Photoelectric Effect Experiment
advertisement

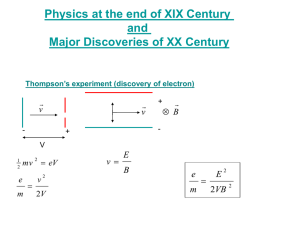
The Photoelectric Effect Aim: To test the predictions on the photoelectric effect of the classical theory of light. Classical theory analogy: Light waves are like the waves washing onto a beach with rocks (electrons) embedded in the sand (metal). Download the java applet to use in the experiment from Photoelectric Effect Java Applet PART A – INTENSITY Hypothesis 1. Classical theory predicts that as intensity increases (waves get bigger), the number of electrons being ejected would increase and hence the photocurrent will increase. Hypothesis 2. Classical theory predicts that as intensity increases (waves get bigger), the ejected electrons will have more kinetic energy (or velocity, since KE = ½mv2). Method: 1. Check the box “Show only highest energy electrons” (this is just to simplify the experiment). 2. In the ‘Options’ menu, check “Control photon number instead of intensity”. 3. Set the “Wavelength” to 150nm. 4. Set “Target” to Sodium. Notice that in these 5. Set the “Intensity” to 20%. experiments, no 6. Record the photocurrent. external voltage is 7. Observe the speed of the electrons. applied (the battery 8. Repeat steps 5-7 using intensities of 40%, 60%, 80% and 100%. voltage remains at 0.00 9. Repeat steps 5-8 using the other target metals. at all times). Results: Hypothesis 1. Photocurrent increases with intensity Photocurrent (?) Intensity (%) sodium zinc copper platinum calcium 20 40 60 80 100 Copy and paste your results table into Excel and graph it. Paste your graph below. Conclusion: ______________________________________________________________________________________________________ Hypothesis 2. Kinetic energy of photoelectrons increases with intensity For sodium metal, did the speed of the electrons increase as the intensity increased? ____________ Was this result repeated for all the other metal targets? _________ Would you consider your results for the KE experiment to be reliable? ____________ Why? _____________________________________________ Conclusion: ______________________________________________________________________________________________________ FREQUENCY – PART B Hypothesis 1. Classical theory predicts that frequency (how often waves arrive) has no effect on the number of electrons being ejected if the light (wave) is not intense enough. Hypothesis 2. Classical theory predicts that frequency (how often waves arrive) has no effect on the kinetic energy of electrons being ejected if the light (wave) is not intense enough. Method: 1. Check the box “Show only highest energy electrons” (this is just to simplify the experiment). 2. In the ‘Options’ menu, check “Control photon number instead of intensity”. 3. Set the “Intensity” to 20%. 4. Set “Target” to Sodium. 5. Set the “Wavelength” to 800nm. 6. Record the photocurrent. 7. Observe the speed of the photoelectrons (if any). 8. Repeat steps 5-7 using wavelengths of 700, 550, 400, 250 and 100nm. 9. Repeat steps 5-8 using the other target metals. Results: Hypothesis 1. Frequency has no effect on photocurrent Colour Frequency Photocurrent (?) Wavelength (Hz) (nm) sodium zinc copper platinum calcium 800 infra red 700 red 550 green 400 violet 250 UV 100 UV Copy and paste your results table into Excel and graph photocurrent vs frequency. Paste your graph below. Conclusion: ______________________________________________________________________________________________________ Hypothesis 2. Frequency has no effect on kinetic energy. For sodium metal, did the speed of the electrons increase as the frequency increased? ____________ Was this result repeated for all the other metal targets? _________ Would you consider your results for the KE experiment to be accurate? ____________ Why? _____________________________________________ Conclusion: ______________________________________________________________________________________________________ SUMMARY Summarise your results into the table below. Experiment Photocurrent Wave theory Experimental prediction observation Increase intensity Increase frequency Print out this experiment and paste it into your notes. KE Wave theory prediction Experimental observation