PP 16: Thermochemistry
advertisement

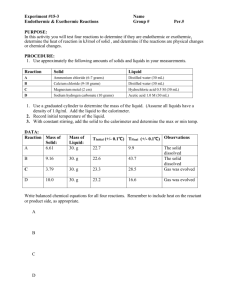
PP 16: Thermochemistry Drill: Calculate the volume of gas released at 227oC under 83.1 kPa pressure when 320 kg of NH4NO3 is exploded forming N2, O2, & H2O. Drill: Draw LDDs for: SCl2 CH2Cl2 HSO4-1 C4H6 Thermochemistry: The study of heat transfer in chemical reactions Thermochemical Terms Heat (H): A form of energy that can flow between samples of matter energy Heat cannot be measured directly; thus, we measure heat change (H) Enthalpy Change: Heat that can flow in/out of a system (H) System: That part of nature upon which attention is focused Surroundings: That part of nature which we are not focused Reaction Coordinate: A graph of energy change versus time in a chemical reaction Time Exothermic Reaction: Chemical reactions that release, give off heat, or lose heat Products will contain less heat than the reactants or H < 0 or negative Endothermic Reaction: Chemical reactions that absorb, take in heat, or gain heat Products will contain more heat than the reactants or H > 0 or positive Thermochemistry Topics: • • • Heat Change Calorimetry Hess’s Law & the Thermochemical Equation (TCE) Heat Change: Calculating the change of heat within a system Specific Heat [C]: The heat required to raise one gram of a substance 1oC Specific Heat Formula: H = mCT Heat of Fusion (Hf): • The heat required to melt one gram of a substance at its normal MP • Hf: (J/g or J/kg) Heat of Fusion Formula: H = mHf Heat of Vaporization (Hv): • The heat required to boil one gram of a substance at its normal BP • Hv: (J/g or J/kg) Heat of Fusion Formula: H = mHv C: (J/goC, J/kgK) Problem: Calculate the heat change when 10.0 g H2O goes from –10.0oC to 120.0oC. MP = 0oC cice = 2.06 J/gK BP = 100oC cwater = 4.18 J/gK Hf = 334 J/g HV = 2260 J/g csteam = 2.02 J/gK Problem: Calculate the heat change when 5.00 g H2O goes from –100.0oC to 200oC. MP = 0oC cice = 2.06 J/gK BP = 100oC cwater = 4.18 J/gK Hf = 334 J/g HV = 2260 J/g csteam = 2.02 J/gK Drill: Calculate the heat change when 25 g H2O goes from 140.0oC to 60.0oC. MP = 0oC cice = 2.06 J/gK BP = 100oC cwater = 4.18 J/gK Hf = 334 J/g HV = 2260 J/g csteam = 2.02 J/gK Calorimetry: The study of how to measure experimental heat transfer in a system Calorimeter: • The device used to measure heat transfer • A calorimeter is an adiabatic system • Adiabatic Systen • A system that exchanges zero heat with its surroundings • Hsystem = 0 • A calorimeter is used to measure experimental yields Heat or enthalpy change (H ~ q): Hsystem = Hall parts Calorimetry calculations: Hsystem = 0 Hsys = Hcal + Hrxn Hrxn = -Hcal (all parts) Hrxn = -mcDTcal (all) Problem: Calculate Hrxn when Q reacts in a 1.5 kg calorimeter containing 2.5 kg water. The temperature changes from 22.50oC to 26.50oC. (Cwater = 4.18 J/gK & Ccal = 2.00 J/gK) Problem: When X reacts in a 2.0 kg calorimeter containing 1.5 kg water, the temperature changes from 22.50oC to 26.50oC. Calculate Hrxn (Cwater = 4.18 J/gK & Ccal = 2.00 J/gK) Problem: When Z reacts in a 2.0 kg calorimeter containing 2.0 kg water, T = 20.0oC to 0.0oC & 50.0 % of the water freezes. Calculate Hrxn (Cwater = 4.18 J/gK, Ccal = 2.00 J/gK, & Cice = 334 J/g)) Drill: When Z reacts in a 1.0 kg calorimeter containing 2.0 kg water, T = 30.5oC to 20.5oC. (Cwater = 4.18 J/gK & Ccal = 2.00 J/gK) Problem: When 40.0 g NH4NO3 dissolves in 460.0 g water at 25.0oC, the temp. falls to 22.5oC. Calculate the Ho per mole of NH4NO3. Csoln = 4.00 J/gK Thermochemical Equation (TCE) Thermochemical Equation Terms: Heat of Reaction (Hrxn): The heat or enthalpy change of a chemical reaction Heat of Solution (Hsoln): The heat or enthalpy change when a substance is dissolved Heat of Combustion (Hcombustion): The heat or enthalpy change when a substance is burned Heat of Formation (Hof): The heat required to form one mole of a compound from pure elements Heat of formation is measured in kJ/mole. The Degree Symbol: Indicates standard conditions & molar quantities by itself or from a balanced equation. Gibb’s Free Energy (G): • Energy of a system that can be converted to work • Determines spontaneity Exergonic Reaction: A reaction in which free energy is given off & G < 0 or G is negative. Engergonic Reaction: A reaction in which free energy is absorbed & G > 0 or G is positive. Reaction at Equilibrium: G = 0 Gibb’s Free Energy of Formation or Energy of Formation (Gof): • The Gibb’s Free Energy required to form one mole of a compound from pure elements • Gibb’s Free Energy of formation is measured in kJ/mole. Entropy (S): A measure of disorder generally measured in J/K. Entropy of Formation (Sof) measured in J/moleK: The entropy of one mole of a substance Interrelation Term (G): G is the term that interrelates thermochemistry, chemical equilibria, & electrochemistry Problem: When 10.0 g NH4NO3 dissolves in 400.0 g water at 25.0oC, the temp. falls to 20.0oC. Calculate the Ho per mole of NH4NO3. CHOH = 4.18 J/gK CAN = 0.50 J/gK Drill: Define: Heat of reaction, Heat of solution, Heat of formation, Free Energy of formation, & Entropy Thermochemical Equation (TCE): An equation to determine changes in heat, energy, etc Thermochemical Equation (TCE): Horxn = Hfoproducts - Hforeactants Gorxn = Gfoproducts - Gforeactants Sorxn = Sfoproducts - Sforeactants Thermochemical Equation (TCE): Is called the stoichiometry of heat change because this equation is used to determine the theoretical change of a chemical reaction. The 3rd step in stoichiometry involves converting what’s given to what’s asked for. The TCE is used the determine the heat change per mole Use the TCE for the 3rd step of stoichiometry to convert moles given to heat or free energy. Interrelating Equation: G = H - TS Drill: Identify type of rxn when: G < 0 G > 0 G = 0 Thermochemical Equation (TCE) & Interrelating Equation: Xorxn = Xfoproducts - Xforeactants G = H - TS Problem: Use the table below to Calculate H, G, & S for the reaction when 13.6 g of CaSO4 is converted to CaO + SO2 + O2 at 25oC Cmpd CaSO4 SO2 CaO Hfo (kJ/mole) -1434.1 -296.8 -635.1 Gfo (kJ/mole) -1321.8 -300.2 -604.0 Problem: Use the table below to Calculate H, G, & S for the reaction when 19.7 kg of BaCO3 is converted to BaO + CO2 at 25oC Cmpd BaCO3 CO2 BaO Hfo (kJ/mole) -1216.3 -393.5 -553.5 Gfo (kJ/mole) -1137.6 -394.4 -525.1 Drill: Calculate Horxn & Gorxn for the following reaction: __A + __B2C3 __AC2 + __B Cmpd B2O3 AC2 Hfo (kJ/mole) -150 -250 Gfo (kJ/mole) -175 -225 Problem: Calculate Ho, Go, & S for the following reaction at -23oC: _AD2 + _BC _AC2 + _BD Also calculate SfoBD. Cmpd BC AD2 AC2 BD Hfo (kJ/mole) -150 -250 -300 -175 Gfo (kJ/mole) -125 -225 -250 -150 Sfo (J/moleK) 75 50 80 ? Drill: Calculate Horxn & DGorxn for the following reaction: Cmpd QR PR2 Hfo (kJ/mole) -250 -450 Gfo (kJ/mole) -225 -425 _P + _QR _PR2 + _Q Practice for Lab calculations: In lab, a calorimeter was assembled with some water in a cup. The initial temperature of the system was measured. A small amount of solid KOH was added to the system, & the final temperature of the water was measured. Use the results from the experimental data table to determine Hexperimental. Use the results from the heat of formation data table to determine Htheoretical. Hfo Tables Experimental Data Table App. Cup H2 O KOH Thermo Cmpd Hfo(kJ/mole) Mass 5.00 g 50.00 g 2.00 g 20.00 g KOH -424.7 K+ -252.4 OH- -230.0 Initial Temp. =- 22.0oC Final Temp. = 30.0oC Problem: Use the information from the table below to calculate the theoretical Ho, Go, & So for the reaction involving the burning of C3H8 : Cpd Hfo Gfo C3H8 CO2 -103.8 -393.5 - 23.5 -394.4 All units (kJ/mole) H2O -241.8 -228.6 Drill: Calculate Ho, Go, S, & at -23oC when: Compd Hfo (kJ/mole) Gfo (kJ/mole) BC -150 -175 _A + _BC _AC + _B AC -250 -225 Problem: When 35.0 g NaOH dissolves in 965 mL water in a 2.5 kg calorimeter, the temp. went from 22.5oC to 26.5oC. Calculate: Hosoln (Csoln = 4.00 J/gK) (Ccal = 2.00 J/gK) Problem: Use the data in the table below & the following reaction to calculate Ho, Go, & So, Teq, & Ho of 48 g PbO2: PbO2 + CO CO2 + Pb Cpd Hfo (kJ/mole) Gfo (kJ/mole) PbO2 CO CO2 -277.4 -217.4 -110.5 -137.2 -393.5 -394.4 Problem: Use the data in the table below & the following reaction to calculate the potential H, G, & S for the reaction & Sfo for O2 when burning 8.8 kg of C3H8: Cpd C 3 H8 -103.8 - 23.5 269.9 Hfo (kJ/mole) Gfo (kJ/mole) Sfo (J/mole K) CO2 -393.5 -394.4 213.6 H2O -241.8 -228.6 188.7 Drill: Use the data in the table to the right to calculate H, G, & S at 298 K for the production of 3.4 g of NH3 from N2 & H2. Compd Hfo (kJ/mole) Gfo (kJ/mole) NH3 -46.1 kJ/mole -16.5 kJ/mole Test Review: Problem: Use the data in the table below & the following reaction to calculate Ho, Go, & So: __ N2O5 + __ H2O __ HNO3 Hfo Gfo Cpd (kJ/mole) (kJ/mole) N2O5 -11.3 -10.4 H2O -285.8 -237.2 HNO3 -174.1 -151.5 Problem: When 10.8 g N2O5 is added to a 1.50 kg calorimeter containing 1.00 kg of H2O, the N2O5 reacts with the water forming HNO3. The temperature of the system changes from 23.000oC to 23.750oC. Calculate the Hrxn/mole N2O5. (Ccal = 1.00 J/gK) (C = 4.18 J/gK) Problem: The solution for the 1st problem is the theoretical yield for the production of HNO3 in the balanced reaction using molar quantities. The solution for the 2nd problem is the experimental yield for the production of HNO3 from 10.8 grams of N2O5. Calculate the % yield of the reaction. Problem: When 25.0 g CaCO3 is made from CaO & CO2 in a 500. g calorimeter containing 250.0 g of water, the system changes from 25.0oC to 45.0oC. Calculate Hrxn/mole of CaCO3: (Ccal = 1.50 J/gK) (CH2O = 4.18 J/gK) Problem: Use the information from the data table & the following reactions to solve the Hrxn for the entire reaction mechanism: Substance Hof (kJ/mole) Reaction Mechanism C + A 2H+P Sub. Hof Sub. Hof C + A 2H+P A -100 H -150 D + B 2K+P B -150 K -200 H+K P+Q C -50 P -250 D -125 Q -300 Heat Change Problem: Calculate the heat change when the temperature of 1.0 kg H2O is changed from –100.0oC to 200.0oC. Problem: Use the information from the data table to calculate Ho, Go, & S at 227oC for the production of ammonia from N2 & H2. Compd Hfo (kJ/mole) Gfo (kJ/mole) NH3 -46.1 -16.5 Extra: 1st Law Thermodynamics: Total energy change = heat + work E = q + W) Derivation from the work formulas: • • • • W = Fd P = F/A V = Ad W = PV = nRT 2nd Law Thermodynamics: Total entropy in a system always increases assuming no energy is added to the system Thermodynamic Rxns are State Rxns: State Reaction: Reactions that are independent of the path; thus not dependent on intermediates