Fission and fusion – practice questions Question 250S: Short
advertisement

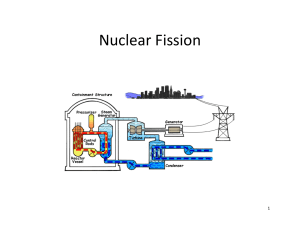
Fission and fusion – practice questions Question 250S: Short Answer What these are for: These questions will give you some simple practice in handling the ideas and calculations that physicists meet in nuclear fission and fusion. The process of fission in one type of nuclear reactor proceeds as follows: a nucleus of uranium 235 92 U captures a single neutron. The resulting nucleus is unstable and splits into two or more fragments. These fragments could typically be a pair of nuclei, 90 36 Kr and 144 56 Ba for example. Neutrons are also ejected as a result of the fission. It is these neutrons that go on to cause subsequent fission events and maintain the chain reaction. 1. Write down two balanced equations (the first to the unstable uranium; the second to the final products) that represent this fission process. 2. Calculate the total mass of the original uranium isotope and the neutron. The table gives the atomic masses (in atomic mass units) of the particles found in this question. (1 atomic mass unit (u) º 931 MeV.) Particle n Mass (u) 1.008 665 Kr 89.919 528 Rb 95.934 284 1 0 90 36 96 37 138 55 Cs 137.911 011 144 56 Ba 143.922 941 235 92 235.043 923 U 3. Calculate the total mass of the four products. 4. Calculate the change in mass. Does this represent energy gained or lost by the system? 5. Convert the mass change into the energy released (in MeV) in the fission event. 6. These particular barium and krypton isotopes are not the only products possible in nuclear fission. Repeat the calculation steps 1–5 with the following possible products: Caesium-138 and Rubidium-96. Nuclear fusion is the process in which nuclei combine to give heavier elements. In one fusion reaction, two atoms of deuterium (hydrogen-2) fuse together to give one atom of a helium isotope (helium-3) together with one other particle. 7. Write out a balanced equation for this fusion process and say what the fourth particle is. 8. Calculate the energy release in this equation. Values you need are in the table. Particle n Mass (u) 1.008 665 H 2.014 102 H 3.016 050 He 3.016 030 1 0 2 1 3 1 3 2 Another possible fusion process is represented by: 1H 0 n 1H 2 1 3 (the formation of hydrogen-3, tritium, by a nucleus of deuterium absorbing a neutron). This equation is certainly balanced. But can it occur in practice? 9 Calculate the change in mass in this reaction. 10. Is the reaction possible or not? Fusion in a kettle? Question 260S: Short Answer A change of scale When you are confident with basic calculations of fission and fusion energy changes, you should work through these questions that try to put the energies of these changes into a more human scale for you. You will also need to understand the conversion of atomic mass units to energy and the meaning of the term ‘electron volt’. Try these One of the reactions that fuels the stars is the fusion of two protons to give deuterium. In turn the deuterium goes through a series of reactions, the end product being helium. This is also a process that releases energy. In this question you are asked to consider the energy that would be released if all the deuterium in the water contained in an electric kettle were to be converted by fusion into helium. The kettle contains 1 litre of water. The data you need are listed below. 1 atomic mass unit (u) = 931 MeV 1 eV = 1.6 x 10–19 J NA = 6.02 × 1023 mol–1 Particle Mass / u 1 1.007 825 H 1 2 1 3 2 1 0 H 2.014 102 He 3.016 030 n 1.008 665 2 1. Two deuterium nuclei 1H can fuse to give one nucleus of helium Write down the balanced equation that represents this reaction. 2. Calculate the mass change that occurs in this reaction. 3. Convert this energy into joules. 3 2 He with the ejection of one other particle. This gives you the energy released when two deuterium nuclei fuse. The next steps take you through an estimate of the total energy released if all the deuterium in the kettle water were to fuse to make helium-3. The ratio of deuterium atoms to hydrogen in water is roughly 1 to 7000. 4. What roughly is the mass of 1 mole of water? (H = 1 u; O = 16 u. Ignore the effect of the small number of water molecules containing deuterium.) 5. How many moles of water are contained in the litre? 6. How many molecules of water (H2O) are in the kettle? 7. How many molecules of deuterium oxide (D2O) are in the kettle? 8. Each heavy water molecule has two atoms of deuterium; what total energy is released if all the deuterium in the kettle is converted to helium-3? Now to put this number in a new perspective. It requires 4200 J to increase the temperature of 1 kg of water by 1 K. 9. How many litres of water could be heated through 100 K by the fusion energy you calculated in question 8? Fission in a nuclear reactor – how the mass changes Question 270S: Short Answer Some rather harder questions - These extended questions will test your ability to deal with calculations involving the physics of nuclear fission. Use the following conversions and values for some of the questions: · 1 eV = 1.60 x 10–19 J · 1 atomic mass unit = 1.66 x 10–27 kg · c = 3.00 x 108 m s–1 Particle Mass (u) 235 235.043 94 U 92 H 1.007 825 He 3.016 030 n 1.008 665 1 1 3 2 1 0 Try these In the 1970s, Magnox power stations produced about 20 TW h of electrical energy in the UK every year by fission of uranium. (This energy supplied roughly the electrical needs of Greater London.) 1. The overall efficiency of the process that converts the energy for heating released in the fission to the final electrical product is 40%. How much energy, in joules, is produced each second in the company’s reactors? 2. Each fission releases about 200 MeV of energy. How many atoms of 235 92 U need to fission in each second to produce the heating energy you calculated in question 1? 3. What was the mass of these atoms before they underwent fission? 4. What is the total mass change due to fission in Magnox reactors each second? In the Magnox reactor, the fuel rods do not contain pure 238 92 U and 235 92 235 92 U . The uranium comes from mined ore that contains a mixture of U . The fuel delivered to the reactor contains 0.7% of 235 92 U . The fuel rod stays in the reactor for about 3 years and is then removed to allow reprocessing. This time consider an electrical output of 1 GW from a set of Magnox reactors. 5. Calculate the number of uranium nuclei disintegrating every second. 6. Calculate the mass of 235 92 7. Estimate the mass of 235 92 8. Estimate the total mass of both uranium isotopes required in the core for a 3 year cycle. 9. Is your estimate in question 8 likely to be an upper or a lower limit? U that undergoes fission every second. U required in the core for a 3 year cycle.