Updated 11-5-13 Appendix D (Control)
advertisement

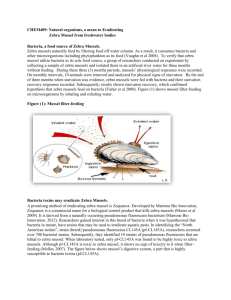
Appendix D-1. Control Options1 Thermal Shock Hot water treatment can kill dreissenid mussels. Long-term exposure to temperatures of 32°C and above are lethal to adult zebra and quagga mussels. Time to death depends upon acclimation temperature of the animal as well as rate of temperature increase. Dreissenid mussels will die in about 1 hour when placed in water of 37°C. At winter acclimation temperatures (5 to 10°C), temperatures of 33°C and above will kill zebra mussels within 13 hours. For further information, see Table 1 below.2 Veligers are more susceptible to high temperatures and will experience high mortality if the ambient water summer temperature rises above 28°C.3 Freezing Adult dreissenid mussels die when aerially exposed to freezing temperatures. In winter, populations can be controlled by dewatering and exposing zebra mussels to freezing air temperatures. Zebra mussels die in 2 days at 0°C and at minus 1. 5°C, in 5 to 7 hours at minus 3°C, and in under 2 hours at minus 10°C. Duration to mortality is less for single mussels than for clustered mussels.4 Oxygen Starvation Stripping oxygen from ambient water, primarily in closed environment such as intake pipe, can be accomplished by use of oxygen scavenging chemical such as sodium-meta-bisulfite and hydrogen sulfide gas.5 Time to death depends on the level of remaining oxygen, ambient temperature and the fitness of the mussels treated. Time to 100% mortality of zebra mussels at 25°C is 12 days at oxygen saturation of 5%.6 At 15°C, the time to 100% mortality increases to 70 day at the same oxygen level. Oxygen stripping can also be achieved by cycling ambient water through oxygen-stripping pumps. The developer of the technology, Wilson J. Browning of Amark Corp., Norfolk County, VA, claims the equipment can cycle 200 million gallons of water and remove oxygen. Addition of CO2 has also been proposed as a tool to remove oxygen from water in an industrial setting. Information on the impact of low oxygen levels on survival and settlement of dreissenid veligers is sparse. From studies on veligers of other species, oxygen levels well above hypoxia levels impact both the development and settlement of veligers. (Note: Portions of the material in this section were taken from California’s Zebra Mussel Early Detection and Public Outreach Program Final Report (Messer, C. and T. Veldhuizen, 2005 and Mackie and Claudi 2010). Additional information including the data in Tables 2, 3, and 4 was compiled by Bruce Sutherland, consultant to the Pacific States Marine Fisheries Commission. ) 2 McMahon, R. F., T. A. Ussery, and M. Clarke. 1993. Use of emersion as a zebra mussel control method. Technical Report No. EL-93-1, US Army Corps of Engineers, Waterways Experiment Station, Vicksburg. 3 Mackie, G. L., and R. Claudi. 2010. Monitoring and control of macrofouling mollusks in fresh water systems. CRC Press, Taylor Francis Group, Boca Raton. 4 Payne, B.S. (1992a). Freeze survival of aerially exposed zebra mussels. Technical Report ZMR-2-09, U.S. Army Corps of Engineers Waterway Experiment Station, Vicksburg, MS. 5 USACE-ZMIS at http://www. wes. army. mil/el/zebra/zmis/idxlist. htm. 6 Johnson P. D., and R. F. McMahon. 1998. Effects of temperature and chronic hypoxia on survivorship of the zebra mussel (Dreissena polymorpha) and Asian clam (Corbicula fluminea). Canadian Journal of Fisheries and Aquatic Sciences. 55:1564–1572. 1 Oxygen Starvation - Benthic Mats Researchers from the Rensselaer Polytechnic Institute in New York are investigating the use of benthic mats that would cover the sediment and adult mussels, thus depriving them of oxygen. Preliminary laboratory bioassays carried out in aquaria demonstrated that benthic mat covering of zebra mussels for 2 weeks resulted in mortality rates of 14. 9–100%, while mortality rates were 2. 2% or lower for control aquaria without mats. In laboratory studies in which mussels were covered for 4 weeks, mortality rates of 20–100% occurred, and did not vary significantly with duration of covering or size class. Measurements of several water chemistry parameters beneath mats, including dissolved oxygen, ammonia, calcium and magnesium and pH, indicated that dissolved oxygen concentration was the only parameter to exhibit both significant change and a consistent trend over the course of the study, declining from nearly 100% saturation to a mean of 16. 5% saturation, and remaining at this level for the duration of the experiment (Sandra Nierzwicki-Bauer, personal communication, 2008). In field studies carried out in New York’s Saratoga Lake, divers created treatment and control zebra mussel colonies at 2m depths on a rocky substrate by placing rocks with attached mussels on fiberglass screens atop gravel beds. During a field trial where two treatment colonies, composed of about 30,000 mussels each, were covered with 4m2 mats, mortality rates exceeded 99% after nine weeks of covering. As observed in the laboratory tests, dissolved oxygen concentrations declined significantly under the mats, correlating strongly with increased mortality (Sandra Nierzwicki-Bauer, personal communication, 2008). Desiccation Desiccation is a viable option for eradicating adult mussels from areas that can be dewatered for several days. Alternatively, desiccation can also act as a population control method in areas that cannot be completely dewatered. For example, reservoir levels can be lowered to expose dreissenid mussels inhabiting shallow water. Depth of colonization is dependent upon water temperature, oxygen content, and food availability in a particular body of water. Higher population densities tend to be found above the thermocline, but quagga mussels, in particular, have been found exist at depths greater than 100m. Temperature is positively related, and humidity is negatively related, to adult mussel survival. As humidity increases and temperature decreases, survival increases (Table 1). Aerial exposure of zebra mussels to temperatures exceeding 25°C, will result in 100% mortality in 2. 1 days. Temperatures over 32°C are lethal within 5 hours. Instantaneous mortality occurs at 36°C. At temperatures below 30°C, time to mortality is dependent upon relative humidity. D1–1 Table 1. Number of days to 100% mortality of adult zebra mussels aerially exposed to different levels of relative humidity and air temperature.7 Days to 100 % Mortality at Air Temperature, °C Relative Humidity, % 5 15 25 95 26. 6 11. 7 5. 2 50 16. 9 7. 5 3. 3 5 10. 8 4. 8 2. 1 Manual Removal When found in relatively small numbers, manual removal may be an effective way to reduce dreissenid populations and potentially even eradicate them if reproduction has not yet occurred. Manual removal can take place via hand extraction or via mechanical scraping and suction, typically using divers. In Lake George, New York an effort involving hand harvesting by divers appears to have significantly reduced an introduced population. Divers removed 267 mussels in 1999, followed by a peak of nearly 20,000 in 2000. Since then, ongoing removal efforts have yielded fewer than 2,000 mussels per year (Sandra Nierzwicki-Bauer, personal communication, 2008). The apparent eradication of the nonnative sabellid polychaete worm, Terebrasabella heterouncinata, in California, provides analogous evidence to the role of hand removal as a control technique. After this marine pest was found at an intertidal site outside of an infected abalone culture facility, over 1. 6 million native black turban snails (Tegula funebralis)—the preferred native host—were extracted by hand, along with other infested material. This effort reduced the transmission of the pest species to the point that it no longer was detectable in follow-up surveys.8 Predation The relatively soft shells of dreissenid mussels and their exposure (on substrates as opposed to buried in sediment) make them vulnerable to predation. Possible predators of adult mussels include some species of carp, catfish, bullhead, sucker, sunfish, sturgeon, crayfish, and muskrats. A possible predator of veligers is the American shad. However, there is no evidence of predation control in the Great Lakes, Ohio River, and Poland. There is some evidence of population reduction in the Hudson River. Despite the lack of clear evidence of population control through predation, it is recommended that harvest of predatory species in mussel-infested waterbodies be minimized. Acoustic Deterrents Several acoustic deterrents have not been proven effective in commercial installations. If developed, acoustic deterrents could be environmentally friendly and have a low likelihood of harming nonMcMahon, R. F., T. A. Ussery, and M. Clarke. 1993. Use of emersion as a zebra mussel control method. Technical Report No. EL-93-1, US Army Corps of Engineers, Waterways Experiment Station, Vicksburg. 8 Culver, C. S., and A. M. Kuris. 2000. The first apparent eradication of a locally established introduced marine pest. Biological Invasions. 2:245–253. 7 D1–2 targeted organisms. Cavitation is a form of acoustic energy that initiates the formation and collapse of microbubbles. At frequencies between 10 and 380 kHz, this type of energy has demonstrated mortalities of veliger, juvenile, and adult zebra mussels. Exposure times are in the range of seconds for veligers, minutes for juveniles, and hours for adults.9 Sound treatment using low frequency energy has prevented the settlement of zebra mussels and could be a valid option for reducing the spread of the organisms. Sound waves between 20Hz–20 kHz have caused veligers to detach and sink. Ultrasound waves between 39–41kHz have fragmented veligers in a few seconds and killed adults in 19 to 24 hours.10 Vibration is the use of solid-borne acoustic energy in mechanical structures. This treatment will only work on structures that can be subjected to vibration and not suffer structural deterioration. Vibrational energy is effective in killing zebra mussel veligers and juveniles at just below 200 Hz and between 10 and 100 kHz.11 Plasma pulse technology (Sparktec Environmental, Inc.) has shown some effectiveness in controlling zebra mussels in intake pipes. The system works by releasing stored energy and creating a spark between two electrodes. This causes an intensive shockwave, a steam bubble, and ultraviolet light.12 Deployment of the Sparker technology in a pipeline on Lake Champlain prevented settlement of veligers in an area up to 100 feet from the Sparker.13 Attempts to re-test this technology by Reclamation have not been successful to-date. Electrical Deterrents Electrical current could be used both to disable veligers in a stream of water and to create a static field on a surface to be protected from settlers. Number of studies exists documenting some success with both applications. To-date no commercial installation exists using this technology. Filtration - Media Filters This technology is capable of removing all stages of all dreissenid mussels and protecting all downstream systems and components. Sand or media filters are frequently used for protection of individual components in power plants. For protection of moderately sized intakes, infiltration galleries have been constructed in some locations. 9 Kowaleski, J. J., and P. H. Patrick. 1993. Acoustics as a possible mitigation strategy against zebra mussel settling. In Proceedings of the Third International Zebra Mussel Conference, Toronto, Canada, February, 1993. 10 Sonalysts, and Aquatic Sciences. 1991. Zebra mussel deterrence using acoustic energy. Research Report 90-38. Empire State Electric Energy Research Corporation. 11 Kowaleski, J. J., P. H. Patrick, and A. E. Christie. 1993. Effects of acoustic energy on the zebra mussel (Dreissena polymorpha). In Zebra Mussels: Biology, impact and control, T. Nalepa and D. W. Schloesser, eds.pp.167–174. Boca Raton: CRC Press. 12 Mackie, G. L., P. Lowery, and C. Cooper. 2000. Plasma pulse technology to control zebra mussel biofouling. ERDC TN-ZMR-2-23 US Army Corps of Engineers, Waterways Experiment Station, Vicksburg. 13 Schaefer, R., and R. Claudi. 2004. Development of an efficient low-cost Sparker technology for controlling zebra mussels. Presentation at the Thirteenth International Conference on Aquatic Invasive Species, Ennis, Ireland, September 20–24, 2004. D1–3 Filtration - Mechanical Self Cleaning Filters This technology is capable of removing all stages of dreissenids if an appropriate screen size and configuration is used. Most conventional industrial strainers have strainer screen openings that will prevent some translocating mussel adults and most shell debris from fouling the raw water systems. None, however, will protect against the introduction of larval stage organisms. In most instances, it is not possible to retrofit existing strainers with finer screens and hope for successful mitigation. The performance of such modified strainer or filter tends to deteriorate, excessive clogging of the screen may result in stretching and tearing of the material, the backwash system may prove to be inadequate and the pressure drop caused by the strainer may be unacceptable. Different types of filters, designed primarily for the removal of small particles, have been tested for dreissenid veliger control by a number of different organizations. Wedge wire slot filters/strainers have difficulty in excluding veliger stages of dreissenids. This is likely due to the fact that wedge wire type screen filters are being designed to remove inorganic matter, such as quartz or metal shavings, but they are not designed to stop organic matter from passing through the screen. Organic particles, due to their flexible nature, tend to “sneak through” the wedges of the screen. Hydrocyclone or centrifugal separators filters were initially thought to be a mitigation option for facilities that already employ this technology for silt removal. A 1992 study14 tested two different centrifugal separators and concluded that the removal rate of larval mussel stages was no greater than 50%. A later study15 as part of a ballast water project confirmed demonstrated that a 100µm hydrocyclone filter was 30% less effective in removing both biotic and abiotic materials than either the disc or screen-type automatic backwash filter. The lack of success is probably due to the close to neutral buoyancy of dreissenid veligers. Recently, hydrocyclone filters have been used successfully for removal of New Zealand mud snails in fish hatchery applications. Excellent results were obtained by Ontario Power Generation using a continuous backwash, pleated screen filter16 and by the New York Power Authority using a modified clean-in-place bag filter17 in eliminating dreissenid veligers from incoming water. Many filters are very good at removing all or most particles from the water stream, but most filters are not able to process large volumes of water efficiently. Filters that use stainless steel, square weave mesh and automatic backwash seem to have the best balance between particle removal efficiency and volume of water filtered. A number of manufacturers produce such filters, but they must be carefully evaluated to ensure efficacy. A rugged design with a stainless steel mesh no greater than 80 microns in either direction and an efficient self-cleaning system should be capable of preventing the majority of ready-to-settle veligers from entering the downstream system. Acres International. 1992. The study of centrifugal separators for zebra mussel control. Report P9895.29.01. Parsons, M. G., and R. W. Harkins. 2002. Full scale particle removal performance of three types of mechanical separation devices for the primary treatment of ballast water. Marine Technology 39:211–222. 16 Koopmans, R., and R. W. Hughes. 1993. Mechanical filtration as a control measure for zebra mussels. In Proceedings of the Third International Zebra Mussel Conference, Toronto, Canada, February, 1993. 17 Kahabka, J., and A. Talgo. 1993. Filtration as a control method for zebra mussels: A small hydroelectric plant application. In Proceedings of the Third International Zebra Mussel Conference, Toronto, Canada, February, 1993. 14 15 D1–4 40 Mic ron F ilter T es ts 400 Numer of Velig ers 350 300 250 200 B efore 150 A fter 100 50 0 < 100 100-200 200-400 > 400 Velig er S iz e (µm) 80 Mic ron F ilter T es ts 600 Number of Velig ers 500 400 300 B efore A fter 200 100 0 < 100 100-200 200-400 > 400 Velig er S iz e (µm) Results from filter evaluation tests carried out on the Lower Colorado River18 Filtration systems are not appropriate for water streams with continuously high sediment load. Under such conditions, the backwash system may not be able to remove the sediment cake that builds up on the screen. Very efficient backwash systems are capable of coping with higher sediment loads. Claudi, R., T. H. Prescott, and A.C. Taraborelli. 2008. Assessment of the ballast safe filter performance in removing quagga mussel veligers from the raw water of Lake Havasu. Report for Sigma Design Company LLC, Springfield, N.J. and Maritime Solutions, Inc. 18 D1–5 UV Radiation The term ultraviolet is applied to that portion of the electromagnetic spectrum between visible light and x-rays, typically between 190 and 400 nm. This region is commonly subdivided into UVA, UVB and UVC, where UVA corresponds to the longer wavelength (lower energy regime), through to UVC, which corresponds to the shorter wavelength, higher energy end of the UV spectrum. UV radiation is an effective method for preventing downstream settlement of dreissenid veligers. Based on work by numerous authors in the early 1990s, the UVB and UVC portions of the spectrum were found to be most effective for dreissenid veliger control. The basis for the control is thought to be the impact of the UV light on the essential functions of the veliger, thereby inactivating the organism and preventing attachment. The determination of the most effective wavelength and the radiation dose to achieve either immediate or latent mortality of the veligers or juveniles has been the subject of numerous studies. Medium pressure mercury lamps have been shown as capable of delivering an effective dose to eliminate downstream veliger settlement. A full-sized pilot UV system was installed at a power plant on Lake Huron in 1999. Twenty medium pressure lamps were arranged in four frames each containing five lamps (Figure 1). The total volume treated was 760 L/s (12,000 US gpm). The system was sized to deliver a radiation dose of 70–100 mWatt-scm2 to all particles passing through. This system was the only means of protection on this cooling system. During the operation of the UV system, lamps had to be serviced, the system experienced numerous upsets and occasionally it was even taken out of service inadvertently. Despite these issues, there was an 85% reduction in settlement in the system, when compared to control.19 Figure 1. UV lamp rack used in an open channel system for dreissenid veliger control. The UV systems currently being tested for control of various species in ballast water are rugged and dependable. They use UV lights installed inside a pipe, rather than the open array shown above, and they have been engineered to deliver a maximum dose in the least amount of time. Pickles, S. B. 2000. Use of ultraviolet radiation for zebra mussel control at Ontario Power Generation. Presentation at the Tenth International Aquatic Nuisance Species Conference, Toronto, Canada, February, 2000. 19 D1–6 The effectiveness of a UV system is dependent on the characteristics of the raw water being treated. Factors, such as water transmittance, presence and size of suspended solids, iron, hardness and temperature all affect the efficacy of the UV system. Treatment systems must be designed for the worst case scenarios. This means designing for peak flows, end of lamp life intensity, minimum transmittance and maximum suspended solids at the installation location. The aim of the system is to achieve 100% immediate or latent mortality in all ready to settle veligers which pass through. If an adequate dose is not delivered at this point, downstream settlement will occur as UV-based systems have no residual toxicity that could impact areas outside the influence of the lamps. Three separate studies on the Lower Colorado River have shown the extreme effectiveness of medium pressure lamps in preventing quagga mussel settlement downstream.20 Chemical Treatment There are 2 main categories of chemicals used to treat zebra mussel infestations: oxidizing biocides, and non-oxidizing biocides. The most susceptible life stages to chemical treatment are all stages of veligers and post spawn adults. Application rates and exposure time required data for these chemicals have been gathered from laboratory studies, power plant, and water treatment plant applications. Oxidizing Biocides Chlorine, bromine, hydrogen peroxide, ozone, and potassium permanganate are examples of oxidants that are used to control dreissenid mussels. The chemicals have been used by the water treatment industry for disinfection since the late 1800s. Because these chemicals have been in use for so long, their effect on the environment is well understood and documented.21 In mussels, oxidizing chemicals are thought to work by oxidizing the gill lamellae as well as other organs, eventually causing death. Dreissenid mussels can recognize oxidizing chemicals as toxins and close their shells when oxidizing chemicals are present. Periodically, they reopen their shells to “test” the water. Depending upon water temperature, respiration rate, and stored nutrient reserves, dreissenid mussels can remain closed for many days before resuming respiration. Once this occurs, mortality is swift. Therefore, required exposure time for oxidizing biocides can range from 1–3 weeks. Oxidizing chemical can also be used to prevent settlement by either continuous, semi-continuous, or period application. Chlorination in various forms, such as sodium hypochlorite, chlorine gas, chlorine dioxide, and chloramines, is the most common method of dreissenid mussel control. The use of chlorine and its various forms is usually limited to non-open water situations because of its high toxicity to other forms of aquatic life. Treated waters must either be dechlorinated or held until the residual chlorine has dissipated before discharge. An example of chlorine use that may be applicable to a small isolated population of dreissenid mussels is the practice of using tarps to seal off an area and then injecting chlorine into the enclosed area. The Washington Department of Fish and Wildlife used this method 20 Claudi, R., A. Graves, A. C. Taraborelli, R. J. Prescott, and S.E. Mastitsky. 2012a. Impact of pH on survival and settlement of dreissenid mussels. 7(1). doi:10.3391/ai.2011.ICAIS. http://www.rntconsulting.net/Publications/Articles.aspx 21 Claudi, R. and G. L. Mackie. 1994. Practical manual for zebra mussel monitoring and control. Lewis Publ., Boca Raton, FL. ISBN 0-87371-985-9. 256 pp. D1–7 in October of 2004 to successfully eradicate a small population of nonindigenous tunicates in Puget Sound near the City of Edmonds (Pers. Comm., Pam Meacham, WDFW, February 2007). This method was also used in Huntington Harbor, California, to eradicate a marine alga, Caulerpa taxifolia. Patches of Caulerpa were treated by covering them with black PVC tarp and injecting liquid chlorine under the tarp. The edges of the tarp were sealed to the bottom with sandbags. All of the organisms under the tarps were killed by the treatment, and the tarp method avoided impacts to surrounding areas.22 Hydrogen peroxide. Although toxic to dreissenid mussels, hydrogen peroxide is rarely used because of the high dosage rates and high cost. Ozone is effective at the same doses as other oxidants. 0. 5 mg/l has been shown to cause 100% mortality of veligers in 5 hours and adults in 7–12 days. Application of 0. 3ppm of ozone during the mussel breeding season had prevented mussel settlement and eliminated all adults already in place in a cooling water system of a coal fired power plant. Ozone dissipates quickly and is considered less harsh on the environment. High capital cost of the equipment has been a barrier to wider use. Potassium permanganate is also an effective chemical for mussel control and has been used by a number of water treatment plants in North America. At higher doses, potassium permanganate stains water pink. Fear of pink water has been a barrier to the adoption of this control strategy. In 1992–93, the Central Lake County Joint Action Water Agency fed an average dosage of 0. 11 mg/l potassium permanganate (maximum dosage of 0. 22 mg/l) into raw water from May to October to successfully control zebra mussel settlement. The City of Racine, Wisconsin used an average dose of 0. 2–0. 25 mg/l potassium permanganate to control zebra mussels in their water intake facilities. Upstream of the application point, extensive zebra mussel colonization was reported, but the pipes and wet well downstream of the application point were free of attached zebra mussels. Sodium permanganate liquid is commonly used in drinking water treatment to control tastes and odors, remove color, and oxidize dissolved iron and manganese. Sodium permanganate has only been used effectively for zebra mussel control at the WTP in Findlay, Ohio. The City of Findlay increased the sodium permanganate dosage from 0. 3 mg/l to 1. 25 mg/l during the course of a year. This was effective in eliminating zebra mussel colonization downstream of the application. Water treatment plants in Neenah, Wisconsin, and Keokuk, Mississippi, have also used sodium permanganate for mussel control, but their effectiveness is not well documented. Non-oxidizing biocides Non-oxidizing biocides are generally not recognized as noxious substance by the dreissenid mussels. Mussels continue to filter water, exposing themselves to these chemicals. Elimination of adult mussels with non-oxidizing chemicals can be accomplished in hours compared to days for oxidizing chemicals. 22 The most commonly used non-oxidizing compounds are proprietary molluscicides (e.g., Clam-Trol, Bulab, and Bayluscide). These are very effective for controlling dreissenid mussels, but they are also toxic to many fish and other aquatic species at the required concentrations. Most proprietary molluscicides must be detoxified prior to release to the www. sccat. net/eradication. php D1–8 environment. These compounds are usually deactivated by releasing slurry of bentonite clay into the water. The cationic or surfactant active ingredients bind onto the clay, becoming inactive. The clay settles out of the water column and becomes part of the bed sediments. The compounds are eventually degraded by microbial action. As is the case with most chemical treatments, the proprietary molluscicides are less effective at lower water temperatures, so treatment is recommended during warmer months. The chemicals are usually administered with equipment supplied by the vendors. An example of the successful use of non-oxidizing chemicals to control the Asian clam in the southeastern United States can be found in a paper entitled “Strategies for application of non-oxidizing biocides.”23 Additional information on most of these chemicals, such as formula, manufacturer, and application method, is available at http://www. wes. army. mil/el/zebra/zmis/idxlist. htm. pH adjustment—under 7 or over 9. 5—There continues to be some uncertainty as to what pH level can be tolerated by larval stages of dreissenids and at what pH adults will survive for prolonged periods of time. Successful development of zebra mussel veligers requires a pH range between 7. 4 and 9. 4, with optimal conditions at 8. 5.24 In a series of experiments done on the Lower Colorado River and at San Justo Reservoir in California, the effects of both low and high pH was tested on both the settlement prevention and mortality of adult zebra and quagga mussels. Water with continuously adjusted pH below 6. 9 prevented the settlement of quagga mussels, and long term exposure to this pH caused significant mortality in adults. By comparison, settlement was not completely prevented in the zebra mussel population, and the mortality of adults was much low. At pH levels over 9. 6, new settlement by zebra mussels was almost entirely absent. The observed mortality of adult zebra mussels was low, but did tend to increase with increasing pH. In addition, short-term experiments testing the response of zebra mussels to both low and high pH extremes were carried out at San Justo Reservoir. Both extremes in pH caused swift mortality in adult mussels. From these studies it seems that pH adjustment could be used both as a preventative treatment to eliminate settlement by dreissenid mussels and as an end-of-season treatment to eliminate adults. However, high pH treatment would have to be tailored to site specific water quality to prevent or minimize formation of precipitate during treatment. Copper-based algaecides—Various copper based algaecides used both for control of aquatic weeds and algae have been reported as having an effect on dreissenid mussels when applied for control of aquatic weeds and algae. Copper products (copper sulfate and copper carbonates or 23 Green, R. F. 1995. Strategies for application of non-oxidizing biocides. Proceedings, Fifth International Zebra Mussel and Other Aquatic Nuisance Organisms Conference, Toronto, Canada, February 1995. 175–181. 24 Sprung, M. 1987. Ecological requirements of developing Dreissena polymorpha eggs. Archives of Hydrobiology Supplement. 79:69–86. D1–9 chelates) can be used to control mollusks in open water systems, but require a Special Local Need Label (also known as a Section 24-c) issued by the USEPA. In a study done on the Lower Colorado River and at San Justo Reservoir in California, various copper-based products were tested for their effect on both quagga and zebra mussels at concentrations generally used for weed and algae control. At those levels, all of the products resulted in significant mortality of dreissenid adults within 96 hours. Quagga mussels appeared more susceptible than zebra mussels to these copper-based products.25 Endothal—Two formulations of endothal-based algaecides exist. The first formulation is the dipotassium salt of endothal sold under the trade name of Cascade®. The second is the amine salt of endothal under the trade name of Teton for algae control and EVAC for the control of dreissenid mussels. In experiments carried out in mobile flow through laboratories, no significant mortalities were detected when either quagga or zebra mussels were exposed to various concentrations of the di-potassium salt Exposure to product containing amine salt resulted in significant quagga mussel mortalities. Sixty percent mortality was reached after 24 hours at 2 ppm and after 12 hours at 3 ppm at 20ºC. Both 2 ppm and 3ppm treatments showed 100% mortality after 96-hour exposure. These experiments were conducted in the Lower Colorado River. By comparison, zebra mussels in San Justo Reservoir, the site of the second experiment, exhibited significant mortality only at 3 ppm, after 60 hours. At 96 hours, mortality reached almost 40% and seemed to be still increasing when the experiment was terminated. Mortalities for both species increased with increased ambient temperature. For locations where algaecide treatment is required and amine salt of endothal is chosen, dreissenids will also be affected.26 Potassium salts—Potassium compounds are toxic to most bivalves, including dreissenids and corbiculids. Using flow through experiments in mussel infested waters, it was concluded that 20 mg/L caused 100% mortality in adults in 52 days while preventing any new settlement.27 Total mortality of small zebra mussels (7–11 mm) at a power plant on the Moselle River was achieved in 48 hours using 600 mg/L of KCl.28 In addition to causing mortality, potassium in various forms has Claudi, R., T. H. Prescott, S. Mastitsky, and H. Coffey. 2013. Efficacy of copper based algaecides for control of quagga and zebra mussels. Report prepared for California Department of Water resources, RNT Consulting, Inc., Picton, Ontario. 26 Claudi, R., A. C. Taraborelli, and T. H. Prescott. 2013b. Experimental protocol for Endothal efficacy for control of quagga and zebra mussels. Prepared for Bureau of Reclamation, Task Order Number: R11PD81448. IDIQ Contract: R10PC80264. 27 Lewis, D. P., J. Piontkowski, E. Neuhauser, and J. Knowlton. 1996. Chronic exposure of adult and larval mussels at low level potassium concentrations: Laboratory studies. In Proceedings of the Sixth International Zebra Mussel and other Aquatic Nuisance Species Conference, Dearborn, USA, March, 1996. 28 Khalanski, M. 1993. Testing of five methods for the control of zebra mussels in cooling circuits of power plants located on the Mosselle River. In Proceedings of the Third International Zebra Mussel Conference, Toronto, Canada, February, 1993. 25 D1–10 been observed to prevent valve closure in mussels and reduced filtration rates.29 Potassium chloride is used primarily as an end-of-season treatment for a number of closed loop systems, such as fire protection systems. Potassium chloride seems to be particularly effective in these types of semi-static systems due to mode of action and the fact that it does not possess the inherent demand characteristics typical of oxidants. Concentrations between 88–288 mg/l are necessary to cause swift mortality in adults. Such concentrations will likely kill native mussels as well, but are non-toxic to fish. In 2006, KCl was used to successfully eradicate zebra mussels from a rock quarry pond in Virginia. A 100% kill was attained with minimal environmental impacts to other aquatic species and to the drainage waters downstream. This method seems promising if a lethal concentration of KCl can be maintained for a 2–3 week period. More information about this project can be found at: http://www. dgif. virginia. gov/zebramussels/index. asp. Although potassium compounds are non-toxic to higher organisms, such as fish, the toxicity to native bivalves makes the approval for use of potassium salts in once-through systems unlikely. Biobullets—The product known as “BioBullets” takes advantage of the potassium salt toxicity while overcoming the dreissenid mussel reaction to noxious substances by encapsulating a core of potassium chloride (KCl) with an edible non-toxic coating. These approx. 40 micron particles are readily ingested by dreissenid mussels, leading to a much faster death than the application of potassium chloride alone. BioBullets are not only toxic to dreissenids. Other filter feeders may also ingest the BioBullet particles leading to negative impacts on other species, as is the case with majority of other chemical control strategies.30 Copper ion generator—Dissolution of copper and aluminum anodes by electrolysis has been used to protect ship cooling systems from macrofouling for at least 40 years. Based on the marine experience, a series of experiments was conducted to determine if the same technology could be used against dreissenid mussels.31 They concluded that a continuous dose of 10 ppb of copper ion would limit veliger settlement in system protected. This technology was commercialized under the trademark of MacroTech® Copper Ion generator. Wisconsin Energy Corporation We Energies uses this copper ion technology to control dreissenid mussel infestation in its Oak Creek Power Plant service water system.32 Although the copper ion generator does not eliminate all macrofouling in the service water system, the level of infestation is acceptable to the plant personnel. As the toxicity of copper in freshwater systems is greatly influenced by water quality, one would anticipate that different bodies of water would require different levels of copper ion generation to control dreissenids. The discharge of copper ions into the aquatic ecosystem may not be permitted in all jurisdictions. This technology is currently being tested by the Bureau of Reclamation, and results are expected early in 2014. Wildridge, P. J., R. G. Werner, F. G. Doherty, and E. F. Neuhauser. 1998. Acute effects of potassium on filtering rates of adult zebra mussels Dreissena polymorpha. Journal of Great Lakes Research. 21:629–636. 30 Aldridge, A. D. C., P. Elliot, and G. D. Moggridge. 2006. Microencapsulated Biobullets for the control of biofouling zebra mussels. Environmental Science and Technology. 40:975–979. 31 Blume, J. W., P. C. Fraleigh, and W. R.v anCott. 1994. Evaluation of copper ion and aluminum floc for preventing settlement of zebra mussels. In Proceedings of the Fourth International Zebra Mussel Conference, Madison, USA, March 1994. 32 Babinec, J. 2003. Copper ion treatment for zebra mussel mitigation in house service water systems. Power Plant Chemistry. 5:539–547. 29 D1–11 Bacterial Toxin (Zequanox) —The naturally occurring bacterium Pseudomonas fluorescens strain CL145A is the active ingredient in a microbial molluscicide product registered under the brand name Zequanox (ABN MBI-401 SDP). Zequanox was registered in the United States and Canada in 2012 and is currently registered in most states where zebra mussels occur or may spread in the future. It is registered only for the control of zebra and quagga mussels and has not been shown to have any significant activity against Asiatic clam, other invasive mollusks, or native North American bivalves. The bacterium was discovered and developed by New York State Museum (NYSM) and licensed to Marrone Bio Innovations for commercialization. Pseudomonas fluorescens is ubiquitous in the environment, and lab studies have indicated that when zebra or quagga mussels ingest artificially high densities of strain CL145A, a toxin within these bacterial cells destroys their digestive system. Dead bacterial cells are equally as lethal as live cells, providing evidence that the mussels die from a toxin, not from infection. The current formulation of Zequanox is composed of 50% killed cells of Pseudomonas fluorescens strain CL145A and 50% food-grade tolerance-exempt inert ingredients (carriers in the spray drying process). Future formulations of the product may be freeze-dried or liquid. Laboratory trials to date have been very encouraging regarding non-target safety (Malloy 2008). At dosages that produced high zebra mussel mortality (76–100%), no bacteria-induced mortality has been recorded among any of the non-targets, including fish, ciliates, daphnids, and bivalves (Malloy 2008). Mortality in sensitive aquatic species, such as daphnia and rainbow trout, have only been induced at high concentrations (100–200mg/L) during very long exposure periods (96hr–30 days). Because predicted environmental concentrations (PECs) are very low and the product’s activity has a half-life of 12 hours, impacts to fish, native mollusks, algae, plants, and other aquatic organisms is highly unlikely. Although originally developed as an environmentally safe alternative for chlorination in power plants, the non-target safety of this bacterial control agent may allow this technology to also be used for zebra and quagga mussel control in open waters, such as lakes and rivers. Zequanox was registered for use in industrial sites in 2012, and an expansion of allowed uses, to include open waters, is currently pending approval at the Environmental Protection Agency in early-mid 2014. Further information on this control method can be found at www.marronebioinnovations.com. No-Growth Materials (anti-fouling paints)—Can be effective in preventing zebra mussel attachment, however, the leachate can be toxic to other organisms. Anti-fouling paints are expensive to use and only feasible in certain situations. The following three tables provide a more detailed look at these control methods including target populations, application rates, efficiency and toxicity. Table 2 details non-chemical methods. Table 3 describes chemical control methods, and Table 4 identifies some of the most common commercial products. Anti-fouling coatings—Historically, the majority of developmental work in antifouling paints/coatings has been directed towards prevention of barnacle growth on ships. In fresh water, antifouling coatings’ primary use has been for the prevention of dreissenid mussel attachment to structures exposed to raw lake water. D1–12 The following substrate preference by dreissenids was established33 by using settling plates; copper < galvanized iron < aluminum < acrylic < PVS < teflon < vinyl < pressure treated wood < black steel < pine < polypropylene < asbestos < stainless steel. For pipes the preference was as follows; copper < brass < galvanized iron < aluminum < acrylic < black steel < polyethylene < PVC < ABS. Anti-fouling coatings containing the biocide TBTO (tri-butyl tin oxide, an organotin oxide), have been used successfully for prevention of mussel attachment and growth in Europe. They are banned in Canada and in most of the states in the U. S. Copper has also been used successfully in antifouling coatings, but leaching of copper ions from such coatings may result in unacceptable copper concentrations in the discharge water. Johnson Screens Company has developed a copper alloy under the commercial name “Z-Alloy” for water intakes structure which has been reported as both successful and as having failed. Tests done by the US Bureau of Reclamation in 2008 on wedge wire screens made of Copper-Nickel (90-10) have shown quagga mussels settling heavily on this material within four month.34 The overall trend is toward the use of environmentally benign foul release coatings which form a physical barrier to attachment. The most promising coatings at this time are nontoxic, silicone based paints that prevent, or greatly decrease the strength of attachment. The silicone -based coatings usually require several different layers to be applied to a perfectly clean, white metal surface or very clean and almost dry (10% or less moisture level) concrete. This tends to make them very costly ($40 -$100/m2). In addition, the foul release coatings tend to perform better in areas of high or moderate flow, rather than in quiescent areas. US Bureau of Reclamation is currently conducting coating trials on the lower Colorado River.35 The following three tables provide a more detailed look at these control methods including target populations, application rates, efficiency and toxicity. Table 2 details non-chemical methods. Table 3 describes chemical control methods and Table 4 identifies some of the most common commercial products. Kilgour, B. W., and G. L. Mackie. 1993. Colonization of different construction materials by zebra mussel, Dreissena polymorpha. In Zebra Mussels: Biology, impact and control, T. Nalepa and D. W. Schloesser, eds. pp.167–174. Boca Raton: RC Press. 34 US Bureau of Reclamation. 2009. Preliminary evaluation of quagga mussel fouling potential for Cu-NI (90-10) wedge wire screen. Research Note: Zebra and quagga mussel research program. http://www.usbr.gov/mussels/research/docs/researchnotes/ZQMRP-2009-RN-01-CU-NI.pdf 35 http://www. usbr. gov/lc/region/programs/quagga/researchnotes/04CoatingsStudy. pdf 33 D1–13 TABLE 2: Non-chemical treatment methods for dreissenid control. METHOD Primary TARGET AGE Thermal shock All 100% Freezing Juveniles Adults 100% Oxygen starvation – stripping water of oxygen Oxygen Starvation Benthic mats Desiccation All Juveniles Adults Juveniles Adults Manual removal Juveniles Adults All Veligers Variable Acoustic Deterrents Low frequency sound Acoustic Deterrents Ultra sound Acoustic Deterrents Vibration Acoustic Deterrents Plasma pulse technology Electrical Deterrents Low voltage electricity Filtration Media filters Filtration Self-cleaning mechanical filters UV radiation Veligers Veligers, translocators Not commercially available Not commercially available Not commercially available Prevents settling – Not commercially available Not commercially available 100% Veligers, translocators 100% Veligers Bacterial toxin (Pseudomonas fluorescens) All 100% prevention of settlement 95% Predation Acoustic Deterrents Cavitation All Veligers Juveniles Veligers Veligers Potential EFFICACY CONTACT TIME /CONCENTRATION COMMENTS 13 hours @ 33 °C (winter) 1 hour @ 37 °C (summer) 2 days @ 0 °C 5-7 hours @ –1. 5 °C under 2 hours @ –10 °C 2 weeks + @ 0 mg/l Lethal to most aquatic species Up to 99% 9 weeks Initial tests promising for limited infestations 100% Immediate @ 36 °C 5 hours @ 32 °C 2. 1 days @ 25 °C N/A Must dewater system for several days Low Not commercially available Continuous veligers in seconds @ 10–380 kHz juveniles in minutes adults in a few hours 4 to 12 min @ 20 Hz–20 kHz Inhibits settling veligers in seconds @ 39–41 kHz adults in 19-24 hours intermittent @ 200 Hz & 10–100 kHz intermittent high energy pulses immediate results @ 8 volt AC Prevents settling Removal of all particles greater than 80 microns Removal of all particles greater than 80 microns 6 hours Must dewater system Must isolate population Ongoing efforts in Lake George, New York and Lake Powell Harvest of potential predatory species should be limited May affect other species, reduced success in high flows, needs power source Not lethal, needs power source May impact other species, needs power source Structural integrity may be threatened Not lethal, private technology Not lethal, needs power source Removes all plankton, high total suspended solids may be a problem Removes all plankton, high total suspended solids may be a problem Lethal to many species, effectiveness may be limited by turbidity and suspended solids Low toxicity to other organisms, few treatments needed, not yet available in commercial quantities. Extensive information on treatment methods, including information sources, application methods, hazards, etc. is available on the US Army Corps of Engineers website. Information on the bacterial toxin, Pseudomonas fluorescens is available on the National Energy Technology Laboratory website. D1–14 TABLE 3: Chemical treatment methods for dreissenid control. NON- OXIDIZING CHEMICALS TARGET AGE EFFICIENCY CONTACT TIME/ CONCENTRATION Veligers 100% Adults 100% 69. 9% 52. 4% Potassium salts (KCL) All Potassium ion (KH2PO4) Potassium ion (KOH) Copper ions All All Veligers Prevent settlement 50% 95-100% 100% 100% 100% Copper-based algaecides All EarthTec® CaptainTM adults adults 100 85% 0. 5mg/l copper equivalent in 96 hours 1. 0mg/l copper equivalent in 96 hours NatrixTM adults 85-100% 1. 0mg/l copper equivalent in 96 hours Copper Sulfate adults 50-99% 0. 5mg/l copper equivalent in 96 hours Adults – Quagga 100% 36 hours of exposure at 1 ppm 24 hours of exposure at 2 ppm 12 hours of exposure at 3 ppm at ambient temperature of 25º C At 20º C ambient water temperature 96 hours of exposure at 1 ppm 84 hours at 2 ppm 24 hours of exposure to 3 ppm. pH adjustment COMMENTS under 7 or over 9. 5, pH close to limit prevents settlement pH 3 in 96 hours pH 2 in 96 hours ph4 in 96 hours High pH may cause unacceptable precipitation in water with high scaling index 50 mg/l 48 hours @ 150 mg/l 3 weeks @ 95–115 mg/l continuous @ 160–640 mg/l Less than 10 mg/l 24 hours @ 5 mg/l 10 15µg/L continuous prevents settlement Lethal to other mussel species, non-toxic to fish at required dose rate As above As above Lethal to other aquatic species Lethal to other aquatic species, efficacy increases with increasing ambient temperature Equally effective on zebra and Quagga Difference in efficacy between quagga and zebra mussels Difference in efficacy between quagga and zebra mussels Difference in efficacy between quagga and zebra mussels Endothal based algaecides Teton - amine salt of endothal Adult zebra Cascade – di-potassium salt of endothal none 2% 100% 100% 75% 0 Efficacy differs between quagga and zebra mussels. Efficacy increases with ambient water temperature 96 hours at 3ppm at 25º C Extensive information on the chemical treatment methods listed above, including information sources, application rates, toxic effects, hazards, etc. is available on the US Army Corps of Engineers website at www. el. erdc. usace. army. mil/zebra/zmis/idxlist. htm. D1–15 TABLE 4: Chemical treatment methods (commercial products) for dreissenid control. PROPRIETARY MOLLUSCICIDES QUATERNARY AMMONIUM COMPOUNDS Clam-Trol CT 1 TARGET AGE All EFFICIENCY 100% 48 hours after exposure CONTACT TIME/ CONCENTRATION 1. 95 mg/l @ 11 °C for 12 hours 1. 95 mg/l @ 14 °C for 14 hours COMMENTS More toxic to veligers than adults and more toxic to mussels than to trout. Must be deactivated with bentonite clay 1. 95 mg/l @ 20 °C for 6–14 hours Calgon H-130 All 100% after 48 hours 0. 85–1. 12 mg/l 1. 1 mg/l toxic to salmonids, must be deactivated, corrosive, flammable Macro-Trol 9210 All 100% 5–50 mg/l continuous Lethal to aquatic organisms, must be deactivated with bentonite clay Bulab 6002 All 100% 2 mg/l 7–10 days Lethal to fish, especially salmonids. Must be deactivated with bentonite clay 4 mg/l 5–8 days PROPRIETARY MOLLUSCICIDES AROMATIC HYDROCARBONS TARGET AGE Mexel 432 Deters veliger settlement EVAC – endothal formulation All Bulab 6009 All EFFICIENCY CONTACT TIME/ CONCENTRATION Dose at 1–4 mg/l once a day 96 hours LC 50 for rainbow trout 11mg/l, corrosive 100% 0. 3–3 mg/l for 5 to 144 hours Lethal to fish but rapidly degrades, does not bioaccumulate 100% 2 mg/l 4 to 10 days 96 hours LC 50 for rainbow trout 1,1 mg/l, corrosive 4 mg/l 3 to 8 days OXIDIZING CHEMICALS Chlorine TARGET AGE Veligers COMMENTS EFFICIENCY 100% CONTACT TIME/ CONCENTRATION 0. 3mg/l TRC (total residual chlorine) settlement prevention 1mg/l TRC 7 to 14 days D1–16 COMMENTS Lethal to many aquatic species , can be detoxified on discharge with sodium metabisulphite PROPRIETARY MOLLUSCICIDES QUATERNARY AMMONIUM COMPOUNDS Chlorine dioxide ClO2 TARGET AGE EFFICIENCY Adults 100% depending on ambient water temperature All 100% 0. 3mg/l TRC (total residual chlorine) settlement prevention All 100% Veligers 0. 3mg/l TRC (total residual chlorine) settlement prevention 1mg/l TRC 7 to 14 days depending on ambient water temperature Adults Hydrogen peroxide 100% 6 hours High dosage rates required. Lethal to other aquatic species. Short half-life in water 100% Veligers in 0. 3ppm continuous prevents settlement Lethal to other aquatic species, very short half-life in water, generally no need to detoxify on discharge Juveniles Ozone All COMMENTS 1mg/l TRC 7 to 14 days depending on ambient water temperature Adults Chloramine CONTACT TIME/ CONCENTRATION 5 hours @ 0.5 mg/l total mortality Adults in 7 days @ 0.5 mg/l depending on ambient temperature Potassium permanganate All 100 % 2. 0 mg/l for 48 hours Sodium permanganate All 100% 0. 3 mg/l to 1. 25 mg/l D1–17 lethal to other species, at high doses may turn water pink The commercial products listed above have been approved for aquatic use by EPA if applied according to label instructions by a licensed applicator. It is important to note that they may not have been approved by the individual states and must have that approval before they can be applied. The molluscicides have been primarily developed for use at water impoundment and hydropower facilities, treatment facilities, water intake structures, etc. Their use in open water is not generally recommended but might be possible under certain circumstances. For example, the herbicide Endothal, has been shown to be effective against zebra mussels and has been permitted for use in open waters in Washington State to control noxious weeds. Extensive information on the products listed above, including manufacturer, chemical formulation, application rates, toxicity, hazards, etc. is available on the US Army Corps of Engineers website. D1–18