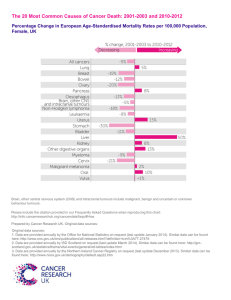
Renal & ureteric neoplasms
advertisement

Renal & ureteric neoplasms Epidemiology of renal neoplasms A classification of kidney tumours is quite a difficult task. A simply classification has been done by Barbaric (1994). This classification was based on pathology (malignant, benign, or inflammatory tumours). Benign renal tumours Benign renal tumours arise from any of cell types in the kidney. The most common benign lesions are renal cysts. 70% of asymptomatic renal masses are simple cysts. They may be single or multiple and unilateral or bilateral. Benign tumours include: adenoma, fibroma, renal oncocytoma, renal angiomyolipoma, lipoma, myoma, lymphangioma, hemangioma. Malignant renal tumours Renal tumours account for around 3% of all neoplasias. Malignant tumours of renal parenchyma are relatively uncommon but remain a problem in urological oncology related to early diagnosis, innaccuracy of pre-operative staging and toxicity of the treatment for metastatic disease. Nephrocarcinoma is the generic category that includes adult parenchymatous malignancies, primarily the classic hypernephroma. Relatively rare are mesenchymal malignancies such as sarcomas, heman-giopericytomas, infiltrative malignancies such as myeloma, and secondary or metastatic malignancies. Renal cell carcinoma also defined as hypernephroma or Grawitz's tumour originates from the proximal convoluted tubule and has an incidence of around 3,6 - 5,6 per 100.000 males and 1,7 to 3,4 per 100.000 females. The tumour mainly affects adults aged 50 - 70 years with the male: female ratio of 3:1. Renal cell carcinoma (RCC) is the most common malignant lesion of the kidney with the percentage of around 90%, followed by Wilms' tumour, which is the most common in childhood but is very rare in adults. There are marked international variations in incidence of RCC, being high in Northern Europe and North America, and low in Africa, Asia and South America. There is some evidence of increasing incidence in males in the last years. The disease can occur at any age. Despite the fact that RCC is the disease of the adults, the tumour may well occur in children as several reports have shown. Renal tumours have been a major subject of cytogenetic and molecular genetic studies, which suggest that these findings may be useful clinically in diagnosing renal tumours and metastases and indicating poor prognostic genetic markers. RCC like other cancers occurs in inherited and sporadic, non-inherited forms. The hallmarks of familial renal cancer are that they occur at an early age, and usually are bilateral and multifocal. Tumours of the renal pelvis Epithelial tumours of the renal pelvis have the same histologic morphology as the ureter and bladder tumours, because they arise from urothelial cells. Transitional and squamosus cell carcinomas of the renal pelvis can be induced by the action of such substances as dibenzanthracen, benzpyrene, methylcholantren, aflatoxin B, metabolites of certain chemical agents such as benzidine. In carcinogenesis RNA viruses, inflammation, stone disease and metaplasia also play a causative role. Tumours of the renal pelvis are about 40 times more common in patients who take large amounts of phenacetin. An association between ta-bacco use and development of urothelial tumours of the renal pelvis is also shown. The renal pelvis tumours are reltively rare. 7-8% of all renal tumours arise in kidney pelvis and 6-12% of all malignant kidney tumours are transitional cell carcinomas. The majority of these tumours develop in the elderly, namely in the 6 th to 8th decade and they mainly affect males with the male to female ratio 2-4: 1. Pathology of renal tumours Renal cell carcinoma (WHO) Renal cell adenocarcinoma, hypernephroma, metanephroma, Grawitz tumour, alveolar carcinoma, adenocarcinoma of kidney, adenocarcinoma renis, and clear cell, dark cell, and granular cell carcinoma). This cancer comprises up to 80% of renal cancer and is infrequent before the fifth decade of life, with the pick incidence in the sixth decade of life. Renal cell carcinoma (RCC) is rare in children. RCC occurs more frequently in men than in women, and is rarely familiar. The strong association is seen between RCC and syndrome of von Hippel and Landau. The etiology in humans is largely unknown but RCC occurs more frequently in cigarette smokers. Ultrastructural and immunologic studies clearly demonstrated that RCC arises from the cells of proximal convoluted tubule. The clear tumour cells contain abundant glycogen and lipid in their cytoplasm. RCC arises usually in the upper pole of the kidney. In about 5% of all cases RCC are multifocal and in 1% are bilateral. It is not uncommon that RCC is presented initially with metastatic disease. Gross examination shows ovoid soft mass. The cut surface may be grey, orange or golden yellow with foci of hemorrhage, necrosis, cystic degeneration and sometimes calcifications. The small RCC tumours are surrounded by pseudocapsule of fibrous tissue and atrophic renal parenchyma. In late stage RCC penetrates its own pseudocapsule, renal fibrous capsule and penetrates adjacent adipose tissue. The infiltrate of kidney pelvis and hi-lar structures in cases presented with hematuria is seen; the infiltrate of renal vein in cases presented with metastatic disease is common. Sometimes large RCC goes without any metastases and small RCC tumours spread to lungs and then to brain, liver and others organs. Nephroblastoma (Wilms' tumour) Nephroblastoma is the most common of the malignant abdominal tumours of young children but accounts for only 6% of renal cancers in all ages. It may be present at birth, 28% occur in the first year and over 96% occur before 6 years of age. Rarely nephroblastoma arises in sites other than kidney such as inguinal canal or within teratoma. On gross examination the tumour is usually large (clinically it appears most commonly as palpable abdominal mass), rounded, and dwarfing but not invading the kidney until late. The cut surface revealed a soft, grey-white to cream coloured tumour tissue foci of hemorrhage and necrosis. Nephroblastoma is the malignant counterpart of embryonic rather than mature tissue. Microscopically the typical nephroblastoma is composed of variable amounts of metanephric blastema and its epithelial and stro-mal derivatives. The blastema cells resemble: normal metanephros with small ovoid cells with scanty cytoplasm and is arranged in nests or trabeculae. The epithelial element is usually trabecular; sometimes the epithelium forms papillary structures or even glomerulilike structures. The stroma is heterogeneous and most often consists of spindle cells of undetermined type, fibroblasts or smooth muscles. Sometimes foci of cartilage, adipose tissue and neural elements are seen. While most of nephro-blastomas show the mixed pattern, the tumour may consist predominantly or entirely of blastemal, epithelial or mesenchymal elements. Areas of leiomyosarcoma or rhabdomyosarcoma and even neuroblastoma (with rosettes) or ganglion cells may be present in nephroblastoma. Malignant renal tumours Renal cell carcinoma (RCC), clear cell carcinoma, Grawitz tumour Renal cell carcinoma is a relatively rare tumour, accounting for approximately 3% of adult malignancies. RCC is more common among urban dwellers and is more cmmon in males, with a male-to-female ratio of approximately 2:1. Familial renal carcinoma has been reported. Patients with von Hippel-Lindau disease have a higher incidence of this cancer polycystic kidney disease appear to be a predisposition for development of the tumour, although this has not been firmly established. Renal carcinoma is a tumour of adults, occuring primarily in those in their forties to sixties, but it may occasionally occur in younger age groups. RCC is typically unilateral, but bilaterality, either synchronous or asynchronous, occurs in approximately 2% of cases. Von Hippel-Lindau disease is characteristically associated with the presence of multiple and bilateral renal carcinomas. The lump frequently extends into the renal vein as a thrombus, which may be propagated for varying, distances into the inferior vena cava, even up to the right atrium of the heart. The more malignant and larger tumours can invade locally, with extension into the surrounding muscles and direct invasion into adjacent organs. Because of the long natural history of renal cell carcinoma and high probability of late relapses or metastases, 10 to 15-year observation time has become widely accepted to judge the effectiveness of therapy. As far as symptoms is concerned, the classic triad of pain, hematuria and flank mass is found in few patients and generally indicates advanced disease. RCC arises from the proximal convoluted tubule and grows rather slowly; therefore it can often achieve an enormous size without causing any symptoms. The most frequent findings are pain (tumour invades surrounding areas or obstructs the outflow of urine owing to hemorrhage and subsequent formation of blood clots) or hematuria (as a result of invasion of the collecting system) secondary to the primary tumour, but symptoms owing to metastatic disease probably occurs more frequently. Raised erythrocyte sedimentation rate, weight loss, cachexia, fever, night sweats, and the sudden development of a varicocele Varicocele in the male patient are not uncommon findings. Hypertension is due to segmental artery occlusion or to elaboration of renin or renin-like substances. The most dramatic syndrome is associated with nonmetastatic hepatic dysfunction and is referred to as hepato-renal syndrome [Stauffer syndrome] (abnormal liver function tests, white blood cell loss, fever, and areas of hepatic necrosis without hepatic metastasis). Hepatic functions return to normal by majority of patients after nephrectomy. Hypercalcemia of rather obscure nature (skeletal metastases? production of specific polypeptides?) and increased levels of erythropoietin were also noted by some patients with RCC. Sarcomas of the kidney constitute only about 1 to 3% of malignant tumours of the kidney but increase in incidence with advancing age. The most common symptoms are essentially those of a large renal carcinoma. Leiomyosarcomas, are the most common variety. These tumours tend to compress and displace the kidney rather than invade it. The survival in such lanes is extremely poor. Lymphomas generally occur in the kidney only as a manifestation of the systemic disease, often bilaterally. It can be silent or can produce he-maturia, enlarged kidneys, and progressive renal failure. Metastatic tumours from a variety of solid neoplasms are often localised in the kidneys, because high blood flow and profuse vascularity of renal parenchyma provide ahospitable environment for deposition and growth of malignant cells. Metastases to the kidneys, because of dominating symptoms of basal disease, are identified mostly at autopsy, and, in that sense, are of minimal clinical value. Radiologic methods of study in renal tumours 1. Ultrasonography This procedure should be initial in diagnosis of the urinary tract. This examination is safe for the patient, noninvasive, repeatable and very efficient. Ultrasonography allows to distinguish solid mass from the cyst. Probability of detection of 3 cm tumour is 95%. The diagnostic capabilities of sonographic Doppler analysis of blood flow allow noninvasive recognition of the vascular pathology. The multiplanar capability of US demonstrates the extent of the tumour. Although US is extensively used in the detection of renal neoplasm, it is less accurate than CT or MRI in the staging of tumours. In 50% of patients the retroperitoneal region may not be adequately visualised. US is also unable to detect muscle inavasion. 2. Plain film Plain film Antero-posterio film must precede any contrast study of the urinary tract. The film should include the entire area from the diaphragm to the pubic symphisis. Plain film allows in many cases to suspect the presence of the tumour by: • enlargement of whole or part of the kidney. • changing of the shape of kidney, abnormal position of the organ • presence of calcifications 3. Excretory Urography – IVP Excretory Urography This examination is sometimes required by clinicians to complete the diagnosis of renal neoplasm. IVP is the type of dynamic examination which allows to evaluate excretory functions of kidneys. In this examination we can observe all kinds of must be done during so-called nephro-graphic phase. This is the period when opacification of the renal parenchyma is at its best. Simple cysts or tumours may be diagnosed by this method with 90% accuracy (3). Polar enlargement of a kidney is one of the most frequent pyelographic changes. The renal outline may be altered and markedly irregular. Calcification of renal carcinoma is not common but may occur and may simulate renal lithiasis or calcifaction of a renal cyst. The architecture of the collecting system varies from simple deformity of a calyx to outright destruction of one or more calyces with their amputation. 4. Computed tomography CT (Tumor of the kidney) Over the past decade, CT has become the most widely used technique for diagnosis and staging renal tumours, partially due to the very high overall accuracy up to 90%, that has been achieved. Indeterminated masses in US should be further evaluated by CT. A large variability in the range of attenuation values exists because of necrosis, hemorrhage, calcification, variable protein content and vascularity. Typically renal tumours are solid in density, that is greater than a typical cyst. The margins of the mass are usually irregular. The density of the mass may be homogeneous or non homogeneous. The tumour can demonstrate local invasion into the fat within Gerota's fascia and to the adjacent organs. The reported accuracy rate for CT in staging renal tumours ranges between 72% and 98%. Stage I tumours are defined on CT lying entirely within the kidney. Stage II Tumour spread into the perinephric space. Renal capsular invasion is difficult to diagnose. Recognised signs on CT include extending tumours to the perinephric space, thickening of the perirenal fascia, soft tissue spreading into the perirenal fat. Stage III tumour spread to the venous system and lymph node involvement. Detection of lymph node involvement on CT relies on detecting an increase in the size of infiltrated node. The most important sign of venous tumour infiltration is a persistent filling defect following intravenous contrast administration within the renal vein or IVC. Stage IV Tumour spread to adjacent organs and distant metastasis are well demonstrated in CT and the specificity is almost 98%. 5. Magnetic Resonance Imaging Due to its high tissue contrast and multiplanar imaging capabilities, MRI provides a detailed display of renal anatomy. Recent technical developments overcoming the problem of respiration motion artefacts and the use of paramagnetic contrast agents have further improved the performance of MRI which is now an alternative or complementary imaging modality to US, excretory urography and CT. On plain MR images solid as well as cystic renal and pararenal masses often present the same signal intensity as the adjacent renal tissue. Therefore detection of such tumours is based only on the visible displacement or alteration of organ contours. Delineation from the surrounding renal parenchyma is successful with the aid of contrast enhanced examination. Additional techniques such as fat suppression, provide further improvement of lesion detection, allowing demonstration of small cyst or solid masses with diameters less than I cm. MRI is highly sensitive in demonstrating hemorrhage, hemorrhagic cyst. Among solid tumours of the kidney, MRI allows detection of only fat-containing angiomyolipoma accurately, other solid lesions show very similar SI patterns. Advantages of MRI in staging of the tumour include direct imaging in sagittal and coronal planes. MRI is faced with the same limitations as CT in identifying early extension into the perinephric space. MRI is the best noninvasive method for the assessment of tumour thrombi in the renal veins or in the inferior vena cava. At present MRI and spiral contrast-enhanced CT have similar accuracy in detecting lymph node and venous invasion. The choice between these techniques will depend on several factors apart from accuracy, such as availability and cost. Currently CT is the most widely used technique, with MRI kept in reserve to solve particular problems. 6. Renal arteriography; aortography; phlebography an inferior vena cavography Visualisation of the renal arteries and renal veins may be accomplished by: • translumbar aortography • percutaneous aortography • selective renal arteriography • percutaneous phlebography It is dynamic study of arterial blood supply of the kidney and the tumour and allows to show the pathology as well as anatomy. Visualisation of the renal veins has become very important in the diagnosis of invasive malignancies of the kidneys and their prognostic evaluation. During these examinations we can provide embolisation of the tumour in nonoperative tumours or before the surgery. Classification and principles of treatment of renal tumours Clinical staging of renal tumours is very important in order to treat them correctly. The objectives of the clinical staging ought to be the following: 1. evaluation of the local extension of the tumour 2. determination of the presence of distant metastases 3. determination of the presence of venous tumour thrombus Clinical staging is important in order to decide on a therapeutical approach but surgical and anatomopathological staging is equally important for our knowledge about prognosis. RCC The UICC present TNM classification of RCC is following: Tl - tumour < 7 cm. in greatest dimension limited to the kidney T2 - tumour > 7 cm. in greatest dimension limited to the kidney T3 - tumour extends into major veins or involve adrenal or perinephric tissues but not beyond Gerota's fascia T3a - tumour invades adrenal gland or perinephric tissues but not beyond Gerota's fascia T3b - tumour grossly extends into renal vein or vena cava T3c - tumour grossly extends into vena cava above the diaphragm T4 tumour invades beyond Gerota's fascia NO - no regional lymph node metastasis Nl - metastasis to a single regional lymph node N2 - metastasis in more than one regional lymph node MO - no distant metastases Ml - distant metastasis During the last decade it has been observed that patients with Tl and T2 tumours have similar survival and that 2,5 cm. cutoff does not adequatly differentiate between stage Tl NO Mo and T2 NO MO. In 1997 TNM classification of renal cell carcinoma the cutoff between Tl and T2 tumours was increased to 7 cm.. The 1997 TNM classification resulted in a redistribution many cases of RCC from stage T2 to stage Tl. This group represents 44% of all cases and 76% of stage T2 cases according to 1987 TNM classification. Many findings suggest that increasing the cutoff from 2,5 cm. to 7 cm. alone does not greatly affect outcome. The benefit of the 1997 TNM staging classification of RCC is a potential cost savings. Some investigators postulate elimination of postoperative abdominal CT for Tl NO MO patients and reduction in followup tests and visits. The impact of these savings needs to be evaluated in prospective studies. After all diagnostic steps, staging of the tumour according to TNM classification may be done and then decision about form of the therapy may be made, too. Surgical therapy is the most effective management form of RCC. In the case of local extension of the tumour at the time of diagnosis succesful therapy in RCC is radical nephrectomy - removal of the entire tumourous kidney including fatty capsule and adrenal gland within Gerota's fascia followed by lymph node dissection. After radical nephrectomy the 5 - year survival rate increased to 52 - 60% compared with 33% for simple nephrectomy and 10 - year survival rate from 7,1% to 49%. The operative mortality is 1 - 5%. Distant metastases, at the time of diagnosis, are found in 30% of patients. The indication for surgery in such cases is judged differently by various authors. Popular is the opinion that removal of all neoplastic tissue is mandatory. sue is mandatory. The hope for spontaneus regression of distant metastases after nephrectomy should not influence the indication for surgery. In j these cases nephrectomy is indicated if surgical removal of the metasta-ses is also planned. In patients with more locally advanced disease complete excision of the tumour by means of an extensive operation including if necessary liver lobectomy and excision of the involved bowel, spleen or other organs will be performed. It is very difficult to attain true radicality and surgery often is a compromise between oncological radicality and debulking. Even in the presence of distant metastases some of urologists proposed aggresive therapy including tumour nephrectomy. In these cases a prolonged life expectancy cannot be achieved. As a palliative procedure in complications due to the tumour (hemorrhage, pain, policythemia, hypercalcaemia) nephrectomy may be indicated. Alternative procedure to palliative nephrectomy is embolisation of the tumour. There are also immunological considerations that a positive effect on the cellular immunity following tumour nephrectomy has been observed. In the case of bilateral renal tumours or tumour in a solitary kidney, the decision may be very difficult. In the case of bilateral tumours, the one side is removed radically but the other side undegoes an organsaving operation. It should be done also on the solitary kidney, if j anatomically possible. Extracorporeal bench surgery and autotransplantation is another possibility. Bilateral nephrectomy is indicated rarely because of the side effects and toxicity of immunosupressive therapy or hemodialysis. In the cases of more advanced metastatic disease other than surgical forms of treatment such as radiotherapy, chemotherapy and immunother-apy may be used. Surgical treatment of renal tumours The basic form of the renal tumour treatment is surgery. In the present time various modalities of surgical treatment are used: radical surgery -radical nephrectomy, nephron - sparing surgery and palliative surgery. Incision The fundamental principles of radical surgery are following: - choice of the best incision - preliminary ligation of the renal artery - lymphadenectomy - complete excision of the tumour - control and removal the tumour extending into the inferior vena cava - excision of resectable distant metastases The most popular parietal incision for radical nephrectomy are: xiphoumbilicus epigastric subcostal incision, transverse abdominal incision and thoracophreno-laparotomic incision. Two last incisions give optimal exposure of inferior vena cava and are the best approaches for right renal tumours (RCC) extending into the inferior vena cava. Optimal parietal incisions allow to expose the main vessels: the vena cava inferior, the abdominal aorta and allow to reach the renal artery at its origin. The most important step of the radical nephrectomy is initial ligation of the renal artery and then ligation of the renal vein. This procedure prevents the spread of the tumour cells. The next steps are: the mobilization of the kidney, the division of the ureter and exenteration of the renal lodge by,,en bloc" removal of the kidney, adrenal and perirenal fat with Gerota's fascia. During radical nephrectomy hi-lar, pericaval or periaortal lymphadenectomy is also performed. The role of lymphadenectomy in renal cell carcinoma is controversial in the context of oncological urology. The reported incidence of retroperitoneal lymph node metastases ranges from 6% to 43,5%. The differences in the frequency of nodal involvement may be explained by the extent of lymphadenectomy. Recently the incidence of nodal metastases is minor because the diagnosis of RCC often occurs in the early stage. However in 7% of patients with tumour confined to the kidney (Tl, T2, M0) nodal metastases were seen. Cumulative overall survival after radical nephrectomy and extended lymphadenectomy was appreciated on 46% and 25% at 5 and 10 years respectively. In patients with tumour stage pTl NO M0 survival rates at 5 and 10 years of 80% and 50% respectively were noted, while in patients with tumour stage pT2 N 0 M0 survival rates were 68% and 59% at 5 and 10 years respectively. Patients with tumour extending to perirenal fat (T3) have shown a 70% survival rate at 5 years after radical nephrectomy. It is a significant improvement compared to the 14% survival rate at 5 years after simple nephrectomy. In patients with lymph node metastases survival rates of 52% and 26% at 5 and 10 years respectively were observed. Lymphadenectomy, an integrating part of radical nephrectomy does not increase operative mortality and morbidity, allows for improved surgical staging of the tumour and lower frequency of local recurrences. In the last years there have been some reports, which indicate that almost all patients with lymphatic spread of RCC have additional distant metastases. Therefore, the therpeutic effect of extensive retroperitoneal lymphadenectomy in association with radical nephrectomy seems to be low. However, limited lymph node dissection may be useful as a staging procedure. Thus it seems rational that radical nephrectomy with lymphadenectomy (extended or limited) is indicated in T2, T3 and in some T4 cases of RCC tumours. Conservative surgery of renal tumours. Renal cell carcinoma may develop synchronously in both kidneys in about 1,8 to 3% of cases. Frequency of renal cancer in solitary kidney is not well known.The classic indications for nephron sparing surgery are: bilateral renal tumours, tumour in solitary kidney and in the case when radical nephrectomy would result in renal insufficiency necessitating dialysis or transplantation. Conservative surgery for RCC may be identified with two types of procedures: enucleation of the tumours (tumourectomy) and partial nephrectomy. Both these techniques may be performed in situ or ex situ. Usually, clamping of the renal artery is unnecessary but if it was indicated regional hypothermia should be considered. Partial nephrectomy The classic indications for conservative surgery of RCC are justifiable. In the last years there has been a tendency toward wider application of kidney sparing surgery. Elective kidney sparing surgery means that this form of conservative treatment is used when the contralateral kidney is normal. The arguments against elective nephron sparing surgery are following: rradical nephrectomy for Tl and T2 RCC will probably be curative in 100%, the chance of developing contralateral tumour is small, higher complications rate for conservative surgery, the possibility of tumour recurrence, the possibility of multifocal RCC in the kidney. An argument favoring nephron sparing surgery for RCC is its efficacy in providing for cure of cancer in series of electively treated patients. The criteria for the indications of elective nephron sparing surgery are following: the tumour should be solitary, well demarcated on CT, the tumour should be; well localized and easily resectable, the tumour size should not exceed 4,3 cm. in diameter (3-5). Elective kidney sparing surgery in the case of small, low grade, well localized RCC may be usually curative. Approximately one third of patients with RCC have distant metastases at diagnosis. In patients with multiple metastases survival is not positively affected by their complete excision. In these cases surgical treatment of metastases has only a pallitive role. In the case of solitary synchronous or metachronous metastasis the complete removal of metastatic foci would significantly improve the probability of longer survival. The patient with synchronous metastasis has worse prognosis if compared to this with metachronous disease. The beneficial effects of excision of metastatic renal cell carcinoma are limited and the cure of patients with me- j tastatic disease is relatively uncommon. Non-surgical treatment of renal tumours Non-surgical treatment of renal cell carcinoma comprises irradiation, transfemoral catheter embolisation of inoperable and symtomatic tumours, cytostatic treatment of metastatic disease, hormonotherapy and immunotherapy. Embolisation of the kidney Transfemoral catheter embolisation of the kidney has the following applications: 1. curative superselective erribolization in non malignant renal diseases 2. radiologic nephrectomy as the form of palliative embolisation of renal tumours (bleeding, pain, paraneoplastic syndromes) 3. preoperative embolisation of renal tumours Embolisation of the kidney is in the majority of cases only palliative procedure and very seldom gives a curative effect. Radiotherapy In 1923 Waters observed that radiation could reduce the tumour mass of renal cell carcinoma. For a long time it was thought that RCC is radio-resistent. The value of pre- or postoperative radiotherapy for the prognosis of the patients with RCC is still discussed. Postoperative radiotherapyn locally advanced renal tumours is used in some oncological centers. More often is indicated palliative radiotherapy of metastases. Bone pain from metastases is well palliated in this way. Hormonal therapy Many reports from the 70's have indicated good response in patients with RCC receiving progesterone or androgens. The gestagens: medroxypro-gesterone and hydroxyprogesterone as well as testosterone were applied. The first optimistic results in prospective studies have not been confirmed. On the other hand high dose of progesterone treatment is nontoxic anabolic therapy which offers the improvement of patient's general condition. Chemotherapy Many cytostatic drugs were used in the treatment of metastatic RCC. Positive results of such treatment were observed in 10% patients. In many studies vinblastine (VB) as a single drug or in combination with lomustine (LM, CCNU) was the most effective with response rate from 15-30%. The continous infusional chemotherapy with 5- fluoro-2-deoxyuridine (FUOR) gives a response rate of 2033% in patients with metastatic RCC. In general results of combined chemotherapy are not superior to those obtained by single drug treatment. Immunotherapy The biology of renal cell carcinoma is considered to be influenced by immune system of the tumour host. Occasional spontaneous regressions of metastases are arguments for this assumption. Multiple modalities for active or passive immunotherapy were described. In conventional immunotherapy of RCC were used following agents: BCG, transfer factor (TF), polymerised tumour cells, tumour cells + Corynebacterium parvum, immune RNA (I- mRNA), tumour necrosis factor (TNF), thymosine factor 5. In the current opinion use these agents was largely unsuccessful. The forms of adoptive immunotherapy are LAK and TEL treatment in solid renal tumours. Lymphokine activated killer cells (LAK) treatment gives a 31% response rate (CR + PR) in RCC. An alternative to the use of LAK cells generated from peripheral blood lymphocytes is using the lymphocytes which infiltrate the tumour (TIL). LAK and TIL adoptive immunotherapy has been criticised mainly on account of the toxicity and expence. In the present time the most often form of immunotherapy is the use of biological response modifiers such as interferons and interleukins. A large number of patients have been treated with interferons for advanced RCC (interferon alfa, interferon beta and interferon gamma). Doses of interferons ranged from 1 MIU dalily to 16 MIU daily although the majority of patients were treated with 3 MIU daily or the same dose 3 times each week. Observed responses rates ranged from 5 to 29%. There is no evidence that larger doses than 5-10 MIU daily produce higher response rates. The toxicity of IFN with increasing doses and prolonged treatment is higher. Interleukin-2 (11-2) is produced by activated T cells. Some authors reported the clinical result with 11-2 in RCC. It was used in intravenous infusions but toxicity of such treatment was high. Recently systemic immunotherapy with interleukin 2 (11-2), interferon alfa (INF alfa) and 5-fluorouracyl gives promising results with response rate about 4048%. However, systemic immunotherapy is limited by severe side effects. Longlasting response is rare but only systemic com-i bined immunotherapy is the most efficient in the treatment of metastaticj RCC. Neoplasms of the ureter -epidemiology and pathology Ureteric tumours are relatively uncommon and account for about 1% of all urothelial tumours. Males are about 3 times more likely to suffer from ureteric tumours than females. The peak incidence in men occurs in the 75 to 79-year-old age range. According to Annual Cancer Statistic Review, the disease affects 10 white men per 100000 per year. In families afflicted with Balkan endemic nephropathy, a much higher incidence of upper urinary tract transitional cell carcinoma is seen. Petkovic (1975) reported that the incidence rates for some villages affected by Balkan endemic nephropathy were 100 to 200 times greater than for similar towns nearby. The pathological analysis indicates that more than 90% of ureteric tumours are transitional cell carcinomas. According to the current classification, three grades of malignancy are distinguished. Grade I is characterised by the increased number of epithelium layers but the separate cells are not much different from the cells of normal urothelium. In tumours identified as grade II the urothelium is thickened and the cell, which reveal various amounts of cytoplasm and diverse sizes of nuclei, are patterned irregularly. Random arrangements of the cells, whose nuclei differ considerably from one another in size and shape, are typical of grade III tumours. Nevertheless they still can be recognised as transitional cell carcinomas. The term anaplastic carcinomas refers to tumours that cannot be recognised as urethelial as the pathological view is blurred. Papilloma is a benign urothelial tumour. The tumour consists of fibrovascular stalks lined by normal urothelium. Such rumours are never anaplastic. They are not invasive. Nor do they metastasise. But they are very uncommon. The incidence of papilloma in the ureter is not more than 3%. Inverted papillomas occur in the ureter as well as in the bladder. They are usually benign but they may occur in association with other urothelial carcinomas. Grainger et al. (1990) reported an 18% incidence of malignancy within inverted papillomas of the ureter. Thus patients with inverted papillomas of the ureter should be followed closely for upper urinary tract and bladder transitional cell carcinomas. In 1952 Melicow identified carinoma in situ. According to UICC, the name is used to denote anaplasia of the superficial layers of urothelium without exophytic growth or invasion. Carcinoma in situ is often associated with low-differentiated tumours or it may precede their development. Thus it requires early and aggressive treatment. Squamous cell carcinomas are far less common in the upper urinary tract than urothelial tumours. Their incidence varies from 0,7 to 7%. In the renal pelvis they occur approximately six times more frequently than in the ureter. They can originate from leucoplakia or straight from urothelium. Squamous cell carcinomas are frequently associated with infected staghorn calculi that have been present for a long duration. Squamous cell carcinomas are often invasive and soon metastasise. Therefore their response to treatment is rather poor. Adenocarcinoma in the ureter is extremely rare. As with squamous cell carcinoma, adenocarcinoma usually is associated with calculi, long-term obstruction and inflammation. In 70% of cases adenocarcinomas excrete mucus. They usually infiltrate the ureteric wall and metastasise early. The response to radiation or chemotherapy is poor. Nonepithelial tumours are hardly ever spotted. The following cases have been described so far: leiomyosarcoma, carcinosarcoma, leiomyo-mas, neurofibromas, plasmocytomas and angiosarcomas. Symptomatology and diagnostic procedures of ureteric tumours The most common symptom of upper tract urothelial tumours is gross or microscopic hematuria. It occurs in about 75% of patients suffering from the disease. Messing and Valencourt (1990) indicated that micro-hematuria was very likely to be found in all cases of upper urothelial tumours provided a sufficient number of urine examinations were carried out. About 30% of patients complain of flank pain, which is usually dull and lasting. Acute colic caused by massive bleeding from the tumour and resulting obstruction of the collecting system by a blood clot is more typical of collecting system tumour rather than of ureteral tumours. Prostration and invasion of periureteric tissue are observed in patients with symptoms of advanced disease. Occasionally the symptoms are due to metastasis. Nothing is usually found on the physical examination. The tumour is perceived below the ribs or in the flank only in very advanced stages of the disease. About 10-15% of patients are asymptomatic. The tumour is diagnosed incidentally during an examination undertaken for some other reasons. Since symptoms characteristic of urothelial tumours are very scarce imaging studies become particularly useful as diagnosing procedures. Excretory The most common radiological examination is excretory urography. Ureteric tumours cause obstruction and nonvisualisation of the collecting system. In extreme cases total loss of renal function is observed. The differential diagnosis includes ureteric stricture, filling defect, radiolucent stone, blood clot, sloughed renal papilla or fungus ball. If excretory urography is not revealing due to loss of renal function caused by the ureteric tumour retrograde urography is recommended. Contrast material should be injected via a ureteric catheter. The ureteric orifice should be checked as some bleeding might occur. Retrograde urography visualises the ureter below the tumour and if the stricture is not total the assessment of the malignancy length is possible. According to Murphy retrograde urography makes it possible to diagnose the ureteric tumours in 75% of cases. Antegrade pyelography is not advisable in patients suspected of having upper tract transitional carcinoma because th risk of seeding tumour cells along the needle tract is considerable. Computed tomography is less useful because of small volume of the tumour mass. However, in some cases is helpful. Transiotional cell carcinomas are recognised as soft tissue masses with an average density of 46 HU and a range of 10 - 70 HU. Uric acid stones, which are radiolucent on standard urography, are opaque on CT scans because their radioden-sity is usually greater then 100 HU. Neither ultrasonography nor NMR is performed as they do not offer any material advantage over CT in the diagnosis and staging of patients with upper tract urothelial tumours. Recently endoscopic procedures have been used increasingly in the diagnosis of ureteric tumours. Both rigid and flexible ureteroscopes are used. According to Blute the correct diagnosis of ureteral tumours based on ureteroscopy was made in 90% of cases. The procedure makes it possible to localise the tumour, to assess its morphological features and to perform the biopsy. On the other hand ureteroscopy is an invasive procedure requiring full anesthesia. The risk of potential complications is high (Blute's sample: 7%). Even trivial amount of hematuria can sufficiently cloud visibility, which makes the biopsy impossible. Small tissue fragments obtained by means of small endoscopic biopsy forceps may be difficult for the pathologist to interpret. Thus diagnostic ureteroscopy should be reserved for patients in whom the diagnosis remains in doubt after using conventional diagnostic techniques. Another noninvasive diagnostic procedure commonly performed is cytopathology. However with low grade tumours the cytology is read as normal in up to 80% of patients. As with bladder cancers, there is a correlation between tumour grade and positive cytology results. Thus in diagnosing primary tumours it is of little help. The accuracy of the examination increases significantly the successive tumour grades. In patients with grade III tumours the specificity of cytopathology varies, according to different authors, from 60 - 87%. Ureteral catheterization for collection of urine directly from the upper urinary tract provides more accurate cytologic results, but is still associated with substantial false negative and false positive findings. The drawback of cytology is its low specificity. False positive results are obtained in patients with urinary stones. Hematuria, bacteriuria, crystalluria and alkaline reaction of the urine make the assessment of the sample difficult. Finally, mention should be made about brush biopsy introduced by Gill.With this technique, a fine brush mounted of the end of the guidewire is passed through a ureteral catheter into the ureter. The lesion is sampled by moving the brush back and forth within the catheter. Sheline reported that brush biopsy had a sensitivity of 91% and a specificity of 88%. As ureteroscopy has become easily available brush biopsy is no longer as popular as it used to be but performed in some urological centres due to the low complication rate. Treatment of ureteric tumours Ureteral tumours almost always produce symptoms and signs, that need additional examinations and then allow to establish diagnosis in comparatively short time. Diagnosed upper urinary tract urothelial tumours must be cured. So far collecting data let to make following generalisation. With low-grade and low-stage tumours good results are achieved either with conservative or radical surgery. Intermediate tumours do better with radical surgery. Bad results are observed in group patients with high-grade, high-stage tumours irrespectively of chosen sort of treatment. Traditional management of upper urinary tract urothelial tumours is nephroureterectomy with excission of bladder's wall cuff, that surrounds orifice of the ureter. If one performs this operation it is important not to open ureter and avoid implantation of neoplasmatic cells into surrounding tissues. Cystotomy anterior wall of bladder facilitates removal of distal and intramural part of the ureter as well as minimalises the risk of injury the lower part of contralateral ureter. 5-years survival rate in patients treated with total nephroureterectomy in stage Tis, Ta, Tl was 91%, in stage T2 was 43%, in stage T3, T4 and Nl or N2 was 23%. Conservative methods of surgery have become more popular since seventies. They are used for low-grade, low-stage tumours of upper and middle or distal part of the ureter. Partial nephrectomy is performed if tumour is localised in pelvis. Local recurrences are observed in 25-40% of cases, more often in renal pelvis tumours. Recurrent tumours are successful treated with repeat local excision. Conservative methods are especially appropiate for patients with solitary kidney (anatomically or functionally), bilateral or small low-grade tumours. Upper urinary tract urethelial tumours are treated also endoscopically. Ureteroscopic method is preferred.It allows not to disseminate neoplas-matic cells to adjacent tissues, if one avoids perforation of the ureter wall. Low-grade tumours are removed using ureteroscope. Patients should be in rigorous follow-up because of high recurrence rate and mul-tifocality of ureteral tumours. Percutanous treatment of renal pelvic tumours is accepted only if there is a small, solitary, lowgrade tumour and ureteroscopic access encounters problems. Percutanous method needs full endoscopic control of ureter and bladder. Instillation therapy with cytotoxic agents or BCG is nowadays experimental method without determined indications and efficacy. This kind of treatment can be indicated in patients with multiple, superficial tumours or carcinoma in situ, if there is disturbed renal function or tumours are bilateral. For topical treatment percutanous access (nephrostomy tube) rather then retrograde one (ureteral catheter) is preferred. The first one reduces the number of complications. Systemic chemotherapy according to M-VAC regimen, as in urothelial bladder malignancies, can be potent in advanced (T3-T4) tumours. Durable complete response was observed in 5-10% of patients. Small groups patients with upper tract tumours make impossible the conduction of prospective, randomised trials. Adjuvant radiotherapy with doses of 37 to 60 Gy can minimalize the local recurrence rate after total nephroureterectomy for high-stage and high-grade tumours. Problems with radiotherapy are related to the possibility of injuring neighboring organs and large surface of irradiation. Positive cytolgic findings without endoscopic or radiographic evidence of tumours should be only followed closely. Epidemiology of the urinary bladder malignancies Carcinoma of the bladder is the most frequent neoplasm of the urinary tract. According to United States statistical data it is fourth most common neoplasm among men (5,5% cases) and eight among women (2,3% cases). Carcinoma of the bladder is three times more common among men then women. Neoplasm of bladder can occur at any age of patients but it is most common in elderly persons (55 years and older) and incidence of bladder cancer increases directly with age. The median age at the time of diagnosis is, depending on the country, 60-70 years. The neoplasm diagnosed at age below 30 years is usually low-grade tumour with a good prognosis, but, as in older population, it depends on stage and grade of the tumour. The rise in incidence of bladder cancers is observed in the last 20 years. This rise in incidence of bladder cancer is not dependent on better diagnostic methods. Aging of society influences this incidence, but it is hard to explain fact, that it refers only to men. In population of women, whose life expectancy is longer then men, who are nowadays exposed to industrial and environmental carcinogenes in the same extent, there is a declining incidence of bladder cancer of 2% annually. Bladder cancer is diagnosed 1,5 to 2 times more common among whites then among blacs, depending on gender. Carcinoma in the first group, although is detected more often, in greater percentage is noninvasive. Among black bladder cancer is diagnosed in more advanced form. It doesn't depend on delay in establishing the diagnosis but is rather related to different, more aggressive biology of neoplasm. In 1995 50 500 new cases of bladder cancer were detected in United States. The bladder cancer was the cause of death of 11 200 patient and this accounts for 2,6% all cancers death in man and 1,4% in women. In the same time, in Poland, bladder cancer was diagnosed at 3828 persons (it consists 2,8% of incidence of all malignancies) and was the cause of death of 2252 (it accounts for 3,6% all cancer deaths). Five year survival rate is highest in white men (84%) and the lowest in black women (51%). The difference in survival rate can be related to the fact, that in blacks bladder cancer is seldom diagnosed in less advanced stage and more often exists in more aggressive or other then urothelial form of malignancies, such as squamous cell carcinoma or adenocarcinoma. The treatment last ones malignancies is less effective then urothelial tumours. Additionally, economic conditions can make inaccessible adequate specialistic treatment. During last 50 years incidence rate of bladder cancer has increased of approximately 50% but mortality has decreased of approximately 33%. It is hard to state, to which factors - changing in the biology of cancer, earlier diagnosis, better treatment or alteration in risk factors this phenomenon can be attributed. Symptomatology and diagnostic procedures in urinary bladder tumours Bladder tumours develop secretly. The first most common symptom is hematuria of changeable intensity, often accompanied by bladder irritability, urinary frequency, urgency and dysuria. No connection between the intensity and frequency of hematuria and the stage of the tumour was found. Urinary tract infection is observed in 30% of patients. Pain appears in advanced stages of the tumour. When it is located in the suprapubic region it signals that the tumour infiltrates the perivesical tissues. Flank pain, often accompanied by fever, is due to the ureteral obstruction. As the symptoms of bladder tumours are rather sparse, what seem fundamental in the diagnosis are additional examinations. Exretory urography Urography with cystography are performed in all patients suspected of bladder tumours. The procedures provide information about the renal function and the morphology of the urinary tract. Ureterohydronephrosis signals the muscleinvasive growth of the tumour in 70-90% of cases. Filling defects in the central part of the cystogram can indicate the papillary growth of the tumour. Marginal filling defects are typical of flat tumours, which are always invasive. The most common method used in the diagnosis of bladder tumours is ultrasonography. Sonography Sonography Transabdominal sonography makes it possible to identify exclusively exophytic tumours whose diameter exceeds 10 mm. Transurethral sonography is a more discriminating method. It helps to determine the depth of the tumour invasion into the bladder wall. Cystoscopic view The most revealing diagnostic procedure is cystoscopy. It shows the size of the tumour, its appearance and its surrounding. If a biopsy is also applied the histology of the tumour as well as its grade can be determined. Biopsy (cystoscopic view) The biopsy of non-affected parts of the bladder should be obtained in search of carcinoma in situ. The pathological stage of the tumour must be precisely estimated. This can be achieved by means of transurethral resection. After the procedure a pathologist checks the removed tissues as well as the material taken from the place where the tumour was located. The histological grade determined in this way is consistent with the grade of the tumour removed during the cystectomy. The tumour grade is adequately estimated during the transurethral resection in 96% of patients with superficial carcinomas. The procedure is much less accurate when the tumour infiltrates the bladder muscle or perivesical tissues. In such cases the differentiation between T2/T3a and T3b is successful in 57% of patients. Bimanual examination is still useful as it helps to approximate the local advancement of the tumour. Computed tomography is of little use in the diagnosis of bladder cancers. CT scanning is limited in accuracy because it can detect only gross extravesical tumour extension. CT scans fail to detect nodal metastases in up to 40% of patients having them. Cytological examination is particularly useful for patients who underwent treatment due to bladder cancer. It may also be used as a screening procedure for people who are especially vulnerable to bladder cancers because of their professions. The sensitivity of the method increases with the tumour grade. In grade 3 tumours it varies from 60 to 87%. The drawback of cytological examination is its low specificity. Classification and principles of the treatment of the urinary bladder tumours Staging of the urinary bladder tumours In description of staging urinary bladder tumours system T (estimation of the tumour), N (estimation of the regional lymph nodes metastases), M (estimation of distant metastases) is commonly used. This system have been worked out by UICC (International Union Against Cancer). The following stages are distinguished. Tumour estimation (T): Ta - papillary, epithelial confined tumour, Tis - flat, in situ carcinoma, Tl - tumour invades lamina propria of urothelium T2 - tumour invades superficial muscle layer (inner half), T3a - tumour invades profound muscle layer (outer half), T3b - tumour invades perivesical fat, T4 - tumour invades pelvic viscera like rectum, pelvic walls, prostatic stroma or uterus. The symbol TO is applied, if tumour haven't been found in postcystec-tomy specimen and Tx, if there is no data regarding bladder tumour. Regional lymph nodes estimation NO - there are no metastases in regional lymph nodes, Nl - there is single positive node less then or equal to 2 cm in diameter, N2 - there is single positive lymph node greater then 2 cm but less then 5 cm in diameter, or multiple positive nodes less then 5 cm in diameter, N3 - positive lymph nodes greater then 5 cm in diameter. Symbol NX means, that status of nodes is unknown. Regional lymph nodes are those located in pelvis minor, below the bifurcation of common iliac arteries Metastases estimation The presence of distant metastases (visceral or in lymph nodes above bifurcation of the aorta) is recorded as Ml and the lack of them as MO. MX is used if there is uncertainty regarding distant status. In order to specify staging of the bladder tumour, besides clinical examination, USG and bimanual investigation of patients, following additional procedures are performed: 1. transurethral diagnostic (primary) resection 2. transurethral ultrasonography 3. computed tomography of pelvis minor, retroperitoneum and liver 4. magnetic resonance of pelvis minor and bones - sites suspected of the metastases, 5. chest radiograph. Preoperative staging of bladder tumours is often underestimated (about 30% of cases) or overestimated (about 10% of cases). Strategy of the treatment of the urinary bladder tumours. The discrimination between noninvasive tumours (Ta,Tl) and invasive ones (T2-T4) is very important because of the difference in the treatment of them. Additionally, on the method of treatment influences grading of neoplasmatic cells atypia (symbol G). Noninvasive tumours can be treated with preservation of the bladder. The basic curative method is transurethral resection of the tumour. Beside diathermic current recently energy of laser radiation is used to destroy neoplasmatic, former photosensitized cells. In the case of recurrent, high grade tumours as well as the prophylactic of recurrences, intravesical cytostatic (doxorubicin, mitomycin c, thio-tepa) or immunostymu-lated agents (suspension of the tuberculosis germs bacilli - BCG therapy) are used. Big tumours can be removed with a fragment of bladder wall (partial cystectomy). High graded superficial tumours, early or often recurrent, presenting progression of staging or grading are the indication to radical cystoprostatectomy and urinary diversion in some urology departments. The procedure of choice in invasive tumour of bladder is radical cystoprostatectomy (including regional lymph nodes removal). For advanced forms of disease the systemic chemotherapy, based on M-VAC regimen (metotrexat, vinblastin, doxorubicin, cisplatin) is used as adjuvant (after surgery) or neoadjuvant (before surgery) supported form of treatment. In highly selected group of patients bladder-sparring methods are tested. Usually the combination of surgery - transurethral resection or partial cystectomy - with radiation or/and systemic chemotherapy are examined. Radiotherapy is the basic method of treatment in certain countries. Results obtained with it are poorer regarding survival rate and complications rate in comparing to radical cystoprostatecomy. Patients with advanced bladder cancer may suffer mainly from pain and intractable haematuria, if the bladder haven't been removed. Palliative external radiotherapy is not recommended so as it is ineffective and produces many complications. Massive haematuria, if transurethral ful-guration is insufficient, may be treated with intravesical instalations under anesthesia of silver nitrate, 1% alum and 4% formalin solutions. Transdermal embolisation of arterial vessels is also recommended. Surgical treatment of urinary bladder tumours Three methods of surgical treatment are commonly used in bladder cancer. These are: transurethral resection, partial cystectomy and radical cystoprostatectomy. The choice of the appropriate procedure is determined by the following factors: tumour stage, grade and multifocal growth. Such tests as DNA flow cytometry, ABH blood group and Thomsen-Friedenreich (T) antigen expression, DNA synthesising (S-phase) fraction and abnormalities in p53 gene are less useful in every-day clinical practice. Most authors believe that radical cystectomy is the treatment of choice in invasive bladder cancer. What is identified as invasive cancer is the tumour which infiltrates the bladder muscle. According to TNM classification (1989) it represents stage T2-T4. Transurethral resections are performed in cases of superficial caricinomas whereas radical cystectomy is reserved for invasive tumours. In selected cases partial resection of the bladder is performed. However, the division presented above is fairly simplistic. Local recurrence was observed in 50-80% of patients with bladder cancer - stage Ta and Tl. In 25% of these the disease progressed. Patients with cancer recognised as stage Tl and grade G3 were still alive 5 years after the operation only in 50-60% of cases. It is a well-known fact that noninvasive tumours which are confined to the lamina propria of the bladder, usually regarded by urologists as superficial, may metastasise into the lymph nodes. Smith and Whitmore reported that kind of development in 5% of patients who had undergone radical cystectomy. Lerner diagnosed metas-tases in the lymph nodes in 4% of patients with bladder cancer defined as stage pTa, pTis or pTl. These findings suggest that carcinomas infiltrating lamina propria should be treated as advanced, which means that they require procedures more radical than transurethral resection. On the other hand, some urologists believe that transurethral resection in association with chemotherapy can be an alternative to radical cystectomy as it proves to be successful in the treatment of some patients, even those with invasive bladder cancers. Transurethral resection Transurethral resection as primary therapy should be reserved for patients who have small, solitary, low grade superficial carcinomas and bladder papillomas. The operation is frequently performed in two stages. Stage I involves the resection of the bladder tumour. Stage II is the resection of the scar resulting from the first operation. The procedure makes it possible to remove deep layers of the bladder muscle, which renders the treatment more radical. Transurethral resections are performed in spinal anaesthesia. They are well tolerated and do not require a blood supply. Patients do not stay in hospital longer than a few days after the operation. The combination of transurethral resection with chemotherapy and irradiation was introduced in order to save the bladder. However, the treatment proved to be less successful than expected. Partial resection of the bladder Partial resection Partial resection of the bladder is performed rarely. The treatment is reserved for patients with solitary muscle-infililtrative tumours localised on top of the bladder, far from the trigone or vesical neck. No evidence of carcinoma in situ can be recognised in the remaining part of the bladder. A tumour-free margin of 1,5 to 2,0 cm must be obtained. The results for patients selected carefully for the procedure were not inferior to those of cystectomy. In order to improve the therapeutic results partial resection was combined with chemotherapy. Herr and Scher reported that the combination gave five-year survival in 64% of patients. All of them preserved their own well-functioning bladder. Because of the multifocal growth of bladder cancer, patients undergoing partial cystectomy must be followed by careful surveillance cystoscopies as intravesical recurrences are common. Herr and Scher observed such recurrences in 46% of cases. Radical cystectomy Radical cystectomy in the treatment of bladder cancer has been performed for more than a hundred years. The first operation of that type was carried out by Bardenheuer in 1887. Ineffective anaesthesia, lack of antibiotics and problems with urinary diversion were the reasons for the postoperative mortality rate of 50-60% at the beginning of the century. In 1939 von Hinmen reported that 34,2% of patients who had undergone cystectomy died directly after the operation. In the 60's the postoperative mortality rate was already 10%. Nowadays, in most urological centres it is about 1%. The operation consists in the resection of the bladder together with the prostate and seminal vesicles. Abdominal assess is used. In female patients, the standard operation for invasive bladder cancer is an anterior pelvic exenteration with wide excision of the bladder and urethra in continuity with the uterus, fallopian tubes, ovaries, and anterior wall of the vagina. The connection of the tumour stage with lymphatic metastases is commonly known. In patients with tumours superficially infiltrating the bladder muscle lymph node metastases were observed in 6-36% of cases. In patients with more advanced carcinomas lymph node metastases were diagnosed in 50% of cases. These findings made urologists try to improve the therapeutic results. Thus cystectomy was combined with pelvic lymphadenectomy. The term radical cystectomy denotes the operation involving a total resection of the bladder with the prostate and seminal vesicles and pelvic lymphadenectomy. Many authors used to believe that multifocal growth of the urothelial tumours required preventive urethrectomy in addition to cystectomy. As nowadays neobladders are commonly made, urethrectomy is performed only when the tumour infiltrates the urethra. Even though cystectomy is a serious and mutilating operation it is applied more commonly now because it has become safer (lower mortality rate) and more comfortable to the patient. The implantation of the ureters to the skin as well as the methods consisting in connecting the urinary tract with the alimentary system have been superseded by new methods of urinary diversion described by Bricker (1950), Couvelaire, Cibert and Carney. Kock and Le-isinger described the method of making a continent urinary reservoir called Kock-pouch. In 1985, urologists from Mainz described a new method of making a continent urinary reservoir shaped from the coecumand the terminal part of the ileum. The method was related to the operation performed by Gilchrist in the 50's. It is known as Mainz Pouch. Rowland from the University of Indianapolis modified the operation in that the reservoir was made from the coecum and a part of a colon. The modification is known as Indiana Pouch. Recently neobladders joined to the urethra have been used. The operation makes it possible to avoid urostomy, which mutilates the patient. However, neobladders cannot always be made. As cystestomy used to cause impotence in almost 100% of men, a number of patients did not accept the operation. The nerve-sparing modification introduced by Walsh reduced the risk of impotence as a side effect of cystectomy. The modification consists in that vesical arteries and bladder pedicles are transected immediately adjacent to the seminal vesicle and ureters to avoid injury to the cephalad portion of the neurovascular bundles. Mention should be made of the combination of cystectomy with radiotherapy. This integrated therapy was introduced by Whitmore. The method was used in the 60's and 70's in many urological centres in the world. It was expected to improve the therapeutic results in patients with bladder cancer as well as to reduce the percentage of local recurrences after cystectomy. The bladder was irradiated with 40 Gy over the period of four weeks. The operation was performed four weeks after the irradiation had been completed. 'Short' irradiation lasted for a week, the dosage being 10-20 Gy. In those cases cystectomy was made a few days after the irradiation. The results of the integrated therapy were promising. Whit-more reported a significant drop in the percentage of local recurrences (37% vs. 16%) over the period of 20 years. Five-year survival of patients with bladder cancer increased during that time, too. The theoretical as-f sumption underlying the therapy is that irradiation destroys tumour cells in the lymph nodes, thus preventing their dissemination during the operation. In addition, radiotherapy may diminish the mass of the tumours that were regarded as non-operative prior to the irradiation. However, a number of authors were rather sceptical about the superiority of the integrated therapy over mere cystectomy. In 1984 Montie presented the therapeutic results achieved over the period of 20 years in 99 patients who had undergone radical cystectomy without prior irradiation. The percentage of local recurrences in those patients was only 9% and five-year survival in T3, T4 tumours was 40%. Similar results were observed by Mathur. When comparing the integrated therapy (cystectomy + irradiation-16Gy) with radical cystectomy in the analysis of 197 patients with bladder cancer, Skinner found no statistically significant survival advantage in either of the methods. Thus the better results reported widely have been attributed to better surgical techniques, better selection for the operation, progress in anaesthesia and intensive care rather than to the combination of cystectomy and radiotherapy. Nowadays the integrated therapy in bladder cancer is not particularly common. Instead, radical cystectomy associated with neoadjuvant or adjuvant chemotherapy is applied. At present radical cystectomy appears to be the most effective therapeutic option for patients with invasive bladder cancer. Five-year survival is assessed by different authors as 30-54% of cases. However, the operation is dangerous, technically difficult, for which the risk of perioperative morbidity is high. What seems to be of primary importance is to determine strictly the indications for the operation. These are: 1. invasive bladder cancer irrespective of the tumour grade, 2. recurrences of the tumour after transurethral resection, particularly when the grade increases, 3. high grade tumours coexisting with carcinoma in situ, 4. multifocally growing superficial bladder cancers resistant to intrave-sical chemo- or immunotherapy administered after transurethral resection, 5. recurrences of carcinoma in situ following chemo- or immunotherapy. It seems that nowadays the radical treatment tends to be increasingly applied even in early stages of bladder cancer. Current research on the biology of neoplasms might help to determine the progression risk factors, which in turn will make it possible to decide which patients require the resection of the bladder and which can be administered a less aggressive treatment. Radiotherapy of the urinary bladder tumours Cure rates of up to 80% can be obtained with surgery for T1-T2 well differentiated tumours where lymph node involvement is uncommon. Poorly differentiated or multiple T1-T2 tumours may be treated by local radiotherapy to the bladder and perivesical tissues with five- year survival rates of 40-60%. Lymphatic involvement occurs in about 60% of patients with T3 tumours and radiation is usually given to the whole pelvis. Five-years survival rates after radiotherapy, however are less than 35%. External -beam radiotherapy is often first line therapy in Great Britain and Europe, with salvage cystectomy reserved for treatment failures. Analysis of clinical characteristics has defined several prognostic factors that predict for local control freedom from metastasis and survival in patients treated with definitive irradiation. Most series show that depth of tumour's invasion defined by stage is an important prognostic indicator. Other poor prognostic sign include tumour greater than 5 cm, residual disease after TURB, and radiographic evidence of ureteral obstruction with hydronephrosis. In an effort to improve local control rates with external - beam radiotherapy for patient with invasive bladder cancer hyperfractionated treatment schedules have been used. Edsmyr and coworker reported a Swedish trial of 168 patient with T2 to T4 disease randomly assigned to receive either 1 Gy three times a day to a total of 84 Gy or 2 Gy once a day to a total of 64 Gy. Both regimens were given over 8 weeks with an imposed mid-course 2-week treatment interruption. The 5year survival for the group receiving 84 Gy was 37% and for those receiving 64 Gy it was 16%. Patients with T3 disease particularly appeared to benefit from the hyperfractionated schedule. A study in Great Britain has tested an accelerated hyperfractionated regimen in 24 patients with muscle invasive bladder cancer. Over a 22-day period, patients received doses of 1.8 to 2.0 Gy per fraction to total doses of 54 to 64 Gy to the bladder and 39.6 to 44 Gy to the whole pelvis. Local control was, 56% and survival was 35% at 2 years. Simulation and treatment During simulation and treatment, the patient is supine and the bladder is emptied to ensure reproducibility. At the time of simulation a Foley catheter is inserted and 25 to 30 ml of radiopaque contrast material is instilled in addition to 10 to 15 ml of air for bladder visualisation. A rectal tube with barium contrast medium is placed to visualise the rectum. In general, a four- field box approach is used (Anterior posterior and lateral fields) delivered through 6 to25 MV X-rays. A lot of favour the four field box because of the ease of interpreting imaging films, the ease of making portal size reductions, and the satisfactory dose distribution. Other techniques of irradiation are possible e.g. anterior posterior, rotational, three-field portal arrangements. The 360-degree arc rotation technique was significantly associated with an increased risk of severe complications. Preoperative radiotherapy The rationale for using this neoadjuvant irradiation is to reduce the incidence of tumour bed failures and potential seeding of malignant cell from the operation. Some of retrospective studies have suggested that the addition of preoperative radiotherapy improves the overall outcome, other series have found no benefit when results are compared with modern surgical series. Several prospective trials that compared preoperative radiotherapy plus radical cystectomy to radical cystectomy alone have demonstrated no significant differences between the treatment arms in two studies of so called sandwich irradiation patients with clinical B2, C, D disease received sequentially 5 Gy (single fraction) preoperatively, radical cystectomy, and 45 Gy postoperatively. The 5- year disease -free survival rate for these patients was only 20%. Preoperative irradiation is recommended for T2, largo =4 cm tumours, highgrade lesions, T3 and resectable T4. Complication of radiation therapy Radiotherapy is generally well tolerated, most patients experience dy-suria, urgency, urinary frequency, and diarrhea as acute self limiting symptoms. The incidence of long -term complication is acceptable when proper treatment techniques are used. In general, such complications become clinically manifest within the first 3 years after radiotherapy. Shipley reported an 11% incidence of chronic grade III-IV genitourinary complications among 35 patients with locally advanced bladder cancer treated with conventional fractionated therapy. Palliative Radiotherapy - is for rapid relief of pain or hematuria with minimal incovenience to the patient. This can be accomplished with a short course with a high dose per fraction.: 17 Gy in 2 fractions over 3 days, 30 Gy in six fractions of two fractions per weeks, 21 Gy in three fractions over 5 days. Many suffered acute side effects including diarrhea, vesicovaginal fistulas, contracted bladders. Brachytherapy Interstitial treatment may be used alone, in combination with low or moderate dose external irradiation, or to treat the suturejine in patients undergoing partial cystectomy. In carefully chosen patients, the technique has provided excellent tumour control and bladder function. Experience with radium needle implants has been reported by Van der Werf Messing from Rotterdam. 328 patients with T2 and 63 patients with T3 muscle invasive bladder cancer were treated with preoperative radiation of three fractions each at a dose of 3.5 Gy followed by radium. Results local recurrence at 5 years was 16% for T2, and 28% for T3 tumours. Survival -56% of patients with T2, and 37% of those with T3 disease alive at 5 years. (Survival was similar to patients undergoing definitive external-beam radiotherapy or cystectomy.) Other isotopes using in the brachytherapy: cesium (Cs -137) iridium (Ir-192). Mazeron treated 30 patients with T2 and 5 with T3 disease using a single fraction of 8,5 Gy, followed by partial cystectomy and iridium implantation, and attained excellent local control. Rozan reported a multicenter French experience that included 98 patients with Tl, 66 with T2, 26 with T3a, 9 with T3b, and 1 with T4 disease treated by preoperative external beam radiation, partial cystectomy with lymphadenectomy, and placement of an iridium implant into the tumour bed. Preoperative radiation was targeted to the bladder in 44 patients and to the pelvis in 161 patients for a mean total dose of 11 Gy, and a mean dose per fraction of 5,4 Gy. Of the 36 tumour recurrences, 9 were located and the original site, and 19 occurred in a different site. They reported an overall 5- years survival of 67%. Survival was 77% for pTl, 43% for pT3. Serious complications: hematuria - 8, chronic cystitis -17, fistulas - 11 patients. Local treatment of the urinary bladder tumours (chemotherapy and BCG-therapy) Introduction Superficial bladder cancer has a high tendency to recur, and in significant subset of patients, to progress. Analysis of the clinical risk factors of superficial bladder cancer, such as stage, grade, size and multifocality is crucial tc predict the course of the disease. The field of topical therapy can roughly be divided into the chemotherapeutic and immunotherapeutic approaches. Chemotherapeutic and immunotherapeutic agents have been used in the therapeutic ablation of existing or residual tumours and in prophylaxis against their recurrence. Intravesical therapy can also be used tc treat existing superficial bladder tumours. Currently, this is reserved primarily for the management of carcinoma in situ, which is often almost impossible to resect endoscopically in its entirety or for the rare patieni with so many superficial tumours that endoscopic resection is not feasible and cystectomy is either not appropriate or not desired by the patients. With these exceptions, however, intravesical therapy is used primarily in an adjuvant form, although this may often be threatening existent or persistent disease that is just not yet detectable. Adjuvant intravesical therapy or intravesical immunotherapy is indicated in patients who are at a high risk for tumour recurrence by virtue olhaving multiple tumours, recurrent tumours, high grade tumours associated with urothelial atypia, or carcinoma in situ. Intravesical chemotherapy Since 1948, when Semple first used podophyllin in an attempt to reduce the recurrence rate of superficial bladder cancer, many different agents have been employed. Generally, intravesical chemotherapy began in the 1960 with the introduction of intravesical thiotepa. From many different substances used in treatment, only the most frequently used and successful will be described. Table 1 summarises the advantage and disadvantages of the compounds that have been studied for chemotherapy and chemoprophylaxis. All agents for intravesical chemotherapy are about equally effective. The possible exception is that mitomycin may be marginally superior to thiotepa in the treatment of patients with stage Ta tumours and for high-grade tumours. When used as prophylaxis against tumours recurrence, recurrence rates have been reduced to 30-44% compared with about 70% in controls. However, the study by Pawinski et al.(9) reported on 2535 patients from the European Organisation for Research and Treatment of Cancer (EORTC) and Medical Research Council studies on chemotherapy with a median follow-up of 7,8 years, who were analysed for the duration of disease-free interval and survival, with regard to recurrence, the overall advantage of chemotherapy over TUR alone was 6%. Nowadays, little evidence exist to indicate, that any type of intravesical chemotherapy prevents progression to muscle invasion. From analysing a variety of randomised, controlled studies that collected progression data, it appears that patients treated with surgery alone and patients treated with both surgery and intravesical chemotherapy experience roughly a 7% chance of developing muscle - invasive disease . Table 1: Advantages and disadvantages of therapeutic compounds used in treatment of bladder cancer Molecular Name Advantages Disadvantages Therapeutic use weight Systemic absorption leading Thiotepa Small Inexpensive to myelosuppression and +/renal failure Systemic side-effects; Epodyl Small Inexpensive +/myelosuppression AdriaMinimal Large Chemical cystitis; expensive ++ mycyn absorption Minimal Epirubicin Large Chemical cystitis; expensive ++ absorption Mitomycin C MitoxantronC Minimal Chemical cystitis, bladder (absorption ulceration; expensive Minimal Large Chemical cystitis, expensive absorption Relatively Local toxicity, BCG-itis BCG Large inexpensive (absorption) Cytokines Relatively Effective Systemic toxicity; extremely Interferon small expensive Large ++ ++ +++ + Intravesical Bacille Calmette-Guerin (BCG) therapy For many years it has been known that patients dying of tuberculosis have a very low incidence of incidentally discovered tumours at autopsy. In 1929 it was suggested that response of the body to the tuberculosis bacillus might convey benefit in preventing cancer. In 1976 Morales et al. presented the first results of intravesical BCG instillation in the treatment of superficial bladder tumours. Subsequently Lamm et al. and Brosman, amongst others, reported successful prophylaxis of recurrent bladder tumours with BCG instillations. BCG is an attenuated strain of Mycobacterium bovis that has stimulatory effects on immune responses. Several strains of BCG have been used including the Pasteur, Tice, Armand-Frappier and Moreau strains. All are derived from the original strain developed at the Pasteur Institute and the precise mechanism by which BCG exerts remain unknown. The fact that intravesical therapy without scarification or conversion of previously negative Mantoux test, is as effective as in the other circumstances, suggests that it is a purely local mechanism. Some studies suggests that one mechanism involves the release of cytokines into the bladder, belonging to both the interferon and inrleukin families. BCG is commonly given in three clinical settings: prophylaxis in tumour-free patients, treatment of residual tumour in patients with papillary TCC and treatment of patients with carcinoma in situ. Intravesical BCG treatment, compared with many other instillations, does sometimes cause major side-effects and patients must be seen regularly. Although most patients only suffer bladder irritation, irreversible bladder contracture and systemic BCG-itis may occur. Such severe symptoms requiring antituberculous therapy occur in up to 6% of patients. BCG is perhaps most useful in the treatment of patients with carcinoma in situ In several studies BCG induced complete response rates in about 72% of patients with ca. in situ. Intravesical BCG in conjunction with transurethral resection of the prostate may be effective in the treatment of patients with carcinoma in situ involving the prostatic urethral mucosa or prostatic ducts. Conclusion Because well-differentiated and moderately differentiated superficial tumours recur after complete endoscopic resection in roughly 50% of patients but rarely progress to more aggressive disease, it would make sense that intravesical therapy be withheld in such patients until the frequency or multiplicity of recurrences becomes so great that the expense, discomfort, or risk of repeated TUR exceeds that of intravesical therapy. Alternatively, because high-grade lesions, particularly Tis or stage Tl, have substantial risk of stage progression, intravesical therapy is indicated immediately. Because only BCG has been shown to delay or prevent progression it should be the first agent used.