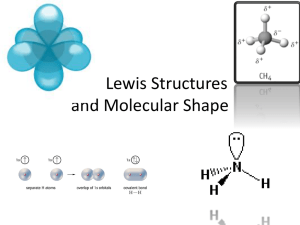
Lewis Structures & Molecular Geometry Worksheet
advertisement

Name__________________________________ Procedure: In this experiment you will draw Lewis structures for a series of molecules and determine the electronic (VSEPR) geometry, hybridization, molecular geometry and ideal bond angle(s) for the central atom or other specified atom. You will use molecular modeling kits and take pictures of each structure to help visualize the actual geometry. The octet rule must apply for C, N, O and F! For all others, it is a good guideline, but may be violated. Molecular Formula 1.NH 3 - 5+3(1)= 8e (# valence e-) Lewis Structure .. H—N—H | H ED and Molecular Geometry Polarity IMF(s) Tetrahedral/ Trigonal pyramidal Hybrid- Ideal Bond Angles ization 3 sp Picture of Ball and Stick Molecule 109.5° 2. C2H2 3. CH2Cl2 Molecular Formula Lewis Structure ED and Molecular Geometry Hybrid- Polarity ization IMF(s) Ideal Bond Angles Picture of Ball and Stick Molecule RnF4 OF6 CH3NH2 BBr3 2 Molecular Formula Lewis Structure ED and Molecular Geometry Hybridization SCl2 AtF3 SeF4 BrF5 3 Polarity IMF(s) Ideal Bond Angles Picture of Ball and Stick Molecu Molecular Formula Lewis Structure ED and Molecular Geometry Hybridization PCl5 CH2O C2H6O (CH3OCH3) C2H6O (C2H5OH) 4 Polarity IMF(s) Ideal Bond Angles Picture of Ball and Stick Molecu Molecular Formula Lewis Structure ED and Molecular Geometry Hybridization ClO3-1 BeCl2 NO3-1 BH3 5 Polarity IMF(s) Ideal Bond Angles Picture of Ball and Stick M Molecular Formula Lewis Structure ED and Molecular Geometry Hybridization H2O CO2 CH3CH2OH 6 Polarity IMF(s) Ideal Bond Angles Picture of Ball and Stick M CO32- 1. From the Lewis Structures drawn in the lab, which substance(s) showed resonance. 2. Which substance from this acitivity would have the strongest London Dispersion force. Name three reasons why iyou selected this substance your explanation 3. In the space below, draw two water molecules hydrogen bonding with each other. 4. In the space below, choose another substance from this lab capable of hydrogen bonding, and draw hydrogen bonding with water 7 5. Choose the two substances from this activity who are likely to have very low boiling points. In terms of their intermolecular forcaes, explain why you chose them. 8