Assignment - WordPress.com
advertisement

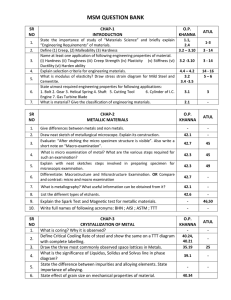
CHAPTER 1 INTRODUCTION 1) State the importance of study of “Materials Science” and briefly explain “Engineering Requirements” of materials 2) Name at least one application of following engineering properties of material. (i) Hardness (ii) Toughness (iii) Creep Strength (iv) Plasticity (v) Stiffness 3) What is modulus of elasticity? Draw stress strain diagram for Mild Steel and Cementite. 4) Define 1) Toughness 2) Hardness 3) Hardenability 4) Malleability 5) Creep 6) Elasticity 7) Ductility . 5) Give differences between metals and non metals. 6) Define (1) Creep, (2) Malleability (3) Hardness 7) Explain selection criteria for engineering materials. 8) Draw neat sketch of metallurgical microscope. Explain its construction. 9) Distinguish between hardness and harden ability 10) What are the factors affecting hardnability? 11) Discuss selection criteria for materials used in engineering applications 12) State the importance of study of materials science and briefly. Explain Engineering requirements of materials. 13) Explain selection criteria for engineering materials. CHAPTER 2 Metallic Materials 1) Evaluate: “After etching the micro specimen structure is visible”. Also write a short note on “Macro-examination”. 2) Evaluate “After etching the micro specimen structure is visible”. Also Write a short note on macro- examination. 3) Explain the Spark Test and Magnetic test for metallic materials. 4) Explain with neat sketches the arrangement of atoms in B.C.C, F.C.C. and H.C.P. lattice. Define unit cell. Show that a F.C.C. structure is always more close packed than B.C.C. structure. 5) Explain with neat sketches steps involved in preparing specimen for microscopic examination. 6) Draw the three most commonly observed space lattices in Metals. 7) Write full names of following acronyms: BHN ; AISI ; ASTM ; TTT 8) What is metallography? What useful information can be obtained from it? 9) What is coring? Why it is observed? 10) What is micro examination of metal? What are the various steps required for such an examination? CHAPTER 3 Iron-Carbon diagram 1) With the aid of steel portion of an iron-iron carbide equilibrium diagram, showing solid state transformations compare the transformations during cooling under equilibrium conditions from solidus to room temperature of typical hypereutectoid steel and hypoeutectoid steels. Compare the resulting microstructure at room temperature and related properties. 2) State Critical Reactions of Iron Carbon Phase diagram. 3) Draw Iron-Carbon Diagram and explain eutectic reaction in it. 4) Explain allotropic transformation of iron. 5) Draw TTT diagram for eutectoid steel. Explain it briefly by considering few cooling rates. 6) Differentiate between austempering and martempering. 7) Briefly explain why ferritic and austenitic stainless steels are not heat treatable. 8) Draw microstructure of (i) 04 % carbon steel and (ii) eutectoid steel at room temperature. 9) Draw iron – iron carbide equilibrium diagram with all necessary details. 10) Briefly explain cooling of 1.2 % carbon steel from liquid state to room temperature. 11) Give composition, properties and uses of Y alloy. 12) Define Allotropy. Explain allotropy of Iron. 13) Draw Iron Carbon Equilibrium diagram. 14) Draw microstructure of eutectoid steel 15) Draw microstructure of eutectic cast iron. CHAPTER 4 Crystallization of metals 1) What is Gibb’s phase rule? Define system, phase and degree of freedom. 2) Show that the degree of freedom at eutectic point in a binary phase diagram is zero. 3) The following data is for Pb-Sn alloy system : (Lead-Tin Solder)(39.15) · Melting point of lead (Pb) - 327⁰C · Melting point of Tin (Sn) - 232⁰C · Eutectic alloy is formed at 183⁰C with 62% Sn –38% Pb · Maximum solid solubility of tin in lead at 183⁰C –19% · Maximum solid solubility of lead in tin at 183⁰C –3% · Maximum solubility of tin and lead at room temperature is negligible. (1) Draw the phase diagram with the help of above data and label all the points, lines and regions on it. (2) For 70%Pb – 30%Sn alloy composition, determine the amounts of proeutectic and eutectic constituents at room temperature. 4) Define Critical Cooling Rate of steel and show the same on a TTT diagram with complete labelling. 5) Explain in brief: Factors affecting formation of solid solution 6) Draw cooling curve of (i) pure metal and (ii) An alloy of two metals which are completely soluble in liquid and solid phase 7) Define critical cooling Rate of steel and show the same on a TTT diagram with complete labelling 8) What is Gibb’s phase Rule? Define system phase and degree of freedom so that the degree of freedom eutectic point in a binary phase diagram is zero. 9) What is cooling curve? How does the time temperature cooling curve of an alloy of eutectic composition different from that of a pure metal? 10) What is solid solution? Discuss in brief types of solid solution with neat sketch. 11) What is Gibb’s phase rule? Explain its importance. 12) What is solid solution? Explain types of solid solution. Also give 13) Hume Rothery’s Rules. 14) What is phase diagram? Explain Lever rule. 15) Say at point “Q” in (Liquid+Solid) region in a phase diagram, a line passing through point “Q” and parallel to the base is drawn. The line intersects the liquidus and solidus at points P and R respectively. Can you determine %Solid at point Q if PR is 6 cm and QR is 2.4 cm in length? If answer is YES, determine % Solid and if NO, justify your answer. 16) What is the significance of Liquidus, Solidus and Solvus line in phase diagram? State effect of grain size on mechanical properties of material. CHAPTER 5 Cast Iron: 1) Differentiate between gray cast iron & spheroidal cast iron in terms of microstructure, properties, composition & applications. 2) Give composition properties and uses of malleable cast iron 3) Give composition and use of White metal and Monel metal 4) Differentiate between white cast iron and grey cast iron. 5) Classify types of cast iron. Discuss any on. Draw its microstructure also. 6) Give composition, properties and uses of malleable cast iron. CHAPTER 6 Wrought Iron 1) 2) 3) 4) What is “Wrought Iron”? Enlist the properties and uses of it. 07 What is “ Wrought iron “? enlist the properties and uses of It. Explain properties and application of Wrought iron. List properties of wrought Iron. CHAPTER 7 Steel 1) Define “Alloy”. Also state composition, properties & uses of any two copper alloys. 2) State composition and specific applications of : Muntz metal ; German silver ; Naval brass 3) State the difference between impurities and alloying elements. State importance of alloying. 4) What are the properties of bearing alloys II. What are the properties of aluminium alloys 5) What are the purpose of Alloying ? Give effects of nickel as an alloying element. 6) Explain the effects on steel by alloying elements Silicon, Sulphur, Magnesium and Phosphorous. 7) What are the purposes of alloying? Give effects of Nickel as an alloying element. 8) State function of following alloying elements in steel : Sulphur ; Nickel ; Chromium ; Boron 9) Specify, with reasons alloy suitable for the manufacture of : Bolts and Nuts ; Lathe Bed ; Milling Cutter 10) Give composition and uses of Monel metal and Nichrome. Enlist properties of a good bearing material. CHAPTER 8 Heat Treatment of Steels 1) List the purposes of heat treatment. Differentiate between annealing & normalizing. 2) Discuss mechanisms of quenching of steel. State the advantages and drawbacks of water & oil as quenching media. 3) It is required to obtain very thin hardened surfaces on gears of automobiles. Suggest suitable methods with salient features. 4) Differentiate Annealing and Normalising on the basis of a. Rate of Cooling (ii) Microstructure after cooling (iii) Grain size distribution (iv) Internal Stresses (v) Mechanical properties and (vi) Applications. 5) What are the purposes of heat treat. Differentiate between annealing and non Normalizing 6) Which are various surface hardening processes? Explain induction hardening process with sketch 7) Case carburizing heat treatment is not generally carried out for medium carbon steels. Why? 8) Which are various Surface Hardening processes? Explain Induction Hardening process with sketch. 9) Explain flame-hardening process in brief 10) Explain the difference between Annealing and Normalizing. 11) Explain in brief: Sintering Process CHAPTER 9 Non-ferrous alloys: 1) Explain structure and properties of Malleable cast iron. 2) Explain modified aluminum silicon alloys. 3) How will you classify brasses based on the composition of zinc Explain the properties & application of the main type of brasses. CHAPTER 10 Powder Metallurgy 1) What is powder metallurgy? Describe various steps involved in powder metallurgy with each step controlling properties of final sintered component. 2) Define Powder Metallurgy. State advantages, limitations and applications of Powder Metallurgy. 3) Enlist methods of manufacturing metal powder. Discuss any one in detail. 4) Discuss the advantages, disadvantages and limitations of powder metallurgy. 5) Explain any two methods for production of metal powders. 6) Which are advantages and limitations of powder metallurgy? 7) Which are the objectives of Heat Treatment? 8) Give difference between Annealing and Normalising. 9) Enlist the products made from powder metallurgy. Explain all four steps of power metallurgy. 10) What is powder metallurgy? Describe various steps involved in powder metallurgy 11) with each step controlling properties of final sintered component 12) Which are merit and demerit and application of powder metallurgy. 13) Give advantages, limitation & applications of powder metallurgy. CHAPTER 11 Corrosion of metals 1) What are the factors affecting rate of corrosion? Explain the hydrogen evaluation & oxygen absorption mechanism of corrosion. 2) Discuss the Cathodic protection method of corrosion prevention for underground pipelines. 3) Write short note on Metallic coatings. 4) Explain cathodic protection against corrosion. 5) Explain use of Galvanic series 6) What is cathodic protection? 7) Explain Cathodic protection against corrosion. 8) Enlist different method of metal coating for corrosion prevention. Discuss any one in detail. 9) Explain metal and organic coatings. 10) What are the factors affecting rate of corrosion? Explain the hydrogen evaluation & 11) oxygen Absorption mechanism of corrosion. 12) Discuss the cathodic protection method of corrosion prevention For underground Pipelines 13) State and explain principle of cathodic protection CHAPTER 12 Non-destructive testing 1) State the advantages or importance of nondestructive testing over destructive testing of materials. Differentiate between X-ray radiography & γ-ray radiography of materials 2) Explain Ultrasonic method of inspection with reference to its working principle, advantages, limitations & applications. 3) What is non destructive test? List various non destructive tests. 4) Explain X ray Radiography. 5) Explain Ultrasonic testing with advantages and limitations. 6) Describe with neat sketch how would you carry out a Jominy harden ability test on a steel sample. 7) Write the procedure for Jominy end quench practical and discuss its conclusion. 8) Which non-destructive test is used for finding defects on welded joints? Explain it. 9) Explain principle and procedure of Ultrasonic test. 10) What is non destructive test? List various non destructive tests. Explain X-Ray Radiography. 11) Explain Ultrasonic Testing with Advantages and limitation. 12) Explain the NDT method widely used for inspection of castings. 13) Suggest and explain a simple and economical NDT method to determine minute surface defects in large size component.