PTA 107 Topic 4: Heat and Temperature Intro Return Exams We will
advertisement

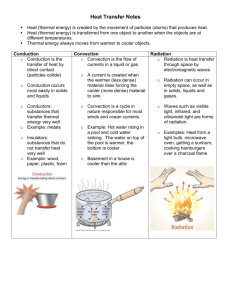
PTA 107 Topic 4: Heat and Temperature 1. Intro a. Return Exams i. We will not review them ii. Do not discuss them with your neighbors iii. Hand out the paper describing how you can get extra credit. b. Read the Case Study and the two articles i. Case Study: Cold Packs ii. Counting Calories iii. Moderating Effects of the Climatic Environment iv. You could be tested on the content of these articles 2. Review of Atomic Theory a. Substance – Matter that has a uniform and definite composition. Pure substances are composed of only one kind of matter. Each substance has its own property which does not change b. Molecule – the smallest unit of a substance, compound or element that can exist and still retain the characteristic properties of the substance. A tightly bound group of atoms c. Substances can be classified into two groups i. Elements – the simplest form of matter that exists in under normal conditions. Elements cannot be separated into simpler substances. They are the building blocks for all other substances. ii. Compounds – Pure substances that are composed of two or more elements that are chemically combined in definite proportions d. Mixture – a physical blend of two or more substances. A combination of different kinds of matter where the constituent substances retain their individual physical characteristics and can be separated by exploiting those properties. i. Homogeneous mixtures – uniform mixture throughout ii. Heterogeneous mixtures – composition of the mixture varies significantly throughout the volume 3. Interaction of Molecules a. Cohesion – The attractive force between the same kinds of molecules. Solids and liquids have stron cohesion. Cohesive forces prevent all matter from being gaseous. b. Adhesion – attractive forces between unlike molecules 4. Phases of Matter a. Solids – a fixed definite shape and volume. The shape does not depend on the shape of the container. Strong cohesive forces. Most solids expand slightly when heated. The mass and spacing of the molecules determines the solid’s density. Most solids are relatively uncompressible. b. Liquids – Materials that have a variable shap, but a fixed volume. Enough cohesion to hold the molecules together, but not rigidly. Liquids take the shape of their container. Most liquids are relatively uncompressible. c. Gases – Very weak cohesive forces. Molecules are relatively far apart. Gases are readily compressible. No fixed shape or volume. The term vapor is usually used for a gas that is normally in liquid form. d. There is a fourth state of matter: Plasma a fluid of charged particles as in “The Sun is a miasma of incandescent plasma” http://www.youtube.com/watch?v=sLkGSV9WDMA 5. Kinetic Theory of Matter a. Phet Physics States of Matter b. External Energy i. The normal bulk KE and PE that we have already discussed c. Internal Energy i. The total kinetic and potential energy of the individual molecules. It is associated with the random, disorder vibrational and rotational motion of the individual molecule. ii. The temperature of a substance is a measure of the internal molecular kinetic energy of the molecules. iii. At absolute zero molecular motion is minimized. 6. Heat – is a measure of the internal energy transferred from one body to another a. If the internal energy of a body is increased, it is heated. b. If the internal energy of a body is decreased it is cooled. c. Most typically internal energy is transferred by means of kinetic energy transfer. That is particles bouncing against each other. i. The exception is radiational heating or cooling. d. Heat refers to the transfer of internal energy, not on the amount of internal energy an object has. i. A hot iron does not contain a lot of heat, but it does contain a lot of internal energy. If you touch that hot iron with your hand, the rapid transfer of energy to your hand will result in a rapid increase in your hand’s temperature and give you a burn. More later when we discuss specific heat. 7. Temperature a. Temperature is a measure of the average kinetic energy of the molecules in a substance and can be measured with a thermometer or a calorimeter. i. The temperature is independent of the size of an object because it is an average molecular kinetic energy – averaged over all the molecules. ii. Two bodies at the same temperature can contain vastly different amounts of internal energy dependent on their mass and composition. iii. The temperature is a reflection of the translational and rotational energies of the molecules. 1. In gases, the temperature is a direct measure of the average KE of the molecules. 2. In solids and liquids, there may be some PE also involved (in particular during phase changes) iv. Temperature is not heat, although they are related. Temperature is a measure of how fast the molecules are moving/vibrating/rotating and heat is the amount of internal energy an object absorbs or releases. b. Temperature Scales – usually based on two standard reference temperatures (boiling and freezing of either water) i. Kelvin – defined by the temperature difference between absolute zero and the triple point of water and by definition 100 K apart. ii. Celsius/Centigrade – used by most countries in everyday measurements 1. Defined loosely by the freezing and boiling points of water and by definition 100 C apart. a. 1 K = 1 C iii. Fahrenheit 1. Defined loosely Defined loosely by the freezing and boiling points of water and by definition 180 F apart. iv. Temperature conversions 1. K to C: TK = 273 + TC 2. C to K: TC = TK - 273 3. F to C: TF=(9/5)TC + 32 4. C to F: TC=(9/5)(TF - 32) c. Facts regarding the human body control of temperature i. At T<20ºC, blood vessels I the skin constrict to decrease the blood flow near the surface, thus reducing heat loss via conduction. Skin hair is pulled upright by piloerector muscles reducing the wind speed next to the skin and further reducing heat loss by convection. If further warmth is required heat is produced by involuntary muscle contractions – shivering. ii. At T> 25ºC, the body must shed heat. Surface blood vessels dilate, increasing blood flow to the skin. This hastens the conduction of heat from the core to the skin which then loses the heat to the environment. iii. For T>29ºC sweating begins to cool the body by evaporation. The sympathetic nervous system controls sweating and can stimulate secretions up to 4 liters per hour. d. Test Question? i. What is considered a comfortable environmental temperature? ii. The actual responses to a cool, cold, warm or hot environment are influenced by 1. A person’s activity level 2. Age 3. Gender 4. Relative humidity 5. Air movement iii. What is the single most important comfort factor for the environment? iv. Why do the elderly seem to feel the cold more so than younger people? 1. Lean bodies ( muscles are good carbon burners and can produce more heat 2. Less body fat so less insulation 3. Poor circulation 4. Inactivity 5. Prescription drugs 6. Note the effect of “cold snaps” on the body (see notes) e. Thermometers i. Thermostat – uses a bimetallic strip (Demo) ii. Glass/mercury (liquid expansion) thermometer – fever thermometer is most accurate when clse to the “core” of a person. iii. Radiation 1. Uses blackbody radiation models to measure the temperature of the cavity in the ear by measuring two infrared wavelengths. (See Phet Physics Blackbody sim) 2. Demo with heat lamp and aquarium f. Summary of body temperatures i. The body controls temperature using thermoregulation processes to stabilize the core temperature. ii. Surface temperature is usually lower than core temperature. This lowers heat loss to the environment. iii. The body’s core temperature tends to decrease at night and the skin temperature increases at night. iv. Critical Temperatures Temperature Condition <33ºC Hypothermia, can affect metabolic processes 35-36ºC Core Temp in early morning 36 – 37.5ºC Normal Range during the day 38ºC Body Temp during moderate exercise 39-40ºC Body temp during hard exercise or a fever >42ºC Hyperthermia, can result in irreversible organ damage What are these temperatures in Fahrenheit? 1. Measures of Heat a. Heat is a form of energy transfer. b. Units of heat can be Joules, calories, Calories or BTU (Energy needed to raise 1 pound of water by 1 degree Fahrenheit) c. Define the Heat of Combustion as the energy released as heat when a compound undergoes complete combustion with oxygen (oxidation) under standard conditions. Expressed in kJ/kg d. The metric unit of heat is the dietary calorie. i. 1 calorie is the energy needed to raise 1 g of water by 1ºC. ii. The dietary calorie is 1,000 calories or 1kcal. 1Cal = 1,000 cal=1kcal iii. Important number: 1 Cal = 1kcal = 4,181 J iv. The process of digesting foods is essentially an combustion (oxidation) process. v. The nutritional energy from foods can be approximated by measuring their heat of combustion. (Burn a cashew?) vi. When listing the energy content of a particular food adjustments are made for parts of the food , such as dietary fiber, that are not digested. e. Energy Values of Major Food Groups i. Lipids (Fats) 37 kJ/g or 9 kcal/g ii. Protein 17 kJ/g or 4 kcal.g iii. Carbohydrates 17 kJ/g or 4 kcal/g f. Problem (Test problem?)The human heart’s resting power output is about 1.1 watts. What food energy in kilocalories is needed to operate the heart for 24 hours a day at this resting level, assuming the efficiency of the body for food energy use is 25%? i. 1.1 Watts 1.1 J/s ii. 24 hour = 86, 400 seconds iii. Energy needed in a 24 hr period is 1.1 Watts 24 hours 1.1 J/s 86,400 s 95,400 J iv. The body must consume 4 times that energy due to its low efficiency . v. Therefore 380,160 J of food energy must be consumed. 1 kcal 90.9 kcal of food energy must be consumed. 4,184 J vi. 380,160 J vii. Note: The work efficiency of the human body is defined as the ratio of mechanical work done by the body to the energy used for mechanical work plus the heat lost to the environment Work Efficiency Mechanical Work Mechanical Work Heat loss Work efficiency tends to be about 2 to 10%, but can be as high as 25% when engaged in sports. What does this mean? 2. Specific Heat a. This is a property of a substance. b. Defined as the amount of heat needed to raise the temperature of 1 g of the substance by 1ºC. c. The amount of heat needed to effect a certain temperature change in a substance is Q s m T i. Define the symbols ii. Work the units d. Specific heat does not depend on the size of the object (i.e. a small pond and a large pond are both composed of water so they both have the same specific heat. However, they woll have different heat capacities.) cal which is quite high for compared to most materials (e.g. g C cal cal cal Aluminum s= 0.217 , Lead s =0.11 , Ethyl Alcohol s=0.60 g C g C g C e. The specific heat of water is 1.0 f. The larger the specific heat the better a material is for a temperature regulator since it can absorb or release a large amount of heat over small temperature changes. g. Problem (Test Problem?) If a 165 lb person were assumed to have a specific heat of 0.8 cal/gºC, how much extra released internal energy would be required to raise his temperature from 98.6ºF to 104ºF? i. Convert ºF to ºC (3ºC) ii. Convert pounds to grams (1 lb = 2.2 kg) (75 kg= 75,000g) iii. Find the heat needed using Q s m T iv. Express the energy in kcals. v. Answer is 180 kcal (Note a Big Mac has 550 Calories) 3. Two heating methods a. Temperature difference i. Conduction: heat transfer through direct physical contact. Molecule to molecule transmission of energy, Occurs primarily in solids, Most insulators contain air spaces that inhibit the transfer of heat. Why does air (or any gas) conduct heat poorly – large spaces between molecules minimizes the frequency of collisions to pass on kinetic energy. 1. In Physical therapy conduction is used in hot packs, cold packs, paraffin, and ice massage. ii. Convection – heat transfer by the movement of a fluid due to fluid motion either from density fluctuations or external forcing. Common in liquids and gases (both fluids) 1. In Physical Therapy convection is used in hot and cold whirlpools and fluidotherapy. iii. Radiation - heat transfer from a warmer to a colder object through transmission of electromagnetic radiation without the need for an intervening medium. All objects whose temperature is greater that 0K radiate energy. All objects also absorp radiant energy to varying degrees. 1. In Physical Therapy radiation is used in the infrared lamp, laser. Ultraviolet therapy iv. Normally all three heat transfer methods work to contribute to the heat loss or gain of an object. 1. On a clear cold night your body will experience high radiation losses. If it is windy the losses will be even greater due to convection. You protect yourself with layers of clothing to lessen the conduction of heat. 2. Heat flows from the bodies core by conduction (and convection by bodily fluids). From the skin heat is lost by convection to the air and radiation . If you are immersed in water heat can transfer by conduction and convection through the fluid. 3. How does clothing affect the rate of heat loss? 4. How does muscle density affect the rate of heat loss? (e.g. young versus elderly) b. Energy-form Conversion i. Energy can be converted from one form into another 1. KE to PE 2. Work to KE 3. PE to Work ii. On a sunny day the Sunlight warms the Earth by radiation, the warmed Earth conducts heat to the air in contact with the surface, the warmed air rises by convection. At the top of rising convective plumes clouds often form. iii. Energy can also be converted from one form into internal energy (e.g. the kinetic energy of molecules in a substance.) 1. In Physical Therapy, Therapeutic Ultrasound converts sound energy (vibrational waves) into thermal energy in your muscles. Also Diathermy 4. Thermodynamics a. Heat and the 1st Law of Thermodynamics i. The first Law is an application of energy conservation ii. The change in internal energy of a substance equals the heat added minus the work done. iii. U Q W usually better written as W Q U iv. Philosophically the first law of thermodynamics says that you can never turn internal energy entirely into work. There is always a heat loss. Nothing can be 100% efficient. 1. E.g. energy stored in steam to move a locomotive b. 2nd Law of Thermodynamics i. Heat only flows from hot to cold. ii. To get heat to flow the other way (e.g. and air conditioner or refrigerator) you must due work. iii. This sounds like a no brainer. However, if molecular motion is reversible, then one could imagine heat flowing from cold to hot naturally. 5. Atmospheric Pressure a. Effect on boiling and freezing points i. Increased pressure makes a liquid boil at a higher temperature. 1. Cooking in the mountains 2. Geysers ii. Pressure has a very small effect on the freezing point of liquids 6. Articles: Read me. Fair game on exams a. Case Study: Cold Packs b. Counting Calories c. Moderating Effects of the Climatic Environment