Abstract
advertisement

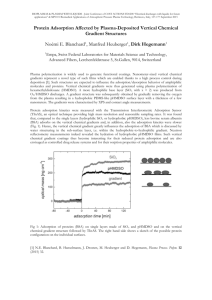
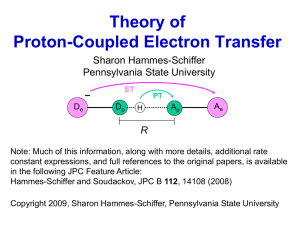
Adsorption and Charge-Transfer Processes on Gold Electrode Surfaces This talk will discuss two aspects of electrochemical research performed in the Burgess lab. In part 1, an example of electric-field driven adsorption (without formal electron transfer) will be described. Dimethylaminopyridine (DMAP) is one of the few examples of a ligand that yields waterdispersible, monolayer protected nanoparticles (MNPs). However, until our studies, very little was known about the adsorption behaviour of DMAP on gold. We have used electrochemical and in-situ spectroscopic techniques to measure the adsorption of DMAP and its conjugate acid (DMAPH+) on polycrystalline gold surfaces. Our results indicate that DMAP adsorption is both quantitatively and qualitatively dependent on solution pH. By coupling our electrochemical results with transmission electron microscopy (TEM) and UV-Vis spectroscopy we have been able to explain the pH dependent and solution-composition dependent stability of DMAP MPNs. Part 2 will focus on a redox-active (quinone) monolayer that undergoes proton-coupled electron transfer (PCET). Theoretical treatments of PCET indicate that both thermodynamic (apparent formal potential) and kinetic (apparent standard heterogeneous rate constants) quantities should be highly pH dependent. Although there are several reports of quinone derivatized self-assembled monolayers (SAMs) in the literature, no systematic studies on the pH dependence of the PCET kinetics have been reported. In this work, we outline a method to produce SAM-aminobenzoquinone monolayers starting from diluted monolayers of an amine terminated SAM. The electroactive monolayer behaves as a nearly ideal Nernstian system (no interactions between redox centres). The ideality of our system has allowed us to perform both thermodynamic and kinetic measurements of PCET and compare them to existing theoretical models.