Minor or Administrative Protocol Deviation
advertisement

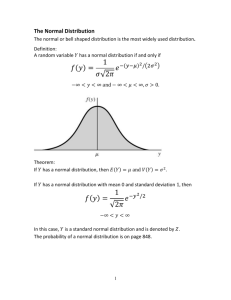
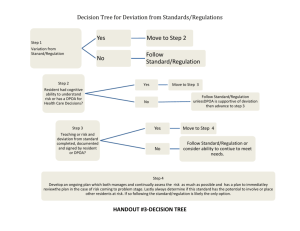
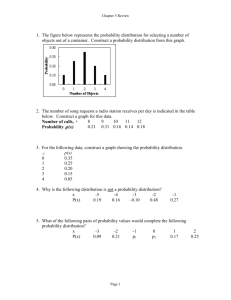
Mother Frances Hospital Tyler Institutional Review Board irb@tmfhc.org / (903) 606-2472 Minor or Administrative Protocol Deviation Log Principal Investigator: IRB# and Protocol title: A minor or administrative protocol deviation is a protocol deviation that was not due to the action or inaction of any member of the research team; or does not affect the scientific soundness of the research plan; or does not affect the rights, safety, or welfare of human subjects. Repeated deviations should be reported to the IRB because they may suggest the protocol’s design may need to be revised or a subject-specific problem. Use the following codes to describe the deviation: A) Follow up visits that occurred outside the protocol required time frame because of the subject’s actions (for example: weather, vacation, transportation, personal issues, etc.) B) Study interventions (lab tests, diagnostic tests) that occurred outside the protocol required time frame because of the subject’s actions C) Biological samples (e.g., blood, urine) obtained at times close to but not precisely at the time points specified in the protocol, especially if the samples are not obtained for safety reasons D) A single instance when biological samples (e.g., blood, urine) were not collected and the samples were not being obtained to monitor subject safety E) Failure of subject to return unused study medication F) Failure of subject to transmit device data within the protocol required time frame G) Subject takes the study drug at a different dose or frequency than instructed and safety was not affected H) Sponsor required reporting outside the protocol required time frame I) Other Add lines as needed. Submit with Continuing Review Date of Date PI Subject ID Code Brief description deviation aware Minor or Administrative Protocol Deviation Log Last revised 01/14/2015 Page 1 of 1