part a: basic manipulations with percents - Bio-Link
advertisement

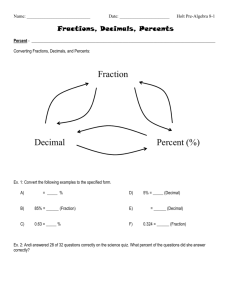
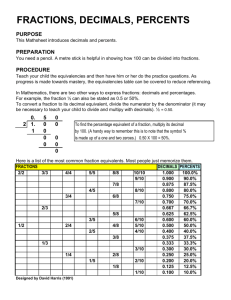
1 Chapters 10 and 11 Problem Set Key: Percents and Introduction to Concentration 1. 2. 3. 4. 5. PART A: BASIC MANIPULATIONS WITH PERCENTS Express the following percents as fractions AND as decimals: 5% fraction = 5/100 decimal = 0.05 0.5% fraction = 0.5/100 decimal = 0.005 0.05% fraction = 0.05/100 decimal = 0.0005 0.341% fraction = 0.341/100 decimal = 0.00341 2.433% fraction = 2.433/100 decimal = 0.02433 Express the following fractions or decimals as percents: 6. 20/50 percent = 40% 7. 4/3 percent = 133.33% 8. 2.0/100 percent = 2% 9. 0.225 percent = 22.5% State the following as percents: 10. 1 part in 100 total parts percent = 1% 11. 2 parts in 25 total parts percent = 8% 12. 18 parts in (18 parts + 42 parts) percent = 30% 13. 0.05 parts in 1 part total percent = 5% 2 PART B: PERCENT ERROR 1. You are doing a new laboratory test for the first time and are trying to determine your accuracy. The test examines the level of calcium in a sample. You receive a sample that has been provided as a reference, and is known to be at a concentration of 23.0 mg calcium/L. You test this reference sample 10 times and get the following calcium concentrations: 23.0 mg/L 23.3 mg/L 23.0 mg/L 23.1 mg/L 22.8 mg/L 23.2 mg/L 23.1 mg/L 23.0 mg/L 22.9 mg/L 23.1 mg/L Determine the percent error of your results, given the formula below: % error = experimental value - true value X 100% true value average = 23.1 mg/L (not rounded = 23.05) % error = 23.1 mg/L – 23.0 mg/L X 100% = 0.43% 23.0 mg/L 3 PART C: Introduction to Concentration 1. Describe how to make 500 mL of 250 g/L NaCl. 0.5 L(250 g/L) = 125 g Dissolve 125 g NaCl in enough water to make 500 mL total solution 2. Describe how to make 100 mL of a 100 g/mL glucose solution. 100 mL (100 g/mL) = 10,000 g Dissolve 10,000 g (10 mg) glucose in enough water to make 100 mL total solution 3. How much KOH is present in 5 L of a 0.1 g/mL solution? 5000 mL (0.1 g/mL) = 500 g KOH 4. How much enzyme is present in 150 L of a 250 g/mL enzyme solution? 150 L ( 250 g/mL)(1 mL/1000 L) = 37.5 g enzyme 5. Which of the following concentrations is equal to 500 g/mL MgCl2? a. 500 mg/L(1L/1000mL)(1000 ug/mg) = 500 ug/mL b. 0.5 mg/mL (1000 ug/mg) = 500 ug/mL c. 50 g/L (1 L/1000 mL) = 0.05 ug/mL d. 5000 ng/mL (1 ug/1000 ng) = 5 ug/mL e. 0.05 g/mL (106 ug/g) = 50,000 ug/mL