STUDY EXPEDITOR

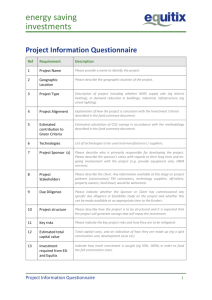
Study Expeditor
PROTOCOL(# or name) :
INVESTIGATOR
EMAIL PHONE
Are you a Washington University full-time faculty member?
Yes No
CONTACT (
for Investigator
l
PHONE
FAX
314-286-1187
CAMPUS BOX
8116
EMAIL FOR CHECK NOTIFICATION
SPONSOR NAME
:
CRO (if applicable):
CONTACT (for Sponsor or CRO)
PHONE FAX EMAIL
----------------------------------------------------------------------------------------------------------------------------- --------------------------------
IRB or Animal Studies approval: Approved Pending N/A Informed Consent : Enclosed Pending N/A
Who developed the protocol?
PI Industry Sponsor Non-industry Sponsor (i.e. other academic institution, foundation, NIH, etc.)
Both PI and Sponsor (If both, who initiated protocol?)
Type of Study: Inpatient Outpatient Both
Treatment Testing Both Data Collection Only
Phase I Phase II Phase III Phase IV N/A
Single Site Multi-Center If Multi-Center , # of sites: 1-10 11 or More Is Wash U Lead Site?
How many patients do you anticipate enrolling (at WU) ?
Device Trial: Yes No If Yes , specify category: A or B. Who holds the IDE#
Drug Trial: Yes No If Yes , Who holds the IND# If no IND#, will there be an IND exemption letter from FDA?
Will there be any
BJH/Children’s (or other non-WU) employees or services involved in any aspect of this study?
(ex. OR, labs, pharmacy, technicians, nurses, diagnostics, or in-patient procedures) YES NO
Will your research be supported or conducted at any facility other than BJH or SLCH? If so which one?
If Yes was answered to either of the prior 2 questions, please explain involvement :
AGREEMENT
PUBLICATION
–Indicate the total length of time you will allow the sponsor to review your publication:
30 – 60 days 60 – 90 days Sponsor-determined time frame ok
If the study is multi-center , what is an acceptable delay before publishing? < 12 mos. 12 - 18 mos.
INTELLECTUAL PROPERTY
– Do you anticipate that you will make an independent discovery or invention (other than the
Sponsor’s anticipated result) related to the study you are performing, or do you expect to make an improvement to or develop a new use for the Sponsor’s drug or product? (A new use is a use beyond the Sponsor’s existing patent and/or FDA approved
treatment with the study drug) Yes No
RELATED RESEARCH
– Are you doing any related research for another party including another private company or the NIH or
PHS that could conflict with this study? Yes No
DISCLOSURE
– Do you* or any investigator* participating in the study have a financial interest in the sponsor consisting of:
Consulting, Speaking fees, Serving on a BOD or SAB, Honoraria, Personal Gifts,
Licensing agreement or royalty income, Equity interests, including stock, stock options, warrants, partnership or equitable ownership interests, or Other fees/compensation *includes family member(s).
CONFIDENTIALITY
–Indicate time period after study you are willing to keep sponsor information confidential:
3
– 5 years
5
– 7 years
more than 7 years (requires additional approval)
BUDGET
–
University overhead for corporate sponsored clinical studies is now 26% effective July 1, 2006. The Budget should include the appropriate University overhead, any departmental overhead, and IRB fees. ( Please note that Department Business
Offices require review and approval for your budget prior to submission to Sponsor. Please involve them early to avoid delays.) .
Please forward documentation of Dept approval of the budget, which is required for CCS files.
Is the budget provided by the Sponsor acceptable to you? Yes No Pending
Is the payment schedule provided by the Sponsor acceptable to you? Yes No Pending
CCS SERVICES
Are you requesting CCS services of this study or IRB submission or other coordinator services? YES NO
(If yes, complete Request for CCS Services Form as found on the CCS web site: ccs.wustl.edu)
PRINCIPAL INVESTIGA TOR’S SIGNATURE : DATE:
FORWARD signed Expeditor, all originals of Agreement, along with the Informed Consent and the Protocol Synopsis (if the Protocol
Synopsis is not available, send the Study Rationale and the Study Objectives) to the Center for Clinical Studies e-mail: ccscontracts@msnotes.wustl.edu
. The electronic version of the above documents is preferred.
Signed originals can be sent to: CENTER FOR CLINICAL STUDIES, IWJ ROOM 301, CAMPUS BOX 8009 FAX: 747-1404.
For more information about CCS services, budgets, or contract terms, visit our web site: http://ccs.wustl.edu
Rev. 11 10.31.07