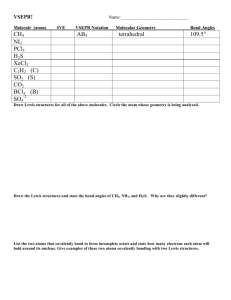
Table for Answering Lewis Structures and VSEPR Theory Practice
advertisement

Table for Answering Lewis Structures and VSEPR Theory Practice Questions Molecule and Identity Calculation of the number of Final Lewis Structure (Get another of the central atom valence electrons in the Lewis piece of paper and FOLLOW THE Structure STEPS IN YOUR NOTES!!! to get this.) Carbon tetrachloride, CCl4 Central atom is C 1 C 1 atom x 4 val electrons = 4e14 Cl 4 atoms x 7 val electrons = 28 e1Total = 32e1- Cl Cl C Cl Cl There is a total of 32 electrons in this Lewis structure. This is not a polyatomic ion so it does not need square brackets and a charge. It is not a resonance structure so only one structure needs to be shown. Predicted VSEPR formula and VSEPR shape (remember to count multiple bonds as one bond when figuring out the VSEPR formula) I know the VSEPR Formula is AX4 so the molecular shape is tetrahedral. Recall: A = central atom X = number of atoms attached to central atom E = number of lone pairs on the central atom 3-D Diagram of the VSEPR shape (include bond dipoles, bond angles and partial charges) Bond angles are all 109.5o in a tetrahedron. Bond dipoles all go from C to Cl with C slightly positive (δ+) and all Cl atoms slightly negative (δ-) because Cl has the higher electronegativity value. Is the Molecule Polar or is it Nonpolar? (include an explanation for your answer.) This molecule contains polar covalent bonds but it is a non-polar molecule because there are no lone pairs on the central atom and the molecule is symmetrical about the central atom so all of the bond dipoles will cancel one another out.