Disease name
advertisement

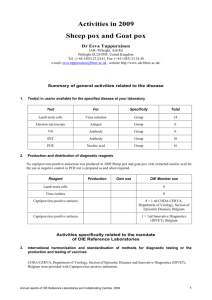
OIE Reference Laboratory Reports Activities in 2011 Name of disease (or topic) for which you are a designated OIE Reference Laboratory: Address of laboratory: Sheep pox and Goat pox Sheep pox and Goat pox Reference Laboratory, Razi Vaccine and Serum Research Institute, Hessarak, Karaj PO Box 31975/148 IRAN Tel.: (98-21) 3117908 Fax: (98-261) 4552194 e-mail address: hr_varshovi @yahoo.com, int@rvsri.com website: Name (including Title and Position) of Head of Laboratory (Responsible Official): Name(including Title and Position) of OIE Reference Expert: Hamid Reza Varshovi, DVM, PhD Head of Animal Viral Vaccines Department Hamid Reza Varshovi, DVM, PhD Head of Animal Viral Vaccines Department Name (including Title and Position) of writer of this report (if different from above): Annual reports of OIE Reference Centres, 2011 1 Sheep pox and Goat pox Part I: Summary of general activities related to the disease 1. 2. Test(s) in use/or available for the specified disease/topic at your laboratory Test For Specificity Total Virus neutralisation test (VNT) based on NI Antibody Group 15 VNT Antigen Group 30 Electron microscopy (EM) Virus detection genus 1 Susceptible animal inoculation Virus detection Group 30 Polymerase chain reaction (PCR) DNA detection genus 48 PCR-RFLP DNA detection Host species 25 Lamb kidney (LK) and lamb testis (LT) and Vero cell cultures Virus isolation - 3 Production and distribution of diagnostic reagents Type of reagent Amount supplied nationally (including for own use) Sheep pox reference virus (MR/ 65 strain) own laboratory use Goat pox reference virus (Goargan strain) own laboratory use Sheep pox & Goat pox virulent challenge viruses own laboratory use GPV antigen 40 ml SPV antigen 50ml GPV hyperimmune serum 80 ml Part II: Activities specifically related to the mandate of OIE Reference Laboratories 3. International harmonisation and standardisation of methods for diagnostic testing or the production and testing of vaccines a) Establishment and maintenance of a network with other OIE Reference Laboratories designated for the same pathogen or disease and organisation of regular inter-laboratory proficiency testing to ensure comparability of results None 2 Annual reports of OIE Reference Centres, 2011 Sheep pox and Goat pox b) Organisation of inter-laboratory proficiency testing with laboratories other than OIE Reference Laboratories for the same pathogens and diseases to ensure equivalence of results None 4. Preparation and supply of international reference standards for diagnostic tests or vaccines Not yet applicable 5. Research and development of new procedures for diagnosis and control Development of Real Time PCR technique for detection and differentiation of SPV and GPV is undergoing. 6. Collection, analysis and dissemination of epizootiological data relevant to international disease control Molecular characterization of Iranian Capripoxvirus isolates based on GPCR gene is accomplished and the findings is published through a PhD thesis. The national Project on molecular epidemiology of SGPV in Iran is planned for 2012 7. Maintenance of a system of quality assurance, biosafety and biosecurity relevant to the pathogen and the disease concerned None 8. Provision of consultant expertise to OIE or to OIE Member Countries Collaboration on Animal diseases prioritization Project of OIE for East Africa (sheep and goat pox section) 9. Provision of scientific and technical training to personnel from other OIE Member Countries At Razi Institute, the training on Capripoxvirus and Parapoxvirus diagnostic methods as well as sheep pox & goat pox vaccine production is available. 10. Provision of diagnostic testing facilities to other OIE Member Countries None requested in 2011 11. Organisation of international scientific meetings on behalf of OIE or other international bodies None 12. Participation in international scientific collaborative studies None 13. Publication and dissemination of information relevant to the work of OIE (including list of scientific publications, internet publishing activities, presentations at international conferences) No presentation or publication for 2011 _______________ Annual reports of OIE Reference Centres, 2011 3