Introduction to Ground Water
advertisement

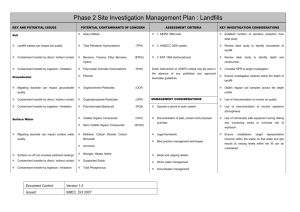
Week 1 - Introduction to Groundwater Water in three states: liquid, solid (ice), and water vapor in the air. Clouds are water droplets, condensed from vapor-saturated air. Water changes from one state to another. So water can be solid, liquid or gas. Water also moves. We call this the Water Cycle. http://ga.water.usgs.gov/edu/watercyclekoreanhi.html cycle – 주기 http://ga.water.usgs.gov/edu/watercycleprint.html circulation – move around something like a circle. Here is an animation of the Hydrologic Cycle http://techalive.mtu.edu/meec/module01/HydrologicScene.html 7 1.72, Groundwater Hydrology Lecture Packet 1 Prof. Charles Harvey http://ocw.mit.edu/NR/rdonlyres/Civil-and-Environmental-Engineering/1-72Fall2005/4F444E12-8746-459D-A6BD-1F42F7498903/0/1_72_lecture_1.pdf Here is a quiz about the Hydrologic Cycle http://techalive.mtu.edu/meec/module01/HydrologicQuiz.html Here is an animation about Evaporation. http://techalive.mtu.edu/meec/module01/Evaporation.html Here is an animation about Evapotranspiration. http://techalive.mtu.edu/meec/module01/Transpiration2.html Groundwater is an important source of freshwater. A lot of water is used for agriculture 농업 56% is used by animals and plants 20% is lost when the water is moved 24% flows back into the stream or ground A lot of water is used for making electricity. This is 87% of all industrial water use 8 Water can dissolve many things. Water consists of two atoms of hydrogen and one of oxygen, - a chemical formula of H2O. Water can dissolve at least small amounts of almost all substances that it contacts. Ground water usually has the large amounts of dissolved solids. The composition and concentration of substances dissolved in unpolluted ground water depend on the chemical composition of precipitation, on the biologic and chemical reactions on the land surface and in the soil, and on the composition of the aquifers and confining beds through which the water moves. The concentrations of substances dissolved in water are in units of weight per volume. In the International System (SI), the most commonly used units are milligrams per liter. A milligram equals 1/1,000 (0.001) of a gram, a liter equals 1/1,000 of a cubic meter, 1 mg/L equals 1 gram m -3. and so The concentration of most substances dissolved in water is small. The weight per weight unit commonly used is parts per million (ppm). 9 Natural inorganic constituents commonly dissolved in water that are most likely to affect use of the water. Substance Major natural sources Effect on water use Important concentration (mg/L3) Bicarbonate (HCO3) and carbonate (CO3) The solution of carbonate rocks by water containing carbon dioxide. 150-200 Calcium (Ca) and magnesium (Mg) Soils and rocks containing limestone (CaCO3), dolomite (CaMgCO3), and gypsum (CaSO4). Small amounts from igneous and metamorphic rocks In inland areas, from seawater trapped in sediments at time of deposition. In coastal areas, from seawater in contact with freshwater in productive aquifers. Both sedimentary and igneous rocks. Not widespread Control the ability of water to neutralize strong acids Bicarbonates of calcium and magnesium decompose in steam boilers and water heaters to form scale and release corrosive carbon dioxide gas. With calcium and magnesium, can cause carbonate hardness Main cause of hardness and of boiler scale and deposits in hot-water heaters. in large amounts, increases corrosiveness of water and, in combination with sodium, gives water a salty taste 250 In low concentrations, reduces tooth decay, at higher concentrations, causes color of teeth. 07-1.22 Chloride (Cl) Fluoride (F) 25-50 Iron (Fe) and manganese (Mn) Iron present in most soils and rocks; manganese less widely distributed. Stain laundry and are bad in food processing, dyeing, bleaching, brewing, and other industrial processes Fe>03, Mn>0.05 Sodium (Na) Same as for chloride. See chloride. In large concentrations, may affect persons with heart disease, and other medical problems. Sodium may damage irrigated crops 69 (irrigation) 20-170 (health) Sulfate(SO4) Gypsum, pyrite (FeS), and other rocks containing sulfur (S) compounds Low concentrations give water a bitter taste and higher concentrations have a laxative effect With calcium, forms a hard calcium carbonate scale in steam boilers 300-400 (taste), 600-1,000 (laxative) 10 Pollution of Groundwater Pollution of ground water is any loss of quality of the water because of what people do. Most pollution of ground water comes from: the disposal of wastes o on the land surface, o in shallow holes including septic tanks, or o through deep wells and mines; the use of fertilizers and other agricultural chemicals; leaks in sewers, storage tanks, and pipelines; and animal feedlots. The size of any pollution problem depends on: the size of the area affected and the amount of the pollutant involved, the solubility, toxicity, and density of the pollutant, the mineral composition and hydraulic characteristics of the soils and rocks through which the pollutant moves, and the effect or potential effect on ground-water use. Affected areas range in size from: point sources, such as septic tanks, to large urban areas having leaky sewer systems and numerous municipal and industrial waste-disposal sites. 11 Nearly all substances are soluble to some extent in water. Many chemical wastes are highly toxic even in very small concentrations. Contamination Basics: Concentration We need to know both the type and amount of contaminant in the groundwater system. We can measure amounts of contaminants in ppm and ppb, which are parts per million and parts per billion. ppm is the amount of contaminant units in 1,000,000 (1 million) units of groundwater. ppb is the amount of contaminant units in 1,000,000,000 (1 billion) units of groundwater. pph (parts per hundred), is the amount of contaminant units in 100 units of groundwater. We call it percent, %. (1pph = 1/100 =1%) This animation gives an idea of the different concentrations. http://techalive.mtu.edu/meec/module04/Concentration.html Which is a larger concentration, 1 ppm or 1 ppb? Effects of Contamination Why do we care if there was only 1ppm (1/1,000,000) of a particular contaminant? It seems like too small of a number to matter. With groundwater contaminants, both chemical and biological, a very small concentration of contaminant could still lead to serious health effects. Different kinds of contaminants have different concentration limits at which they start to effect us or the environment. In dealing with groundwater contamination problems, it is necessary to find out both the specific type of contaminant and the concentration of the contaminant. In this animation vary the concentration of different contaminants in the groundwater. Check out Mr. Blue's reactions to the contaminants. Mr. Blue reacts in the same way a healthy person would to these chemicals. A person who is not as healthy, such as an older person or a small child, might 12 suffer the effects even more. http://techalive.mtu.edu/meec/module04/ContaminationEffects.html Which chemical needs to exist in the least concentration in the groundwater in order to avoid hurting someone? Density The density of a liquid substance - that is, the weight per unit volume of the substance relative to that of water - affects its underground movement. Densities range from: o petroleum products that are less dense than water o other substances that are denser than water. Substances less dense than water float at the top of the saturated zone; o if, like petroleum, they are immiscible, they will spread in all directions as a thin layer. Substances denser than water move downward through the saturated zone to the first extensive confining bed. Contaminant Movement Contaminants can enter the groundwater by seeping in from the surface or by flowing in from another part of the aquifer. Once in the aquifer they move with the groundwater flow. As contamination moves it disperses. This means that the concentration decreases as it moves farther away from the source of the pollution. For that reason there are different concentrations of contaminants at different points in the aquifer. The minerals and physical characteristics of soils and rocks through which pollutants move may affect the pollutants in several ways. If a pollutant enters the ground at a "point," it will be dispersed longitudinally and laterally in granular materials so that its concentration will be reduced in the direction of movement. The visual representation of these different concentrations is called a contamination plume. How the plume looks depends on the type of contamination source, the specific contaminant(s), where the aquifers are located, and different soils in that area. These animations show the two main categories of plumes based upon their source. Here are two animations of the Dispersal of a Pollutant from a Point source http://techalive.mtu.edu/meec/module04/OneTimePlume.html (Click Play) http://techalive.mtu.edu/meec/module04/ContinuousPlume.html (Click Play) Organic Substances Organic substances and other biodegradable materials can be broken down both by oxidation and by bacterial action in the unsaturated zone. Soil, especially clays and organic matter, may absorb trace metals and some complex organic 13 pollutants and reduce their concentration as they move through the underground environment. The hydraulic characteristics of the soils and rocks determine the path taken by and the rate of movement of pollutants. Substances dissolved in water move with the water except when they are tied up or delayed by adsorption. The movement of pollutants is most through the most permeable zones; the farther their point of origin from a ground-water discharge area, the deeper they move into the ground-water system and the larger the area affected. The factors related to the movement of pollutants must be carefully considered in the selection of waste-disposal sites, animal feedlots, and sites for other activities that may cause ground-water pollution. Here is an animation to allow you to Pick the Best Site for a New Well http://techalive.mtu.edu/meec/module06/WellSiting.htm 14 15 Review Match the letters with the name. 16