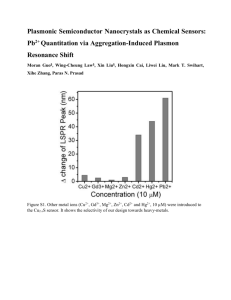
Handheld high-throughput plasmonic biosensor using
advertisement

Supplementary Information for Handheld High-Throughput Plasmonic Biosensor using Computational On-Chip Imaging 1 Department of Electrical and Computer Engineering, Boston University, MA 02215, USA 2 Departments of Electrical Engineering and Bioengineering, University of California, Los Angeles (UCLA), CA 90095, USA 3 Division of Chemistry and Chemical Engineering, California Institute of Technology, Pasadena, CA, 91125 4 Pontificia Universidad Catolica del Peru, Departamento de Ciencias-Quimica, Avenida Universitaria 1801, Lima 32, Peru 5 Bioengineering Department, Ecole Polytechnique Federale de Lausanne (EPFL), Lausanne CH-1015 Switzerland a Authors contributed equally to this work. Correspondence: Professor A. Ozcan, Departments of Electrical Engineering and Bioengineering, UCLA, CA 90095, USA, and Professor H. Altug, Bioengineering Department, Ecole Polytechnique Federale de Lausanne (EPFL), Lausanne CH-1015 Switzerland. E-mails: Professor A. Ozcan, ozcan@ucla.edu and Professor H. Altug, hatice.altug@epfl.ch 1 FDTD calculations for near/far-field response of the nanohole arrays We analyze far- and near-field responses of the periodic nanohole arrays through 3dimensional finite-difference time-domain (3D-FDTD) simulations. In simulations, the dielectric constant of silicon, titanium and gold is taken from Reference [1]. For the unit cell consisting of a nanohole, periodic boundary condition is used along the x- and y- directions and perfectly matched layer boundary condition is used along the direction of the illumination source, z. In the simulations, the mesh size is chosen to be 2 nm along the x-, y- and z-directions. Protein absorption and multiplexing experiments Initially, the plasmonic chip is cleaned by Nano-Strip™ from Cyantek Corporation (composed of 90% Sulfuric Acid, 5% Peroxymonosulfuric Acid, <1% Hydrogen Peroxide and 5% Water) and rinsed with DI-water to dissipate any organic contamination on the surface. Then, a solution of protein A/G in Gibco® phosphate buffered saline (PBS) 1X sterile solution-pH 7.4 is spotted on the specific membranes on the plasmonic chip by using BioForce Nano eNabler and 30 µm AFM tips yielding round droplets, each with a diameter of 30–35 µm and incubated for 1 hour. Protein A/G is a recombinant fusion protein which constitutes of binding domains of both protein A and protein G. Protein A/G sticks on the gold surface by physisorption. We then wash the chip 3 times with PBS for 3 minutes each time and 1 time with DI-water to remove the unbound protein. After that, the chip is immersed in a 0.5% V/V bovine serum albumin (BSA) solution in PBS for one hour to passivate the chip surface outside the A/G patterned areas followed by a similar washing process already described. Finally, protein concentrations of IgG are spotted on the chip surface and incubated for 1 hour and a final washing process is performed afterwards. Protein IgG only sticks on the protein A/G due to the high affinity of protein A/G to the Fc regions of protein IgG. 2 Surface Plasmon grating orders For a periodic nanohole array with a period P, the momentum mismatch between the incident ⃗ 𝑆𝑃𝑃 = photons and the surface plasmon polaritons is overcome when the Bragg condition, 𝑘 ⃗ 𝑖𝑛𝑐,|| ± 𝑖𝐺𝑥 ± 𝑗𝐺𝑦 is satisfied.2 Here, 𝑘 ⃗ 𝑆𝑃𝑃 is the surface plasmon wavevector, 𝑘 ⃗ 𝑖𝑛𝑐,|| is the 𝑘 wavevector of the incident photons parallel to the metal surface and 𝐺𝑥 and 𝐺𝑦 are the lattice vectors of the square lattice (|𝐺𝑥 | = |𝐺𝑦 | = 2𝜋/𝑃) with the grating orders (𝑖, 𝑗). The dispersion relation of SPPs is given by 𝑘𝑆𝑃𝑃 = (𝜔/𝑐)√𝜀𝑑 𝜀𝑚 /(𝜀𝑑 +𝜀𝑚 ) where 𝜀𝑑 and 𝜀𝑚 are the permittivity of the dielectric and metal layers, respectively. Therefore, we observe transmission resonances in the spectrum (Figure 2a in the main text) satisfying this condition with the following formula, 𝜆𝑚𝑎𝑥 = (𝑃√𝑖 2 + 𝑗 2 )√𝜀𝑑 𝜀𝑚 /(𝜀𝑑 +𝜀𝑚 ). As an additional note, in the main text for simplicity, we perform the near-field analysis for a linearly polarized light instead of an unpolarized one as we employ in our experiments. In the main text, Figure 2a shows the calculated distribution of the y-component of the magnetic field intensity, (|Hy|2) as it is the dominant component for an x-polarized light source. Fabrication of nanohole arrays In our lensfree imaging platform, we use plasmonic microarray pixels containing nanohole arrays with hole diameter of 200 nm and array period of 600 nm. The apertures are fabricated through a 120 nm thick gold, 5 nm interlayer of titanium and an 80 nm thick silicon nitride (SiN) layers. We fabricate the plasmonic chips via lift-off free fabrication process as illustrated in Fig. S1a. We first prepare the suspended SiN membranes using photolithography, reactive ion etching (RIE), and wet etching (using KOH), consecutively. Electron beam lithography is then performed on polymethylmethacrylate (PMMA) covered SiN surface. Later, nano-apertures 3 through thin SiN layer are achieved by RIE etching and the remaining PMMA is removed by oxygen based plasma cleaning. Finally, 5 nm-thick Ti (adhesion layer) and 120 nm-thick Au layers are deposited by a directional e-beam evaporator. Fig. S1b demonstrates that the apertures are well defined and unit cells are uniform over large areas. Fig. S1c shows the CMOS image of 100 µm x 100 µm plasmonic microarray pixels separated by 100 µm spacing and the SEM image of a single pixel containing 28,000 aperture elements. Figure S1 Fabrication of the nanohole arrays and microarray pixels. (a) Lift-off free fabrication process of the nanoholes. (b) SEM image of the fabricated apertures. (Inset) Dimension of a single nanohole. (c) Left figure: CMOS image of the diffraction patterns of 6 microarray pixels and right figure: SEM image of a single pixel. 4 Diffraction pattern calculation Far-field of an aperture is the region outside of the Fraunhofer distance which is defined as 𝑑𝑓 = 2𝐷2 ⁄𝜆 where 𝐷 is the largest dimension of the aperture and the 𝜆 is the wavelength. At the LED wavelength (𝜆 = 683 nm), for the square pixel of 100 µm x 100 µm, 𝑑𝑓 is calculated as 30 mm. Hence, for our plasmonic pixel, we are still in the near-field. The near-field of an aperture is divided into reactive and radiative near-fields. In the reactive near-field, the relationship between electric and magnetic fields is complex and hard to determine. In the radiative near-field in contrast (evanescent field contribution is negligible), electric and magnetic fields are still different from the far-field, but they are predictable. The radiative near-field (Fresnel region) is 1/2 the region far from the distance which is calculated by 𝑑𝑓𝑟 = 0.62(𝐷3⁄𝜆) . For our plasmonic pixel, we calculate 𝑑𝑓𝑟 as 0.75 mm. In our lensfree platform, we use a chip of 0.5 mm thickness and a CMOS camera which has an active area covered by a 0.5 mm thick glass. Hence, the distance between active plasmonic microarray pixel and the CMOS active area is approximately 1 mm as shown in Fig. S2a. Since this distance is in the Fresnel zone, we utilize a convolution approach for calculating the Fresnel diffraction patterns at z = 1 mm as shown below.3 𝐼𝑛𝑣𝑒𝑟𝑠𝑒 𝐹𝑜𝑢𝑟𝑖𝑒𝑟 𝐼𝑛𝑡𝑒𝑛𝑠𝑖𝑡𝑦 𝐹𝑟𝑒𝑠𝑛𝑒𝑙 ⟶[ ]⟶[ ] ⟶ [ 𝐹𝑜𝑢𝑟𝑖𝑒𝑟 ] ⟶ 𝑇𝑟𝑎𝑛𝑠𝑓𝑜𝑟𝑚 𝑎𝑡 𝑧 = 0 𝐾𝑒𝑟𝑛𝑒𝑙 𝑇𝑟𝑎𝑛𝑠𝑓𝑜𝑟𝑚 𝐼𝑛𝑡𝑒𝑛𝑠𝑖𝑡𝑦 𝑀𝑒𝑎𝑠𝑢𝑟𝑒𝑑 𝑏𝑦 𝐶𝑀𝑂𝑆 𝑎𝑡 𝑧 = 1 𝑚𝑚 In the main text (Figure 2d), we apply the convolution approach for a plasmonic pixel containing approximately 28,000 nanoholes (dimension of 100 µm x 100 µm) as shown with an SEM image in Fig. S2b-left. Fig. S2b-right shows the near-field intensity distribution (at 𝑧 = 0) calculated at the LED wavelength for a 5 x 5 nanohole array. 5 Figure S2 Diffraction pattern calculation. (a) Schematic view of the plasmonic chip on top of a CMOS camera. (b) Left figure: SEM image of a 100 µm x 100 µm microarray pixel containing nanohole arrays and right figure: near-field intensity distribution calculated at the LED dominant wavelength. Polarization Independent Nano-Apertures As shown in Fig. S3a, for an unpolarized light, electric field is in random directions. In contrast, for a polarized light, electric field oscillates in the plane of polarization at all times as illustrated in Fig. S3b. Using a polarizer, blocking waves of polarizations, we pass light of a specific polarization. According to Malus law, the light that passes through the polarizer has ideally an intensity half (practically even smaller) of the unpolarized light. As shown by the nearfield distributions (Fig. S3a- and S3b-bottom), the intensity of the local fields supported by the aperture system under unpolarized light is two times larger than the one supported by the system under an x-polarized light source. Furthermore, since the circular shape apertures are symmetric 6 in all directions, the underlying physical phenomena such as SPs and LSPs do not vary with the polarization unlike i.e. rectangular-shaped apertures. As shown in Fig. S3b-bottom, for the xpolarized light source, the hot spots supporting highly enhanced local fields concentrate around the rims along the x-direction. As the polarization angle varies (not shown here), hot spots move with respect to the polarization direction. Consequently, in the presence of an unpolarized light, hot spots are observed at the aperture walls along all directions. This is highly advantageous for biosensing applications as the volume of the high near-field enhancement increases allowing stronger analyte-optical field overlap. Figure S3 Unpolarized vs. polarized LED source. Illustration of CMOS based imaging systems under (a) unpolarized and (b) polarized LED sources. In bottom, electric field intensity (|E|2) is calculated at the top surface of the nanohole (hole diameter = 200 nm and period of the array = 600 nm) under unpolarized and polarized sources of the same wavelength. 7 Detection limit of the spectrometer based biosensing system Fig. S4a shows the spectral response of a plasmonic pixel and Fig. S4b shows the spectral location of the SP(-1,0) mode measured at different times. Here, we use a Nikon Eclipse-Ti microscope coupled to a SpectraPro500i spectrometer. As indicated by the figure, for each measurement, the spectral position of the peak wavelength varies around a mean peak wavelength of 656.1267 nm with a standard deviation of 0.1992 nm. Adding and subtracting twice the standard deviation to the mean peak wavelength, the detection limit of the spectrometer based system is set to be around 0.8 nm. Figure S4 Detection limit of the spectrometer based sensing platform. (a) Spectral response of the microarray pixel measured at different times. (b) Spectral position of the SP(-1,0) mode measured at different times. Refractive index sensitivity of the plasmonic nanoholes Refractive index sensitivity (RIS) determines how the spectral position of the plasmonic mode supported by the nanohole array varies with respect to the refractive index of the surrounding medium. It is defined as the ratio of the change in the resonance wavelength of the plasmonic mode to the change in the refractive index of the medium (𝑅𝐼𝑆 = ∆𝜆⁄Δ𝑛). Fig. S5 shows the transmission resonance of the bare nanohole array (λres = 693 nm) and the nanohole array 8 embedded in DI-water (λres = 898 nm). For the nanohole array, by embedding the chip into DIwater (n = 1.33) giving a resonance shift of 205 nm, we determine RIS as large as 621 nm/RIU. Figure S5 Refractive index sensitivity. Spectral response of the nanohole array in air (black curve) and in water (red curve). Detection of nano-particle based assays In this on-chip biosensing platform, we also demonstrate the detection of nano-particles towards functionalized bead assay applications that can increase the sensitivity of the on-chip biosensing platform below µg/mL. Here, we choose 100 mg/mL solution of streptavidin coated polystyrene beads (with a particle size mean 370 nm diameter). These nano-particles are captured by a biotinylated sensing surface as illustrated in Fig. S6g. Fig. S6a and S6b show the SEM images of the bare plasmonic pixel and the one covered with the bead, respectively. Fig. S6c and S6d show the electric field intensity distribution calculated at the LED wavelength for the bare aperture and the aperture with spherical beads at the top surface of the metal layer. For the bare aperture, the plasmonic modes strongly localize around the rims of the nanoholes at the top surface and extend extensively into the air. When the beads are attached to the binding protein at the metal surface, the plasmonic mode red-shifts from the LED wavelength and the near-field enhancements strongly decrease at this specific wavelength. For a low surface density 9 of nanoparticles shown in the SEM image (Fig. S6e), it is observed a 5 nm shift from the bare response (Fig. S6f). Figure S6 Detection of nano-particles. SEM images and near-field intensity distributions calculated at the LED wavelength for (a, c) the bare sensor and (b,d) the sensor covered with the binding beads. (e) SEM image of the sensor covered with low concetration of nano-particle solution. (f) Spectral response of the bare (black curve) and the target (red curve) sensors. (g) Schematic view of the streptavidin coated beads binding to the gold. Our on-chip plasmonic platform can yield high-throughput sensing of biological assays by integrating to plasmonic chips incorporating large numbers of sensor pixels. Fig. S7a shows the schematic view of the plasmonic sensor containing 100 pixels where the target pixels absorb streptavidin coated beads defining the word “BU-UC”. Fig. S7b shows the three dimensional intensity visualization of the patterned chip image demonstrating that the target pixels patterned with “BU-UC” have much lower intensity compared to the reference ones due to the red shift in 10 the plasmonic resonance. Fig. S7c shows the diffraction patterns of the 100 clean plasmonic pixels supporting a uniform intensity distribution over a large area. As shown in Fig. S7d, the target pixels containing the nanoparticles can be easily seen in the image outlined by the dashed lines. This result demonstrates that as the number of plasmonic pixels is scalable with fabrication, a single lensfree image can monitor and quantify thousands of plasmonic structures all in parallel for ultra-high-throughput label-free microarray development applications. Figure S7 Large-scale integration of plasmonic microarrays with lensfree imaging. (a) The schematics of the plasmonic sensor spotted with streptavidin coated beads with the pattern of “BU-UC”. (b) Three dimensional visualization of “BU-UC” patterned chip image. Diffraction patterns of 100 (c) clean plasmonic microarray pixels and (d) plasmonic pixels that are selectively spotted in the form of “BU-UC”. REFERENCES 11 1. Palik ED. Handbook of Optical Constants of Solids; Academic: Orlando, 1985. 2. Ebbesen TW, Lezec HJ, Ghaemi HF, Thio T, Wolff PA. Extraordinary optical transmission through sub-wavelength hole arrays. Nature 1998; 391: 667–669. 3. Goodman JW. Introduction to Fourier Optics; McGraw-Hill: New York, 1968. 12