Repository_ApplicationSupplementForm
advertisement

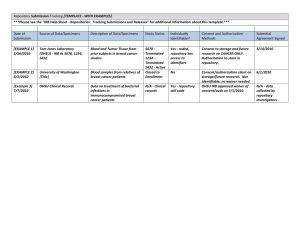
Repository Application Forms Application Supplement These additional agreements are required for all approved Repository applications for biospecimens. Agreement to Return Data Investigators agree to return data derived from received specimens back to the Repository within three years of close of funding. (This point is included in the Material Transfer Agreement that will be executed between the Repository and the Applicant’s Institution.) s I understand and agree to this policy. IRB Review and Human Subjects Training Specimens made available through the SWAN Repository were collected from individuals who provided documented informed consent that the specimens were being collected and would be made available to scientists to expand our knowledge of the public's health. Nonetheless, investigators are required to provide documentation of approval by an Institutional Review Board prior to the actual release of data or specimens. There must be evidence that the project has been submitted for review by the applicant's local IRB. A plan for notification of approval is necessary if approval does not accompany the application. Data and specimens will not be released in the absence of documented approval. Additionally, investigators and their key staff must provide written documentation that they have successfully completed training about the ethical use of materials from human subjects, the role of informed consent and the protection of confidentiality. I understand these policies, and I will provide required documentation before specimens are released to me. Genetic Counseling If you are requesting DNA samples or Genetic Data, please provide a discussion that characterizes the likelihood that the line of investigation you are proposing will lead to the need for further clinical evaluation and/or genetic counseling in affected SWAN participants. NOTE that per the Repository’s standing DNA informed consent, SWAN participants cannot be contacted. However, you will need to be prepared to provide individual results if a SWAN participant contacts us inquiring about DNA testing. Our DNA consent form contains this language regarding test results: "LEARNING ABOUT YOUR RESULTS: You can learn about results obtained from your DNA if you wish to do so. You can contact the SWAN Study, at xxx-xxx-xxxx, to see if your DNA was evaluated by scientists. If so, SWAN will provide you with an address to write to for further information. SWAN requires that the scientists using your DNA be prepared to provide information about the study they have done, what it may mean to you, and if you still wish to know the results, share your results with you (and only you), explaining how they may affect you. However, you must contact SWAN; SWAN will not contact you. This is to protect you from learning something that you do not wish to know." Consortium and Contractual Arrangements Some applications for specimens may include consortium and contractual arrangements. Documentation should be submitted which identifies and explains arrangements that are proposed between the applicant organization and the consortium organization(s). For example, two or more laboratories may embark upon a combined effort to address a question. All consortium investigators must sign a letter indicating their knowledge of the consortium and their agreement to adopt and implement the policies of the Repository with respect to the utilization of the specimens, return of the data, acknowledgement of the contribution of the Repository and the SWAN-designated co-investigator. I understand this policy, and I will provide required documentation before specimens are released to me. This policy does not apply to me. Protection of Your Proprietary Information Applicants are discouraged from submitting information considered proprietary unless it is deemed essential for proper evaluation of the application. If the application contains information which the applicant’s organization considers trade secrets or information which is confidential or privileged, identify those sections in the application that contain this information. When information in the application constitutes trade secrets or information that is commercial or financial, confidential or privileged, it is furnished in confidence with the understanding that the information shall be used or disclosed only for evaluation of the application. If specimens and data are awarded as a result of or in connection with the submission of this application, the Repository retains the right to use information to appropriately document methodology or quality assurance. Dfd Rev 09/27/13