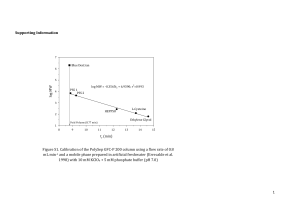
Is the geometry of Vanadium in active site of phosphatases
advertisement

Is the geometry of Vanadium in active site of phosphatases important for inhibitor design and antidiabetic properties of vanadium compounds D. C. Crans Department of Chemistry, Colorado State University, Fort Collins,CO ,USA e-mail: Debbie.Crans@ColoState.edu Vanadate, and vanadium compounds are insulin enhancing agents and representative compounds have been tested in animal systems and human beings.1-3 The specific mode of action and the active species have remained elusive such studies being complicated by the complex speciation. One mode of action of these compounds is believed to be inhibition of phosphatases.1-7 In the following presentation we will investigate the structural aspects of oxovanadate and vanadium complexes coordinated to phosphatases. Specifically, we examine the active sites of the 29 known X-ray structures and analyze the coordination chemistry of the vanadium atoms in the active site and characterize them with regard to trigonal bipyramidal or square pyramidal character. We also compare these systems with small molecule vanadium complexes to determine if the forms in the protein complex is similar to the small molecules characterized.8-10 These considerations demonstrate that a trigonal bipyramidal geometry is observed in the vanadium-phosphatase complexes even though the square pyramidal geometry is more stable for the complexes themselves. These observations were compared to the biochemical data available measuring Ki values for oxovanadates as well as other oxometalates and finally for vanadium compounds.9 We also use the data-mining and shape analysis to investigate the nature of the inhibition by mechanistic investigation of the hydrolysis of the PTP1B reaction surface. Together all these data are used to provide direction with regard to inhibitor design. References 1. K.H. Thompson, J. Lichter, C. LeBel, M.C. Scaife, J.H. McNeill, C. Orvig, J. Inorg. Biochem. 2009, 103, 554-558. 2. B. I Posner; R. Faure; J.W. Burgess; A. P. Bevan; D. Lachance; G. Zhang-Sun; I. G. Fantus; J. B. Ng; D. A. Hall; B. S. Lum J. Biol. Chem. 1994, 269, 4596-4604. 3. G.R. Willsky, L.-H. Chi, M. Godzala, III, P.J. Kostyniak, J.J. Smee, A.M. Trujillo, J.A. Alfano, W. Ding, Z. Hu, D.C. Crans, Coord. Chem. Rev. 2011, 255, 2258-2269. 4. Crans, D.C.; Keramidas, A.D.; Drouza,C. Phosphorus, Sulphur, and Silicon 1996, 109-110, 245-248. 5. McLauchlan, C.C.; Hooker, J.D.; Jones, M.A.; Dymon, Z.; Backhus, E.A.; Greiner, B.A.; Dorner, N.A.; Youkhana, M.A.; Manus, L.M. J. Inorg. Biochem. 2010, 104, 274-281. 6. Lu, L.; Yue, J.; Yuan, C.; Zhu, M.; Han, H.; Liu, Z.; Guo, M. J. Inorg. Biochem. 2011, 105, 13231328. 7. Lu, L.; Gao, X.; Zhu, M.; Wang, S.; Wu, Q.; Xing, S.; Fu, X.; Liu, Z.; Guo, M. BioMetals 2012, 25 (3), 599-610. 8. D.C. Crans, M.L. Tarlton, C.C. McLauchlan, Eur. J. Inorg. Chem., In press (2014). 9. C. C. McLauchlan, B. J. Peters, G. R. Willsky and D. C. Crans, Coord. Chem. Rev. 2015; ASAP, http://dx.doi.org/10.1016/j.ccr.2014.12.012 10. I. Sanchez-Lombardo, S. Alvarez, C. C. McLauchlan, and Debbie C. Crans J. Inorg. Chem. 2015, under revision. 1