Eds.), Colloidal Transport in Porous Media. Springer, Berlin Heidelberg, pp. 143–173 6. Jing, H, Mezgebe, B., Hassan, A. A., Salhe-Demessie, E., Sorial, G., Bennett-Stamper, C. (2014). “Experimental and modeling studies of sorption of ceria nanoparticle on microbial biofilms.” Biosource Technology, 161, 109-117. 7. Cunningham, A. B., Lennox, J. E., Ross, R. J. “Introduction to Biofilms: Section 5 What is Their Impact?” <http://www.cs.montana.edu/webworks/projects/stevesbook/contents/chapters/ chapter001/section005/green/page001.html> Eds. 2001-2010. (accessed June 20, 2014) 16. U. S. Environmental Protection Agency. Basic Information about Disinfection Byproducts in Drinking Water: Total Trihalomethanes, Haloacetic Acids, Bromate, and Chlorite. Washington D. C.: Government Printing Office, 2013. 17. Oldenburg, S. J., (2014). “Silver Nanoparticles: Properties and Applications.”<http://www.sigmaaldrich.com/materialsscience/nanomaterials/silver-nanoparticles.html>Sigma-Aldrich Co. January 9, 2013. 18. Fabrega, J., Fawcett, S. R., Renshaw, J. C., Lead, J. R. (2009). “Silver Nanoparticle Impact on Bacterial Growth: Effect of pH, Concentration, and Organic Matter.” Environ. Sci. Technol. 2009, 43, 7285–7290. 20