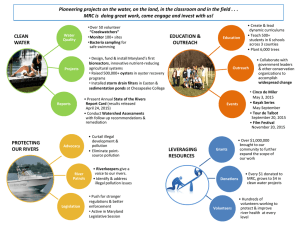
Table 2 - Springer Static Content Server
advertisement

Supplementary material Case reports Patient 1 This 40 year-old woman of Turkish ethnic background is the sister of patient 2. Family history showed no clear evidence of consanguinity within the last two generations but all family members originate from one small Turkish village. Her parents and 2 more siblings are unaffected. Her medical history includes bronchial asthma and hypertriglyceridemia. She developed a progressive asymmetric proximal weakness of the arms with elevated CK (2000 U/L, normal range <155) at age 32 years. Paresis of the upper extremities became symmetric and more severe. Clinical examination at age 40 showed lumbar hyperlordosis, a slight paresis of neck flexors (4-5/5 MRC), arm abductors (3/5 MRC), elbow flexors (2/5 MRC), external rotators of the upper arms (3/5 MRC) and right sided finger extensors II-IV (3/5 MRC). Hip flexors were 4-5/5 MRC, but muscle strength in the legs and feet was normal. (Fig. 1 A, B, C) Lung function tests at age 39 revealed a moderate obstruction but no signs of respiratory insufficiency. ECG, cardiac ultrasound and cardiac MRI were normal. Abdominal ultrasound showed a mild hepatic steatosis without liver enlargement. BMI was 33.6 kg/m2 (adipositas grade I). CK was elevated (1850-2650 U/L, normal<155) and hypertriglyceridemia was detected (triglycerides 318 mg/dL, normal range 50-200; VLDL 41 mg/dL, normal range 7-30). Serum tandem mass spectrometry (TMS) showed a normal carnitine and acyl-carnitine profile. A muscle biopsy from the right biceps brachii was taken at 32 years of age. Patient 2 This 36-year-old sister of patient 1 developed an asymmetric proximal progressive weakness of the upper extremities from age 29 years that became symmetric at age 33. No involvement of the lower extremities was present initially. At age 35 slight weakness of the right lower limb became evident. Aged 36, clinical examination revealed a symmetric weakness of the elbow flexors (2/5 MRC), arm abductors (3/5 MRC), external rotators of the upper arms (2/5 MRC), wrist extensors (4-5/5 MRC), finger extensors on the right side II-IV (3-4/5 MRC) and the left side III (3/5 MRC), right sided ischiocrural group (4-5/5) as well as the foot flexors (4-5/5 MRC) and extensors hallucis muscles (4/5 MRC) on both sides. BMI was 27.0 kg/m2 (overweight). Lung function tests, ECG and cardiac ultrasound were normal. Abdominal ultrasound showed mild hepatic enlargement and steatosis. CK was elevated (2800-4700 U/L, normal<155) and there was a mild hypertriglyceridemia (230 mg/dL, normal range 50-200). Carnitine and acyl-carnitines were normal. Muscle biopsy was taken from the deltoid muscle at 29 years of age. Patient 3 The only daughter of non-consanguineous parents of German descent developed a painless, slowly progressive proximal weakness pronounced on the upper extremities from age 33. In the course of the disease, the lower extremities remained clinically uninvolved whereas a progression of weakness was noted in the upper extremities and shoulder girdle. Examination at age 50 showed a symmetric paresis of the elbow flexors (1/5 MRC), arm abductors (2/5 MRC), pectoral muscles (4/5 MRC) and external rotators of the upper arms (1/5 MRC). The right sided finger extensors III/IV showed a mild weakness (4/5 MRC) (Fig. 1, D, E, F). Getting up from supine position was independent without support of the arms. BMI was 39.4 kg/m2 (adipositas grade II). Lung function, nocturnal pulse oximetry, ECG, long-term ECG and cardiac ultrasound were normal. Creatine kinase was elevated (930-1850 U/L, normal<155) and there was a marked hypertriglyceridemia (triglycerides 484 mg/dL, normal range 50-200; VLDL 112 mg/dL, normal range 7-30; VLDL triglycerides 402 mg/dL, normal range 30-160). Carnitines and acyl-carnitines showed a normal profile. Muscle biopsy from the left biceps brachii was performed at age 37. Patient 4 This 52 year-old British man developed a slowly progressive decline in strength of the upper and lower limbs since 28 years of age. He also has a sensory-neural hearing loss since childhood. There is no neuromuscular disease in his family, but his parents were first cousins. One brother died at 6 months of age of uncertain cardiac disease. On examination at age 35 there were symmetric pareses of shoulder abductors (4/5 MRC), pectoral muscles, elbow flexors and extensors (4/5 MRC). In the lower limbs knee extensors and hip extensors were 4/5 MRC and he showed a waddling gait. Aged 52, the myopathy gradually worsened, but he could still walk independently. Neck muscles had normal strength. He developed a more severe (3/5 MRC) weakness in proximal muscle groups of the shoulder and arm, and a milder, but definite weakness (4/5 MRC) in the finger extensors. Symmetric scapular winging was noticed. He had a Gower`s sign and hip muscles were weak (3/5 MRC). Knee flexors/extensors were relatively retained (4/5 MRC), however ankle and foot muscles were more affected (3/5 MRC) (Fig. 1 G, H, I). He could not walk on heels or tiptoes. Electrophysiology showed signs of an acute myopathy. Except for cystic changes in the pineal gland, brain MRI was normal. ECG and lung function tests were within normal range. Cardiac ultrasound showed a slightly increased left ventricular wall thickness with mild hypokinesia of infero-posterior walls and decreased ejection fraction (40-45%). ACE inhibitor treatment was administered. Jordans’ anomaly was noted in blood smear. CK was elevated (600-1250 U/L, normal<155). Liver function tests were normal. Cholesterol was at the upper limit (5.1mmol/L, normal<5), serum carnitine and acyl-carnitines were normal. Muscle biopsy was taken from the quadriceps femoris muscle at age 25. Patient 5 This 33 year-old woman originated from Northern Iraq. Since age 28 years she developed a progressive weakness and atrophy of the arms, accompanied by mild hip weakness from age 32. Examination at age 33 showed a symmetric paresis of the deltoid muscles (3/5 MRC), elbow flexors (3/5 MRC), arm extensors (4/5 MRC) and finger extensors (3/5 MRC), with a mild involvement of the hip flexors (4+/5 MRC). Electromyography showed myopathic changes in biceps brachii muscles. ECG, cardiac and abdominal ultrasounds were normal. CK was elevated (969 U/L, normal range <155), and there was no evidence of diabetes mellitus or hypertriglyceridemia. Patient 6 The patient is a 45-year-old woman of English ethnic origin from Newfoundland. Her father died at age 57 of dilated cardiomyopathy and heart failure, her mother is alive. The patient developed a slowly progressive myopathy with onset after age 20 with preserved ambulation in her forties. Severe, congestive heart failure and cardiomyopathy were diagnosed at 39 years of age. Diabetes mellitus was noted at age 40 requiring insulin therapy. Her neurological examination at age 43 revealed a myopathy of moderate severity with a prominent involvement of the upper limbs. She could not elevate her arms above her head. The weakness of the upper extremities was more proximal than distal. There was a proximal weakness on the lower extremities. She denied any muscle pain and she was able to walk short distances, but for longer distances she required a walker. Vision and hearing were normal. Electrophysiological examination detected myopathic changes. Her respiratory function was normal. She had insulin-dependent diabetes mellitus and liver was normal. BMI was 22 kg/m2 (normal weight). Serum CK was elevated (800-1200 U/L, normal<120), triglycerides and cholesterol were normal. Carnitine and acyl-carnitines were normal both in blood and skin fibroblasts. A muscle biopsy of the anterior thigh was taken at 41 years of age. Open treatment attempts Supplementation with L-carnitine (2 grams daily in patient 3), coenzyme Q10 (150 mg daily in patient 2 and 3, up to 450 mg daily in patient 1) and riboflavin (100 mg daily in patients 1 and 2) each over a minimum period over 6-months period showed no significant effect on muscle strength, triglycerides and CK levels. Patient 6 received 1 gram L-carnitine daily for 10 months without subjective improvement. Supplementary table 1 Summary of the clinical and genetic data of patients in this study with mutations in PNPLA2 Patient Age Origin Onset (years) Distribution of weakness JB HyperCKemia Hyperlipidemia Diabetes Hepatomegaly CMP 1F 39 Turkey 2F 36 3F 32 proximal>distal upper limbs + 1200-2800 TG, VLDL - - - hom c.613dupC (exon 5) Turkey 29 + 3200-4700 TG - + - hom c.613dupC (exon 5) 50 Germany 33 + 1000-1300 TG, VLDL - - - hom c.1051delC (exon 8) 4M 51 UK 18 + 600-1300 Chol - - + hom c.543delC (exon 5) 5F 32 Iraque 29 + 800-1000 - - - - hom c.695delT (exon 5) 6F 44 Newfoundland 25 proximal upper extremities >prox. lower limbs proximal upper extremities > proximal and distal lower limbs limb girdle and mild distal lower limbs proximal and distal upper extremities, proximal lower limbs limb girdle + 800-1200 - + - + hom c.584C>T (exon 5) hom c.1447G>C (exon 10) 6/6 6/6 4/6 1/6 1/6 2/6 JB: Jordans bodies; CMP: cardiomyopathy; n.d.: no data; PNPLA2 mutations Supplementary table 2 The following intronic primers were used to sequence the PNPLA2 gene PNPLA2 1fw 5´-TTGCTCCACTGACCAGGAG-3´ PNPLA2 1rv 5´-GAGTCCCGCAAACCCTTC-3´ PNPLA2 2fw 5´-GAAGCTTTCTCCGGGCG-3´ PNPLA2 2rv 5´-GACCCGGATTGGATCCTC-3´ PNPLA2 3-4fw 5´-CTCCTCCAGGGTCTGTATGC-3´ PNPLA2 3-4rv 5´-CAAGGGCCTGAAGGATGG-3´ PNPLA2 5fw 5´-TGAGGTAGCCACTGAATGGG-3´ PNPLA2 5rev 5´-TGGGCACTCAGAAGTCAAAG-3´ PNPLA2 6-7fw 5´-ATAGGTTTTGGGGTTGGGAG-3´ PNPLA2 6-7rv 5´-AGAGGGTGGTCAGCAGGTC-3´ PNPLA2 8fw 5´-AGAGGAGAGGACGGTGAGC-3´ PNPLA2 8rv 5´-CTGTCTGTGCCCTGACTTGG-3´ PNPLA2 9fw 5´-AGCAGTGCCAAGTCAGGG-3´ PNPLA2 9rv 5´-CAGCTCCACCTGCTCCG-3´ PNPLA2 10.1fw 5´-AAGAGGAAGCTGGGCAGG-3´ PNPLA2 10.1rv 5´-GCGCAGTGGTAATGGAGG-3´ PNPLA2 10.2fw 5´-TTGCTGAGCACCCCTGC-3´ PNPLA2 10.2rv 5´-AGGCTGGTCAGCCAAGGTAG-3´ Supplementary table 3 Summary of the clinical and genetic data of previously published patients with mutations in PNPLA2 Sex Age Origin Onset (years) <32 Distribution of weakness JB Hyperlipidemia TG Diabetes + HyperCKemia (U/l) + mild general hypotonia F 32 Netherlands F 29 France 29 proximal lower limb + moderate VLDL + + + PNPLA2 mutations het 584C>T (exon 5), 808delC (exon 7) hom 847delC (exon 7) F 37 Algeria 27 + 1400 n.d. - - - hom 865C>T (exon 7) F 35 Japan 35 upper limb: distal and proximal, lower limb: distal limb girdle + 650 n.d. - - - F 44 Iran 30 + 600-800 TG, VLDL + + - M 40 Italy 30 Proximal and distal, lower > upper extremities limb girdle hom c.475_478dupCTCC (exon 3) hom 695delT (exon 5) + 200-400 TG - - + hom c.541delAC (exon 5) M 35 Italy 30 limb girdle and distal lower limb + 200-400 - - - + hom c.541delAC (exon 5) M* 63 Japan 36 mild gereralized hypotonia + 750 n.d. + + + M 27 Japan 20 Distal upper and lower extremities + 750-1700 - - n.d. + hom 799-802delGCCC (exon 7) hom c.475_478dupCTCC (exon 4) F 35 Japan 33 generalized mucle weakness + 650 n.d. n.d. n.d. + M 43 Japan 41 proximal upper limbs n.d. n.d. - n.d. n.d. + F 18 U.S. 10 no paresis + 1800-3000 - n.d. - 3/12 4/12 7/12 4/12 JB: Jordans’ bodies; CMP: cardiomyopathy; n.d.: no data published; TG: triglycerides; VLDL: very low densitiy lipoprotein CMP - Hepatomegaly + - hom c.475_478dupCTCC (exon 4) hom 685C>T (exon 7) hom 1890 bp-insertion (exon 3) Reference (Fischer et al., 2007) (Fischer et al., 2007) (Fischer et al., 2007) (Akiyama et al., 2007) (Campagna et al., 2008) (Campagna et al., 2008) (Campagna et al., 2008) (Kobayashi et al., 2008) (Ohkuma et al., 2008, Ohkuma et al., 2009) (Ohkuma et al., 2009) (Hirano et al., 2008) (Akman et al., 2010)