Research Governance Framework
advertisement

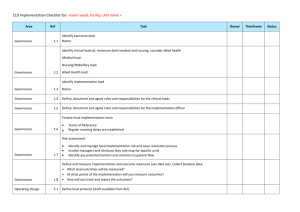
RESEARCH GOVERNANCE FRAMEWORK INTRODUCTION The South Australian Health and Medical Research Institute (SAHMRI) is committed to conducting and supporting methodologically and ethically sound research that adheres to, and is conducted in accordance with, ethical principles, guidelines for responsible research conduct, legislation and regulations. Core components of proper research conduct, as outlined by the Australian Code for the Responsible Conduct of Research (2007), are sound research management and research governance. Responsibility for ensuring research governance requirements are met falls principally to the institution that hosts the research and the researchers who conduct health and medical research. To address this, SAHMRI has developed a Research Governance Framework for all research involving human participants to ensure compliance with these requirements. This Research Governance Framework is intended to provide guidance to researchers, both those employed by SAHMRI and external researchers seeking access to the SAHMRI site, data and/or staff, of SAHMRI’s research governance requirements. SCOPE OF THE FRAMEWORK The SAHMRI Research Governance Framework applies to researchers involved in the conduct of HREC approved health and medical research involving humans that: Is conducted on the SAHMRI site (i.e. the North Terrace Building) and includes the clinical rooms on Level 4; and/or Involves the use of samples/data/records or other material held and managed by SAHMRI. The Framework applies equally to employees of SAHMRI and external researchers who are undertaking research at the SAHMRI site. This Framework does not apply to research undertaken by SAHMRI staff at external institutions e.g. public health organisation. Local policies will apply at these institutions and must be adhered to at all times. Compliance with this Framework is mandatory. 1 RESEARCH GOVERNANCE – A background Research governance implements the principles, requirements and standards of research. It addresses protection of research participants, the safety and quality of research, privacy and confidentiality, financial probity, legal and regulatory matters, risk management and monitoring arrangements and promotes good research culture and practice. (NHMRC, Research Governance Handbook). Research Governance has two core components: Completion and authorisation of a Site Specific Assessment; and Ongoing monitoring of research conduct. Researchers are reminded that ethical review and receipt of approval for their project by a properly constituted Human Research Ethics Committee is a significant part of good research governance and forms a key component of the Site Specific Assessment. Please note that the ethics approval is a separate process to the Research Governance review. The review and authorisation of the Site Specific Assessment can occur at any time, and researchers are encouraged to submit the SSA for authorisation in parallel with the ethics process. The decision to authorise or not authorise the SSA, will be subject to receipt of a copy of the approval from the appropriate Human Research Ethics Committee. Researchers are encouraged to plan sufficient time within their project timetable to obtain both ethics approval and research governance authorisation. The research project must not commence until both HREC approval and Research Governance approval has been granted. Role of the Research Governance Officer SAHMRI has appointed a Research Governance Officer (RGO) to support researchers in compliance with this Framework. The RGO will also: Review the submitted Site Specific Assessment forms and make recommendations to the Executive Director (SAHMRI) regarding authorisation of the project; Liaise with researchers, ethics officers, research sponsors and other key staff and bodies involved in the conduct and management of research (as necessary); Monitor research conducted at the SAHMRI site; Develop processes, systems and methods for the effective governance of research at SAHMRI; and Undertake risk management assessments and procedures to promote responsible research conduct. 2 Site Specific Assessment (SSA) Authorisation The purpose of the SSA is to provide information to SAHMRI about the research being conducted on its site, with its data/records/samples and its staff. This information then allows the Executive Director of SAHMRI, or delegate, to make an informed decision as to whether the research project should be undertaken at the SAHMRI site. It is the responsibility of the Principal Investigator to complete and submit the SSA to the Research Governance Officer at SAHMRI. A copy of the Site Specific Assessment Form can be requested from the Research Governance Officer via email at researchgovernance@sahmri.com or via phone (08) 8128 4110. SSAs will generally be reviewed within 10 days from submission to the RGO. For more complicated submissions, a longer review period may be required. The RGO will ensure that the researcher is advised of the timeframe regarding review. Incomplete SSAs will not be accepted by the RGO and will be returned to the researcher for completion prior to review. 3 REFERENCE DOCUMENTS The key policy documents and guidelines that relate to this Framework are: SAHMRI Procedures for Research Practice; Australian Code for the Responsible Conduct of Research (2007); National Statement on Ethical Conduct in Human Research (NHMRC) (2007); Access to Unapproved Therapeutic Goods – Clinical Trials in Australia (TGA); Values and Ethics: Guidelines for Ethical Conduct in Aboriginal and Torres Strait Islander Research (NHMRC) (2003); International Conference on Harmonisation / Good Clinical Practice Guidelines (ICHGCP Guidelines); Medicines Australia Principles on Conduct of Clinical Trials and Communication of Clinical Trial Results; Australian Clinical Trials Handbook; Privacy laws including the Privacy Act 1988 and National Privacy Principles; 4