Ch. 8 – Formal Charges WS
advertisement

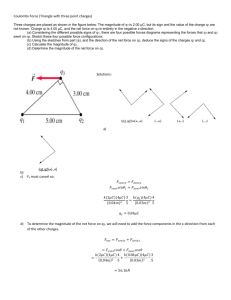
Honors Chemistry Name: _____________________________________ Date: _________________ Mods: ____________ Ch. 8: Determining Formal Charges and “Best” Lewis Structures In some instances, we can draw several different Lewis structures that all obey the octet rule and form valid Lewis structures. This does not refer to resonance; each valid structure contains a major difference from the other(s). How do we decide which of the valid structures is the “best” Lewis structure (aka: most stable)? We calculate formal charges!!!! ------------------------------------------------------------------------------------------------------------------------------------------Calculating Formal Charges in a Lewis Structure: 1. All unshared (nonbonding) electrons are assigned to the atom on which they are found. 2. For any bonds (single/double/triple) half of the bonding electrons are assigned to each atom in the bond. 3. For all neutral molecules: the sum of all formal charges must equal zero 4. For all charged ions: the sum of all formal charges must equal the ionic charge Formal Charge = [# valence e- for the atom] – [(½ Bonding e-) + (Nonbonding e-)] Example) Determine the formal charges for each atom in the molecule below. ------------------------------------------------------------------------------------------------------------------------------------------Choosing the “best” Lewis structure: 1. We generally choose the Lewis structure in which the most atoms bear formal charges of zero. 2. If there are formal charges other than zero, we generally choose the Lewis structure in which any negative charges reside on the most electronegative atom. Example) Use formal charge to determine the best Lewis structure for carbon dioxide. 1. Use formal charges to decide which Lewis structure below best represents the thiocyanate ion, SCN-. 2. Use formal charges to decide which Lewis structure below best represents the cyanate ion, CNO-. 3. Use formal charges to decide which Lewis structure below best represents sulfur dioxide, SO2. 4. For each molecule below, determine the formal charges on each atom.