Group 3 Carbon Cycle Notes

Key Notes
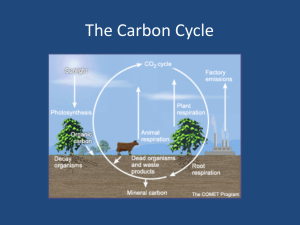
· Carbon can be found in the atmosphere and in the oceans, dissolved, as part of the inorganic carbon molecule
· Carbon from inorganic CO2 is cycled into organic substances by the process of photosynthesis, after in which some of the carbon is released back into the atmosphere through cellular respiration as CO2
· Photosynthesis and Cellular respiration are complementary processes
· Carbon is repeatedly cycled through both, the relationship is called the carbon cycle
· Most carbon is returned to the atmosphere or water as CO2 from body waste and decaying organisms
·
Only in certain conditions decay is delayed and the organic matter can be turned into rock or fossil fuels
· After that production, the carbon is then unavailable to the cycle until it is burned as fuels or by other processes such as uplifting and weathering
·
Combustion is the process by which the fossil fuels are burned and releases the
CO2 back into the atmosphere
· Three main reservoirs: - Atmosphere (smallest), Oceans, Earth’s Crust
· Carbon Dioxide makes up 0.03% of the gasses we breath
· There is a high amount of atmospheric CO2 for the plants to use for photosynthesis
·
The most amount of inorganic CO2 is held as dissolved CO2 in the oceans which is used by the algae and water plants for photosynthesis
·
CO2 reacting with sea water forms the inorganic carbonate ion (CO3-2) and bicarbonate ion (HCO3-). When added with calcium -- CaCO3 it enables the creation of hard structures within the ocean (shells)
·
The remaining carbon ends up as sediment and if heated and crushed will form rocks
·
Discarded shells and bones create limestone, which makes sedimentary rocks the largest carbon reservoir
· Acid rain falling on exposed limestone will release CO2
· The oxygen cycle and the carbon cycle are closely linked as well because oxygen is important in photosynthesis and in cellular respiration
·
Most biotic things require O2 for cellular respiration and is key in the decomposition process
· Oxygen Cycle: the movement of oxygen gas (02) from biotic things, into the atmosphere through photosynthesis, then back into biotic things through cellular respiration.
·
O2 can be found in all parts of the biosphere and is key in many reactions. Oxygen atoms are vital substances such as: Carbon Dioxide, water, glucose.
· CELLULAR RESPIRATION (Converting chemical energy to ATP)
C6H12O6 (aq) +
6 O2 (g) → 6 CO2 (g) + 6 H2O (l) + energy
· PHOTOSYNTHESIS (Converting energy to glucose and oxygen)
6 CO2 (g) + 6
H2O + energy → C6H12O6 (glucose)
+ 6 O2
·
Organic carbon is held in the bodies of living things
·
Carbon is released back into the carbon cycle when the things die as inorganic carbon
· Bogs and other ecosystems similar are the exception to this rule . They store huge quantities of organic carbon because of the lack of oxygen making decomposition very slow
·
Carbon can remain locked in away in the dead plant matter (peat) for years. These deposits can become covered with sediment As the sediment piles up, the organic plant matter will become trapped resulting in carbon containing fossil fuels
· Carbon Cycle: the cycle of the matter in which carbon atoms move from an inorganic form to an organic form then back to an organic form
·
Combustion: the chemical reaction that occurs when a substance reacts very quickly with oxygen to release energy
· Peat: slowly decomposing plant matter produced in low-oxygen environments such as bogs