LAB 3b TRANSDUCTION

LAB 3a TITRATION OF A HIGH TITRE PHAGE
Viruses are noncellular entities composed of nucleic acid surrounded by a protein envelope called the capsid. They are devoid of the sophisticated enzymatic and biosynthetic machinery essential for independent activities of cellular life. This lack of metabolic machinery mandates that viruses must exist as parasites and cannot be cultivated outside of a living cell.
Viruses are differentiated from cellular life on the basis of a) size (can only be visualized with an electron microscope), b) they do not increase in size, c) the ability to pass through bacterial-tight filters, d) replication must occur in a susceptible cell, e) the viral nucleic acid subverts the synthetic machinery of the host cell to produce new viral components, f) viruses contain either RNA or DNA but never both nucleic acids.
Much of the knowledge of the mechanism of animal viral infection and replication has been based on our understanding of infection in bacteria by bacterial viruses, called the bacteriophages or phages. Bacteriophage means to eat bacteria and the name was coined because of the destruction through lysis of the infected cell.
Phage replication depends on the ability of the phage particle to infect a suitable bacterial cell. Infection consists of the following sequential events:
1. Adsorption: the tail portion of the phage particle binds to receptor sites on the host's cell wall.
2. Penetration: The spiral protein sheath retracts, and an enzyme, early muramidase, perforates the bacterial cell wall enabling the phage nucleic acid to pass through the hollow core into the host cell's cytoplasm. The empty protein shell remains attached to the cell wall and is called the protein ghost.
3. Replication: The phage genome takes over the synthetic machinery, which is then used for the production of new phage components.
4. Maturation: The period during which the new phage components are assembled and form complete, mature virulent phage particles.
5. Release: Late muramidase lyses the cell wall, liberating infectious particles that are now capable of infecting new susceptible host cells, thereby starting the cycle over again.
Virulent phage particles that infect susceptible host cells always initiate the lytic cycle as described. Other phage particle, called temperate phages, incorporate their nucleic acid into the host's chromosome. Lysis of the host cell does not occur until it is induced by exogenous physical agents such as ultraviolet, ionizing radiation or chemical mutagenic agents. Bacterial cells containing the incorporated phage nucleic acid, the prophage, are called lysogenic cells.
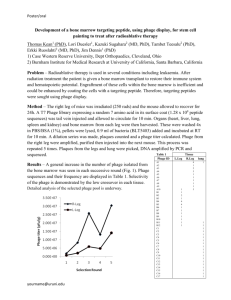
A number of methods have been devised for estimating the titre of a phage stock. The most satisfactory is the plaque count technique. In this method a few virulent virus particles and a great number of susceptible host bacteria cells are distributed over the surface of an agar plate. The procedure requires the use of a double-layered cultural technique in which the hard agar serves as a base layer and the mixture of phage and host cells in soft agar forms the upper overlay. On incubation the virus particles multiply at the expense of neighbouring bacteria. After a few hours, the bacteria will form a more or less continuous turbid film over the plate except where
multiplying virus particles have lysed the cells. At these points, clear holes (plaqueforming units (PFU)) will appear in the turbid background. These may be counted and the original titre of the suspension from which they originated may be calculated.
If the approximate titre of the virus stock is not known, several different dilutions of the virus must be plated to be sure that one of the plates will contain a reasonable number of plaques. If too few plaques are present, the sampling error is large and if too many are present, overlapping will result in unreliable counts. To determine a valid phage count, the number of plaques per plate should not exceed 300 or be less than
30.
Materials:
1 tube of phage F116
1 tube of host bacteria Pseudomonas aeruginosa PA01
5 Tryptic Soy Broth Agar Plates
5 tubes of molten TSBA overlays
8 tubes with 0.9 ml TSB
Automatic pipetter, tips, pasteur pipettes
Procedure:
1. Label all dilution tubes and media as follows: a) 5 TSBA plates: 10 -4 ,10 -5 , 10 -6 , 10 -7 , 10 -8 b) 9 tubes with 0.9 ml TSB: 10 -1 through 10 -9 DO NOT WRITE ON CAPS
2. Remove 0.1 ml phage suspension from the stock tube and deliver into the dilution tube labeled 10 -1.
Mix with the vortex.
3. With a fresh pipette tip, take 0.1 ml from dilution tube 10 -1 and deliver into dilution tube 10 -2 .
4. Continue down the dilution series in the manner above to the 10 -8 tube.
5. Take a TSBA overlay tube from the waterbath (only remove one at a time as needed) and aseptically add two drops of P. aeruginosa culture with a pasteur pipette and 0.1 ml of the 10 -5 TSB phage dilution. Rapidly mix by rotating the tube and pour the contents over the hard TSBA plate labeled 10 -5 , thereby forming a double-layered plate culture preparation. Swirl the plate gently to mix and allow to harden completely before inverting.
6. Using separate pasteur pipettes and tips, repeat step 5 for the phage dilution tubes 10 -6 through 10 -8 .
7.
Incubate all plates overnight at 37 °C.
8. Observe all plates for the presence of plaque-forming units that develop on the bacterial lawn. Count the number of PFUs on each plate in the range of 30-300.
LAB 3b TRANSDUCTION
The transfer of genetic material (DNA) between bacterial cells is a source of variability in bacterial populations and a mechanism of acquiring new properties or capabilities. One example of this, involving the transfer of bacterial chromosomal DNA from one cell to another, is mediated by bacteriophages via a process known as transduction. The process involves inadvertent packaging of partially degraded chromosomal DNA fragments by phage capsids during the assembly stage of viral infection. Because of limitations in the total amount of DNA a capsid can contain, a capsid carrying bacterial DNA may fail to package some or all of the viral DNA.
Following maturation and lysis, the bacterial DNA-containing phage particle, referred to as a transducing particle, is subsequently able to infect another susceptible bacterial cell, injecting the bacterial DNA into this cell. Being of bacterial rather than viral origin, this DNA is not able to initiate the lytic cycle but it can be incorporated into the recipient bacteriums chromosome where it is stably maintained. Expression of genes present on the newly incorporated DNA will impart new properties or abilities on the recipient cell, called a transductant, reflecting the fact that the new genetic information was delivered to the cell via transduction.
In the current exercise, bacteriophage F116L propagated on strain PAO1 of the bacterium Pseudomonas aeruginosa will be used to infect one of two strains of P. aeruginosa , PAO236 and PAO25, both of which carry defects in genes necessary for the biosynthesis of specific amino acids. As such, these strains are unable to synthesize the amino acids and their growth is, thus, dependent upon the inclusion of these amino acids in the growth medium. Such a defect in amino acid biosynthesis leading to a growth requirement for amino acids is referred to as auxotrophy and strains exhibiting auxotrophy are called auxotrophs. The phage-mediated transfer of functional (called wildtype) amino acid biosynthetic genes from strain PAO1 to strains PAO236 or PAO25 will, however, restore the ability of these strains to make the amino acids and thus, render them able to grow without the need for amino acid supplementation.
Materials: (per pair)
1 tube of phage lysate F116L grown in wild type Pseudomonas aeruginosa PAO1
1 tube an overnight culture of recipient bacteria centrifuged and resuspended in transduction buffer. Either Pseudomonas aeruginosa PAO236 (requiring isoleucine, lysine, valine, histidine, methionine, tryptophan, and proline) OR Pseudomonas aeruginosa PAO25 (requiring arginine and leucine).
7 plates of minimal media with glucose and the appropriate amino acids and bases.
Group 1
Host:
Media:
PAO236
Minimal media with isoleucine (I), valine (V), histidine (H), lysine
(K), methionine (M), tryptophan (W)
Group 2
Host:
Media:
PAO236
Minimal medial with Histidine (H), lysine (K), methionine (M), tryptophan (W), and proline (P)
Group 3
Host:
Media:
PAO236
Minimal medial with isoleucine (I), valine (V), histidine (H), lysine
(K), methionine (M), and proline (P)
Group 4
Host:
Media:
Group 5
Host:
Media:
Procedure:
PAO25
Minimal media with Arginine (R)
PAO25
Minimal media with Leucine (L)
1. The diluted phage F116L is the same dilution series used to titrate the phage in lab 3a.
2. Spread 0.1 ml of recipient bacteria on 6 of the appropriate minimal media plate.
One plate will be the recipient cell control (i.e., no phage will be plated on it).
3. Overspread the first plate with 0.1 ml of the stock tube of F116L (dilution 10 0 ).
4. Overspread the second plate with 0.1 ml of the 10 -1 dilution of phage F116L.
5. Repeat step 4 for the 10 -2 to 10 -4 dilutions.
6. Take the last plate that has had no recipient bacteria spread on it and spread
0.1 ml of undiluted phage stock on it (phage control).
7. Incubate the plates 48 hours at 37°C. Count the plate that has a suitable number of colonies (20 to 300)
Student #: _______________________________
Questions (Hand in)
1. When a recipient cell acquires a piece of naked DNA from the environment, it is called a. conjugation. b. transformation. c. transduction. d. transfection.
2. Define the terms Prophage and Lysogen.
3. Calculate the number of phage particles in the stock culture in lab 3a. (Assume a single phage particle is responsible for initiating plaque formation.)
4. True or False . Marine viruses are more abundant than marine bacteria.
5. True or False.
Bacteriophage randomly attach to the bacterial cell surface.
6. What is an auxotroph? Give an example.
7. Bacteriophage F116L is grown on P aeruginosa PAO1 and used to infect a P. aeruginosa strain PAO221 (requires arginine, lysine and histidine for growth.) the
phage population will contain transducing phage particles that contain genes for; a) histidine only b) arginine only c) lysine only d) histidine, arginine and lysine e) every gene of PAO1
8. Determine the efficiency of transduction of the amino acid biosynthetic genes in lab
3b by calculating the number of transductants yielded per number of phage particles used in the initial infection.
9. Bacteriophage F116L propagated on P. aeruginosa PAO1 is used to infect P. aeruginosa strain PAO221 (requires arginine, lysine and histidine for growth.)
Infected cells are subsequently plated on minimal medium containing arginine and histidine. The transduction of what amino acid biosynthetic gene is being measured in the experiment described? Explain
10. Compare and contrast temperate and virulent bacteriophage.