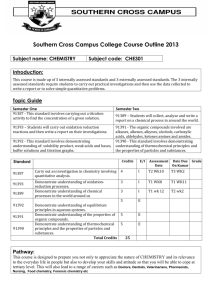
Chemistry T - ACT Board of Senior Secondary Studies
advertisement

Board Endorsed October 2006 – Amended July 2016 Chemistry T Course Type 2 Written under the Accredited from: Science Course 1 January 2007 – 31 December 2011 Framework 2006 Extended 2016 Amended July 2013 (Includes Assessment Task Types approved August 2013) Version 2 Board Endorsed October 2006 – Amended December 2013 Student Capabilities All programs of study for the ACT Year 12 Certificate should enable students to become: creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members and provide students with: a comprehensive body of specific knowledge, principles and concepts a basis for self-directed and lifelong learning personal attributes enabling effective participation in society Approved August 2005 Board Meeting Version 2 1 Board Endorsed October 2006 – Amended December 2013 Type 2 Course Accreditation/Adoption Form B S S S AUSTRALIAN CAPITAL TERRITORY Choose one of the following: adoption of Type 2 course small changes from Written Evaluation of Type 2 course extension of Type 2 course or units modification of Type 2 course (M course) adoption of additional units College: Course Title: Chemistry Course Code Classification: A T M V Unit Title(s) Value (1.0/0.5) Length Moles, Water, Reactions & Acids and Redox 1.0 S Introductory Chemistry 1.0 S Introduction to Chemistry 0.5 Q The Mole, Water & Reactions in Solution 0.5 Q Acids, Redox & Organic Chemistry 1.0 S Acids and Redox 0.5 Q Organic Chemistry 0.5 Q Physical Chemistry 1.0 S Biochemistry 1.0 S Biochemical Structures & Functions 0.5 Q Biochemical Processes & Applications 0.5 Q Industrial Chemistry 1.0 S Foundations of Industrial Chemistry 0.5 Q Investigation of Industrial Processes 0.5 Q Environmental Chemistry 1.0 S Principles of Environmental Chemistry 0.5 Q Investigation of Environmental Incidents 0.5 Q Analytical Chemistry 1.0 S Introduction to Chemical Analysis 0.5 Q Advanced Analytical Processes 0.5 Q Forensic Chemistry 1.0 S Principles & Processes of Forensics 0.5 Q Electrochemistry 1.0 S Extended Research 0.5 Q Dates of Course Accreditation: Version 2 From 1 / 1 / 2007 To Unit Codes 31 / 12 / 2016 2 Board Endorsed October 2006 – Amended December 2013 Extended 2015 Accreditation: The course and units named above are consistent with the goals of the Course Framework and are signed on behalf of the BSSS. Course Development Coordinator: Panel Chair: / Version 2 / / / 3 Board Endorsed October 2006 – Amended July 2016 Course Accreditation/Adoption Supporting Statement Provides support for information on the Course Accreditation/Adoption Form B S S S AUSTRALIAN CAPITAL TERRITORY Written Evaluation for Small changes, reasons for Modification or Adoption of a Type 2 course, or Addition of units to a Type 2 course. For V courses indicate the certificate the college will award. College: Course Code Course Title: Course Length and Composition Number and Length of Units Which units will your college deliver? Duration of Units and Available Course Patterns Must be consistent with Table 1.1 in the Guidelines. Implementation Guidelines Must be consistent with the original course document. Compulsory Units Must remain the same as original document. Prerequisites for the course or units within the course Must remain the same as original document. Arrangements for students who are continuing to study a course in this subject The adopting college may customize this to suit their individual needs. Additional Units The adopting college may write additional units to suit their individual needs but within policy 2.5.5.6 and with panel approval. The course should have coherence between units of study (Policy 2.4.2). Interdisciplinary Units If the adopting college wishes to include a unit from another course see Policy 2.5.12.6. Negotiated Units The adopting college may customize this to suit their individual needs but within policy 2.5.5.6 and with panel approval. Refer to Board Policy 2.5.5.4 if considering including Independent Study units within this course. Suggested Implementation Patterns This must be in line with the original course document. Please indicate any specific needs for your college when adopting this course. For example – if you intend to deliver the course in any delivery time structure other than the way it has been written (i.e. 1.0 units instead of 0.5 units) then these must be submitted with this adoption form. Version 2 4 Board Endorsed October 2006 – Amended July 2016 Contents Course Classification .....................................................................................6 Course Framework .....................................................................................6 Course Developers .....................................................................................6 Evaluation of Previous Course .....................................................................................7 Course Length and Composition .....................................................................................8 Available Course Patterns .....................................................................................9 Implementation Guidelines .....................................................................................9 Subject Rationale ...................................................................................11 Goals ...................................................................................11 Student Group ...................................................................................11 Content ...................................................................................12 Teaching and Learning Strategies ...................................................................................12 Assessment ...................................................................................13 Science Unit Grade Descriptors for T courses ...................................................................................15 Moderation ...................................................................................16 Bibliography ...................................................................................17 Resources ...................................................................................20 Proposed Evaluation Procedures ...................................................................................20 Introductory Chemistry Value: 1.0...................................................................21 Introduction to Chemistry Value: 0.5...................................................................25 Moles, Water, Reactions & Acids and Redox Value: 1.0...................................................................29 The Mole, Water & Reactions in Solution Value: 0.5...................................................................33 Acids, Redox and Organic Chemistry Value: 1.0...................................................................36 Acids and Redox Value: 0.5...................................................................40 Organic Chemistry Value: 0.5...................................................................43 Physical Chemistry Value: 1.0...................................................................46 Biochemistry Value: 1.0...................................................................49 Biochemical Structures and Functions Value: 0.5...................................................................52 Biochemical Processes & Applications Value: 0.5...................................................................54 Industrial Chemistry Value: 1.0...................................................................57 Foundations of Industrial Chemistry Value: 0.5...................................................................59 Investigation of Industrial Processes Value: 0.5...................................................................61 Environmental Chemistry Value: 1.0...................................................................63 Principles of Environmental Chemistry Value: 0.5...................................................................66 Investigation of Environmental Incidents Value: 0.5...................................................................69 Analytical Chemistry Value: 1.0...................................................................71 Introduction to Chemical Analysis Value: 0.5...................................................................74 Advanced Analytical Processes Value: 0.5...................................................................76 Forensic Chemistry Value: 1.0...................................................................78 Principles & Processes of Forensics Value: 0.5...................................................................82 Electrochemistry Value 0.5....................................................................86 Extended Research Value: 0.5...................................................................88 Biochemical Structures & Analysis Value: 1.0...................................................................90 Appendix A - Australian Curriculum Achievement Standards ................................................................93 Checklist for courses submitted for accreditation .................................... Error! Bookmark not defined. Chemistry (T) Unit Student Evaluation ...................................... Error! Bookmark not defined. Version 2 5 Board Endorsed October 2006 – Amended July 2016 Course Name Chemistry Course Classification T Course Framework This course is presented under the 2005 Science Course Framework. Course Developers Name Qualifications College Stephen FORD BEd, Grad Cert Canberra Lake Ginninderra Bill ZIMMERMANN BSc(Hons),DipEd. UNSW Hawker Rosemary WATERS Dip Teaching Sydney Hawker Leslie BARNARD BAppSc,GradDip Canberra Lake Ginninderra Lynne BEAN BSc. DipEd Sydney, GradDipSc ANU Dickson Sarah SUKUMAR BAppSc RMIT DipEd Tasmania Canberra Peter SMITH PhD, Sydney DipEd Canberra Erindale Stephanie MANDER PhD Sydney, DipEd Canberra. CIT Sue STURGISS BSc (Hons) Sydney,DipEd CSU,GradCertEd ACU St Francis Xavier Jan CHEETHAM BSc.DipEd. Flinders St Clare’s Jeannette JOLLEY BSc.DipEd. BEd, Latrobe St Clare’s This group gratefully acknowledges the work of previous course developers. Version 2 6 Board Endorsed October 2006 – Amended July 2016 Evaluation of Previous Course This new course is largely based on very successful Chemistry courses from the majority of colleges in the ACT with minor adjustments to cater for all participating contributors. This is the first Type 2 Chemistry course to be written in the ACT. Are the course and course framework still consistent? The proposed Type 2 Course is consistent with the Science Framework, which has just been written. Were the goals achieved? The Type 1 Course this document is based on all received very positive comments on both their course contents and their assessment procedures on moderation days. Was the course content appropriate? The entire course contents had been approved by the Chemistry Accreditation Panel. How successful were the teaching strategies? Feedback from students, who had gone on to tertiary studies invariably indicated that they felt especially well-prepared, more so than their NSW counterparts, for studies in Chemistry at the ANU, UC, UNSW, ADFA, CSU and other universities. Comments from staff at ANU, ADFA and UC were also very positive and have been for very many years. Competency in practical skills was continually referred to. Occasionally, well-qualified parents of students have indicated their high approval of the courses and their delivery. How appropriate was the assessment program? Moderation Day feedback indicate that the assessment was appropriate and we have passed on a number of our assessment items as exemplars to colleagues across the system. Were the needs of all students met? We believe the needs of all students were met. Across the system only one or two students per year were “V-graded” in Chemistry and, as mentioned above, feedback from past students has been very positive. Was the course relevant? Feedback from students as mentioned above indicate that this was the case, as were comments from academic staff at ANU, ADFA and UC. How many students completed the course in each of the years of accreditation? This question is not applicable as this is a new course. What improvements need to be made to the course? Minor improvements and/or changes are needed to: provide some more flexibility so that this course can be adopted by as many colleges as possible in part by writing more options in Semester 4; allow for some changes since the last course was written, especially Biochemistry; broaden options for study in Industrial Chemistry; acknowledge a change in the Chemistry experiences of students entering the course. Version 2 7 Board Endorsed October 2006 – Amended July 2016 Course Length and Composition Unit Title Unit Value INTRODUCTORY CHEMISTRY 1.0 INTRODUCTION TO CHEMISTRY 0.5 THE MOLE, WATER & REACTIONS IN SOLUTION 0.5 ACIDS, REDOX & ORGANIC CHEMISTRY 1.0 ACIDS and REDOX 0.5 ORGANIC CHEMISTRY 0.5 PHYSICAL CHEMISTRY 1.0 Optional Units BIOCHEMISTRY 1.0 BIOCHEMICAL STRUCTURES & FUNCTIONS 0.5 BIOCHEMICAL PROCESSES & APPLICATIONS 0.5 INDUSTRIAL CHEMISTRY 1.0 FOUNDATIONS OF INDUSTRIAL CHEMISTRY 0.5 INVESTIGATION OF INDUSTRIAL PROCESSES 0.5 ENVIRONMENTAL CHEMISTRY 1.0 PRINCIPLES OF ENVIRONMENTAL CHEMISTRY 0.5 INVESTIGATION OF ENVIRONMENTAL INCIDENTS 0.5 ANALYTICAL CHEMISTRY 1.0 INTRODUCTION TO CHEMICAL ANALYSIS 0.5 ADVANCED ANALYTICAL PROCESSES 0.5 FORENSIC CHEMISTRY 1.0 PRINCIPAL & PROCESSES OF FORENSICS 0.5 ELECTROCHEMISTRY 0.5 EXTENDED RESEARCH 0.5 Any school wishing to offer 0.5 units in third semester,Physical Chemistry, will need to adjust the 1.0 unit into a split appropriate to their delivery of the unit. Version 2 8 Board Endorsed October 2006 – Amended July 2016 Available Course Patterns Course Number of standard units to meet course requirements Minor Minimum of 2 units Major Minimum of 3.5 units Major Minor Minimum of 5.5 units Double Major Minimum of 7 units Implementation Guidelines Compulsory units For a Minor in Chemistry, (2 units), Introductory Chemistry and Acids, Redox & Organic Chemistry are required. For a Major in Chemistry, (3.5 or 4 units), Introductory Chemistry, Acids, Redox & Organic Chemistry, Physical Chemistry and at least one half unit from the Chemistry Options are required. For a Major Minor in Chemistry, (5.5 or 6 units) Introductory Chemistry, Acids, Redox & Organic Chemistry, Physical Chemistry and at least two and one half units from the Chemistry Options are required. Alternatively, a student may undertake a Minor in Chemistry at the ANU College, together with a Major in their home college to complete a Major Minor. For a Double Major in Chemistry, (7.0 or 7.5 units) Introductory Chemistry, Acids, Redox & Organic Chemistry, Physical Chemistry and at least four units from the Chemistry Options are required. Alternatively, a student may undertake a Minor in Chemistry at the ANU College, together with a Major in their home college plus an additional one and one half or two units to complete a Double Major. Prerequisites for the course or units within the course Introductory Chemistry would normally be a prerequisite for Acids, Redox & Organic Chemistry and Acids, Redox & Organic Chemistry would normally be a prerequisite for Physical Chemistry. In exceptional circumstances, and at the discretion of the Executive Teacher responsible for Science, this could be varied. Options in Semester 4 would require Physical Chemistry as a prerequisite. An exception to this would be Environmental Chemistry (which could be done concurrently with Acids, Redox & Organic Chemistry). For those students wishing to include Atomic & Nuclear Physics as a Chemistry unit, a minor in Physics is a prerequisite. Forensic Chemistry is an alternative unit to Analytical Chemistry, and Electrochemistry is an alternative to Industrial Chemistry as there is some overlap of content in these units. In addition, it is recommended that students would be enrolled in a Tertiary Mathematics course. Version 2 9 Board Endorsed October 2006 – Amended July 2016 Arrangements for students who are continuing to study a course in this subject Students continuing this course from Year 11 may take the following units in Year 12: Physical Chemistry followed by at least a 0.5 unit of one of the Chemistry Options. Some Colleges may need to apply separately for an extension of their present course for Year 12 students in 2007 to avoid duplication of content. Units from other courses Students may elect to undertake a minor in Chemistry at the ANU, concurrently with a major in their home college to achieve a Major Minor or as part of a Double Major as detailed above. Negotiated Units Students may, at the discretion/invitation of their chemistry teacher, elect to undertake a “Case Study” or Extended Research. It is envisaged that this would only be appropriate for a few exceptional students each year. This could be undertaken as a half or whole unit in an area of Chemistry of the student’s own choosing, in consultation with his or her teacher, who would act as a mentor. Relationship with other courses Chemistry is one of a number of Science Courses which all come under the Science Framework. Biochemistry may in some instances be counted as a Biology unit or a Chemistry Unit. Atomic & Nuclear Physics may in some instances, be counted as a Physics or Chemistry Unit. Study at ANU College, as previously noted, may contribute the equivalent of a Minor in Chemistry to either a Major Minor or a Double Major. Other colleges may offer units that students could undertake as part of the Chemistry Course. Relationship with courses at other colleges/special institutions (optional) Study at ANU College, as previously noted, may contribute the equivalent of a Minor in Chemistry to either a Major Minor or a Double Major. Other colleges may offer units which students could undertake as part of the Chemistry Course. Some units could be taught at a particular College to a student cohort from a combination of Colleges, timetabling permitting. The CIT are planning to adopt this Type 2 course as part of its Year 12 program. Successful completion with a Chemistry Major and at least a C grade average would give credit transfer for the CIT Diploma unit Introductory Chemistry, a bridging or first year unit in all CIT science diplomas and advanced diplomas. Suggested Implementation Pattern or Sequencing of Units Implementation Pattern Units Involved Year 11, Semester 1 Introductory Chemistry Year 11, Semester 2 Acids, Redox & Organic Chemistry Year 12, Semester 1 Physical Chemistry Year 11 Semester 2, concurrent with Acids, Redox & Organic Chemistry Environmental Chemistry Year 12 Semester 1, concurrent with Physical Chemistry Forensic Chemistry Year 12, Semester 2 Any 0.5 or 1.0 units from Chemistry Options or Atomic & Nuclear Physics The compulsory units outlined ensure that all students undertaking a T minor course will achieve the goals of the course. Version 2 10 Board Endorsed October 2006 – Amended July 2016 Subject Rationale There is an innate human curiosity about and wish to understand the universe. The study of science encourages and enables students to develop complex and sophisticated understanding of the universe through observation, questioning, experimentation, discussion, critical analysis and creative thinking. In a technologically based society, scientific literacy for all citizens is of paramount importance. The Program for International Student Assessment (PISA) defines scientific literacy as “the capacity to use scientific knowledge, to identify questions and to draw evidence-based conclusions in order to understand the natural world and the changes made to it through human history.” (OECD: the 2003 PISA Assessment Framework). Scientifically literate individuals contribute to the quality of their own lives and to society through informed decision-making. Scientific processes challenge current understanding and are continually re-evaluated. Students are constantly encouraged, through their study of science, to examine and reconsider their understanding of scientific concepts and their interrelationships, of scientific inquiry methods and therefore of their world more generally. Science courses also help students understand and apply their learning in other subjects, in a scientific way. The essence of science involves social, cultural, critical and aesthetic perspectives. An appreciation of the aesthetic qualities and understanding of the universe strengthens students’ curiosity and sense of wonder. The study of science equips students to be independent thinkers and life-long learners. Goals This course should enable students to: demonstrate depth and breadth of scientific knowledge; apply knowledge and understanding to solve problems in familiar and unfamiliar contexts; critically research, analyse, evaluate and synthesise information from a variety of sources, including their own work and the work of their peers; develop hypotheses and design, carry out and as necessary modify experiments; follow instructions and make accurate and precise observations while conducting practical investigations, while safely using appropriate equipment and techniques; communicate scientific information to diverse audiences in an appropriate manner using a variety of media and technologies; appreciate the role and implications of science in the wider community – environmental, social, political and economic; work independently and collaboratively. Student Group Chemistry attracts a significant number of students as it is seen as the enabling science. Many of the more able students continue with the Year 12 units if they aspire to studying any of the Science based degree courses at a tertiary level. Other students find it is more satisfactory to complete a minor in Chemistry. Version 2 11 Board Endorsed October 2006 – Amended July 2016 Content In addition to the essential concepts and skills outlined in the Framework, all Chemistry major courses should cover: Periodicity Atomic structure Stoichiometry Chemical change Solubility Organic chemistry Rates of reaction Equilibrium Bonding and structure Types of reactions Acids and bases Electrochemistry Energy The course is structured to facilitate the inclusion of the historical, biological, physical, environmental, analytical and industrial aspects of chemistry. Teaching and Learning Strategies Teaching strategies that are particularly relevant and effective in the teaching of Chemistry include: practical / field work / excursions; inquiry-based learning; collaborative learning; open-ended investigations; visiting scientists; modelling; use of information and communication technologies (ICT), including data loggers, CD ROMs, Videos and the Internet; peer tutoring / student presentations / student as teacher; integration of teacher-student and student-student feedback; teacher instruction – lectures, discussions, skills instruction; teacher demonstrations; student reflection on their learning. These strategies are consistent with the Learning Principles (see introduction in Science Course Framework). Version 2 12 Board Endorsed October 2006 – Amended July 2016 Assessment Assessment Tasks and Assessment Criteria The purpose of including assessment task types (with examples of tasks) and assessment criteria in Course Frameworks is to provide a common and agreed basis for the collection of evidence of student achievement. This collection of evidence enables a comparison of achievement within and across colleges, through moderation processes. This enables valid, fair and equitable reporting of student achievement on the Year 12 Certificate. Assessment tasks elicit responses that demonstrate the degree to which students have achieved the goals of a unit (and the course as a whole). Assessment Task Types (with weightings) group assessment tasks in ways that reflect agreed shared practice in the subject area and facilitate the comparison of student work across different assessment tasks. Assessment Criteria (the qualities that teachers look for in evaluating student work) provide a common and agreed basis for judgement of performance against unit and course goals, within and across colleges. Over a course, teachers use all of these criteria to assess students’ performance, but do not necessarily use all criteria on each task. Assessment criteria are to be used holistically on a given task and in determining the unit grade Assessment Rubrics draw on the general course framework criteria to develop assessment criteria for a task type and a continuum which indicates levels of student performance against each criterion. Version 2 13 Board Endorsed October 2006 – Amended July 2016 Assessment Task Types Assessment for T Courses Suggested task types: Strands Inquiry skills Human endeavour Understanding log book * practical report * research assignment * presentations * investigative project essay * models * test/quizzes * practical skills test * Weighting for 1.0 and 0.5 units Weighting for Project based units 40-60% 40-100% 40-60% 0-60% Key: This table is designed to highlight types of tasks which address different content descriptors and assessment criteria. Teachers are reminded that any single task can incorporate multiple assessment strands. highly relevant - These tasks will have a clear link to the content descriptors and assessment strands. * some relevance - These tasks have some links to the content descriptors and assessment strands. Additional Assessment Advice for T Courses For a standard 1.0 unit, a minimum of three and a maximum of five assessment items. For a half-standard 0.5 unit, minimum of two and a maximum of three assessment items. Each unit (standard 1.0 or half standard 0.5) should include at least 2 different types of tasks. It is recommended that, in standard units, no assessment item should carry a weighting of less than 10% or greater than 45% of the unit assessment. A variety of task types and modes of presentations should be used during the course. It is recommended that an open-ended investigation be undertaken at least once during a minor and twice during a major. This investigation may either be theoretical or practical or a combination of both. Assessment Criteria Students will be assessed on the degree to which they demonstrate: knowledge and understanding; critical thinking; investigative skills; communication skills; effective work practices. Version 2 14 Board Endorsed October 2006 – Amended July 2016 Science Unit Grade Descriptors for T courses Work practices Communica tion Investigative Skills Critical Thinking Knowledge and Understanding A student who achieves an A grade typically A student who achieves a B grade typically A student who achieves a C grade typically A student who achieves a D grade typically A student who achieves an E grade typically demonstrates a limited knowledge of scientific concepts demonstrates thorough and extensive knowledge and understanding of scientific concepts demonstrates broad and in-depth knowledge and understanding of scientific concepts demonstrates broad and general knowledge and understanding of scientific concepts demonstrates general and basic knowledge and understanding of scientific concepts justifies and applies knowledge to familiar and unfamiliar contexts and across different concept areas and experiences, displays originality and lateral thinking in problem solving applies knowledge to familiar and unfamiliar contexts and across different concept areas and experiences, displaying originality and effective thinking in problem solving is able to apply knowledge in a variety of contexts and different concept areas to solve problems is able to use knowledge in different areas to solve problems displays emerging awareness of strategies to solve problems evaluates, synthesises and analyses patterns and trends in data, observations and investigations and makes valid and perceptive inferences analyses and synthesises patterns and trends in data, observations and investigations and makes valid inferences describes and explains patterns and trends in data, observations and investigations and makes general inferences identifies and describes patterns in data, observations and investigations and makes simple inferences identifies patterns in data, observations and investigations applies highly effective analytical and evaluative skills, makes perceptive connections between scientific concepts, draws accurate conclusions and proposes appropriate improvements applies effective analytical skills, makes insightful connections between scientific concepts, draws mostly accurate conclusions and proposes appropriate improvements describes and explains general connections between scientific concepts, draws conclusions and proposes improvements describes connections between scientific concepts, draws conclusions and proposes improvements identifies connections between scientific concepts demonstrates logical and coherent investigations, acknowledges information using referencing conventions and operates equipment highly effectively and safely demonstrates well considered investigations, acknowledges information using referencing conventions and operates equipment effectively and safely demonstrates considered investigations, acknowledges information using referencing conventions and operates equipment safely with some general effectiveness outlines investigations, inconsistently acknowledges information using referencing conventions and mostly operates equipment effectively and safely displays emerging skills in investigations, attempts to acknowledge information and operates equipment with limited awareness of safety procedures presents highly complex concepts accurately and coherently in a wide range of written and non written formats using appropriate terminology with flair presents concepts clearly and logically in a range of written and non written formats using appropriate terminology with confidence presents general concepts clearly in a range of written and non written formats using appropriate terminology generally using terminology appropriately presents basic concepts in a narrow range of written and non written formats using terminology inconsistently presents some basic concepts in a limited range of written & non written formats using minimal terminology organises time and resources to work in a highly productive and safe manner both independently and in a team organises time and resources to work in a productive and safe manner both independently and in a team organises time and resources to work in a generally productive and safe manner both independently and in a team evaluates and analyses risks, acts highly appropriately in all investigations analyses and explains risks and acts appropriately in all investigations identifies and describes risks and acts appropriately in all investigations demonstrates inconsistent organisation of time & resources, works with occasional productivity & some awareness of safety independently or in a group identifies risks and acts mostly appropriately in investigations demonstrates limited organisation of time & resources to work with an emerging awareness of safety demonstrates an emerging awareness of risks, developing approaches to investigations Version 2 15 Board Endorsed October 2006 – Amended December 2013 Moderation Moderation is a system designed and implemented to: provide comparability in the system of school-based assessment; form the basis for valid and reliable assessment in senior secondary schools; involve the ACT Board of Senior Secondary Studies and colleges in cooperation and partnership; and maintain the quality of school-based assessment and the credibility, validity and acceptability of Board certificates. Moderation commences within individual colleges. Teachers develop assessment programs and instruments, apply assessment criteria, and allocate Unit Grades, according to the relevant Course Framework. Teachers within course teaching groups conduct consensus discussions to moderate marking or grading of individual assessment instruments and unit grade decisions. The Moderation Model Moderation within the ACT encompasses structured, consensus-based peer review of Unit Grades for all accredited courses, as well as statistical moderation of course scores, including small group procedures, for T courses. Moderation by Structured, Consensus-based Peer Review Review is a subcategory of moderation, comprising the review of standards and the validation of Unit Grades. In the review process, Unit Grades, determined for Year 11 and Year 12 student assessment portfolios that have been assessed in schools by teachers under accredited courses, are moderated by peer review against system wide criteria and standards. This is done by matching student performance with the criteria and standards outlined in the unit grade descriptors as stated in the Course Framework. Advice is then given to colleges to assist teachers with, and/or reassure them on, their judgments. Preparation for Structured, Consensus-based Peer Review Each year, teachers teaching a Year 11 class are asked to retain originals or copies of student work completed in Semester 2. Similarly, teachers teaching a Year 12 class should retain originals or copies of student work completed in Semester 1. Assessment and other documentation required by the Office of the Board of Senior Secondary Studies should also be kept. Year 11 work from Semester 2 of the previous year is presented for review at Moderation Day 1 in March, and Year 12 work from Semester 1 is presented for review at Moderation Day 2 in August. In the lead up to Moderation Day, a College Course Presentation (comprised of a document folder and a set of student portfolios) is prepared for each A and T course offered by the school, and is sent in to the Office of the Board of Senior Secondary Studies. The College Course Presentation The package of materials (College Course Presentation) presented by a college for review on moderation days in each course area will comprise the following: a folder containing supporting documentation as requested by the Office of the Board through memoranda to colleges. a set of student portfolios containing marked and/or graded written and non-written assessment responses and completed criteria and standards feedback forms. Evidence of all assessment responses on which the unit grade decision has been made is to be included in the student review portfolios. specific requirements for subject areas and types of evidence to be presented for each moderation day, which will be outlined by the Office of the Board of Senior Secondary Studies through memoranda and Information Papers. Version 2 16 Board Endorsed October 2006 – Amended December 2013 Bibliography Suitable Text Books Commons, C. et al. 1999, Chemistry 1, 3rd ed, Heinemann, Melbourne. Commons, C. et al. 1999 , Chemistry 2, 3rd ed, Heinemann, Melbourne. Elvins, C. et al. 1992, Chemistry in context – chemistry one, Heinemann Educational, Australia. Hill, G. and Holman, J. 1989, Chemistry in context, Nelson, Melbourne. Irwin, D. et al. 2001, Chemistry contexts book 1, Longman Sciences, Melbourne. Irwin, D. et al. 2002, Chemistry contexts book 2, Longman Sciences, Melbourne. James, M. et al. 1991 & 1992, Chemical connection, VCE chemistry, Books 1 & 2, Jacaranda Press, Melbourne. Sharwood, J. (ed) et al. 2000, Nelson chemistry (VCE Units 1 & 2), Nelson, Melbourne. Sharwood, J. (ed) et al. 2000, Nelson chemistry (VCE Units 3 & 4), Nelson, Melbourne. Smith, R. 2005, Conquering chemistry, 4th ed, McGraw-Hill, Rydalmere NSW. Thickett, G. 2000, Chemistry pathways 1 and 2, Macmillan, South Yarra VIC. Teacher Resources Aldridge, S. 1994, Biochemistry for advanced biology, Cambridge University Press, Cambridge UK. Australian Academy of Science, 1994, Environmental science, Australian Academy of Science, Canberra. Aylward, G. and Findlay, T. 1994, SI chemical data, John Wiley & Sons, Milton QLD. Anderton, J. Kolomyjec, C. Garnett, P. 1990, Chemistry problems for senior secondary school, STAWA. Brown, K. 1986, Moles: a survival guide for GCSE chemistry, Cambridge University Press, UK. Brown, W. 1997, Introduction to organic chemistry, Sanders College Publishing, Fort Worth. Bucat, R. (Ed) 1984, Elements of chemistry vol I &II, Australian Academy of Science, Canberra. Budavari, S (Ed) 1996, The merck index, 12th ed, Merck Research Laboratories, New Jersey. Carr, M. and Cardell, B. 1992, Biochemistry, Nelson, Melbourne. Clugston, M. and Flemming, R. 2000, Advanced chemistry, Oxford University Press, Oxford. Commons, C. and Hoogendoorn, B. 1998, Demonstrations for secondary schools chemistry, 5th ed, Heinemann, Melbourne. Commons, C. et al. 199l, Chemistry two, chemistry and the marketplace, energy & matter, Heinemann, London. Commons, C. et al. 1999, Chemistry 2: teacher's resource book, 3rd ed, Heinemann, Melbourne. Derry, N. et al, 2000, Chemistry Units 3 and 4, Wesley Longman de Vreeze, D.and McMicking, K. 1998, Instant lessons in chemistry, books 1,2,3, Blake Education, Sydney.. Elvins, C. et al. 1999, Chemistry 1: teacher's resource book, Heinemann, Melbourne. Elvins, C. et al. 1999, Chemistry one teacher’s resource book, 3rd ed, Heinemann, Melbourne. Fogliani, C. (Ed), Australian chemistry resource book, Vol 1-25, Charles Sturt University, Bathurst. Gadd, K. and Gurr, S. 1994, Chemistry: university of Bath: science 16-19, Nelson, UK. Green, N et al. 1984, Biological science 1, Cambridge University Press, UK. Gribben, P. and St Germain, W. 2005, Assignments in chemistry, books 1, 2 and 3, Blake Education, Sydney. Gribben, P. 1999, Interpreting data in chemistry books 1 & 2, Blake Education, Sydney. Version 2 17 Board Endorsed October 2006 – Amended December 2013 Gribben, P and Cassidy, M. 2003, Cambridge HSC study guide chemistry, Cambridge University Press, Melbourne. Harwood, R. 2002, Biochemistry, Cambridge University Press, Cambridge UK. Hornby, M and Peach, J. 1994, Foundations of organic chemistry, Oxford University Press, Oxford UK. James, D. and Matthews, G. 1991, Understanding the biochemistry of respiration, Cambridge University Press, Cambridge UK. Jones, J. 2002, Captivating chemistry: the acidic environment , University of Sydney & UTS, Sydney. Kennedy, E. 2005 Biology in context-the spectrum of life biochemistry, Oxford University Press, Melbourne. Laidler, G. 1991, Environmental chemistry; an australian perspective, Longman Cheshire, South Melbourne. Lainchbury, A. et al. (revised by) 1995, ILPAC equilibrium 1: principles, John Murray, London. Lainchbury, A. et al. (revised by) 1995, ILPAC s-block elements, the halogens, the periodic tables, John Murray, London. McMurry, J. 1994, Fundamentals of organic chemistry, Brookes/Cole, US. Nash, B. and Hargreaves, G. 1990, Chemistry activities, books 1 & 2, Macmillan, Melbourne. Purves, W. et al. 1998, Life; the science of biology, 5th ed, W.H.Freeman, US. Ramsden, E. 1996, Chemistry of the environment, Stanley Thornes Ltd., Cheltenham UK. Roebuck, C. 2000, Excel preliminary chemistry, Pascal Press, Sydney. St. Germain, W. 2003, Literacy in science books 3,4, Blake Education, Sydney. Selinger, B. 1998, Chemistry in the market place, 5th ed, Harcourt Brace Jovanovich, Marrickville NSW. Smith, D. 1986, Conquering chemistry HSC course, Blackline Masters, McGraw Hill, Sydney. Smith, D. et al. 2006, Chemistry in use book 1, McGraw Hill, Australia. Smith, R. 2004, Conquering chemistry preliminary guide, 4th ed, McGraw Hill, Australia. Stryer, L. 1991, Biochemistry, 3rd ed, W.H. Freeman & Co, New York. Tooley, P. 1975, Experiments in applied chemistry, John Murray, London. Tregarthen, T. 2003, Macquarie revision guides preliminary chemistry, Macmillan, Melbourne. Vogel, A. 1961, A text book of quantitative inorganic analysis, 3rd ed Longman, London. Wiecek, C. 1989, Chemistry for senior students, The Jacaranda Press, Sydney. Wilkinson, J. and Nash, B. 1989, The world of chemistry, books 1 & 2, text & practical manuals, Macmillan, Melbourne. Winter, M. 1995, Chemical bonding, Oxford University Press, Oxford. Zubay, G. et al. 1995, Principles of biochemistry, Wm. C. Brown, US. Zumdahl, S .1997, Chemistry, 5th ed, Houghton Mifflin Company, Boston. Journal Articles Relevant articles from Chemistry Review, Phillip Allan Updates, Oxfordshire. Relevant articles from ChemMatters, American Chemical Society, Washington. Version 2 18 Board Endorsed October 2006 – Amended December 2013 Audio visual Material An Introduction to Oxidation and Reduction (video), 1997 Learning Essentials Bonding Part 2 (video) Classroom Video Chemical Energy (video) Classroom Video Chemistry of the Environment (video) Classroom Video Chemistry 2 (video), 1998 Video Education Australasia Corrosion Reactions (video), 1997 Learning Essentials Equilibrium- the Concepts (DVD), 2003 VEA Australasia Equilibrium 2- Applying the Concepts (DVD), 2003 VEA Australasia Ionic & Covalent Bonding Part 1(video), Classroom Video Supercharged World of Chemistry Program 4 (Molarity, Titrations, Limiting Reagents) (video), 2001 Video Education Australasia The Amazing Mole (video), 2003 Video Education Australasia The Amazing World of Reactions in Water (video), 1997 Parts 1 and 2 Video Education Australasia The Revolution in Genetics (video), 1998 Quantum, ABC Science Unit CD ROMS D.Smith, S. Monteath, M. Gould, (CD ROM), Chemistry in Use Book 1 Teacher Resource The McGraw Hill Companies 2006 Web sites http://www.science.org.au/nova/037/037sit.htm http://www.greenhouse.gov.au/renewable/recp/biomass/index.html http://www.greenhouse.gov.au/renewable/ http://www.greenpeace.org.au/climate/solutions/renewables/biomass.html http://www.abc.net.au/rn/science/earth/stories/s123008.htm http://medlineplus.gov/ www.webelements.com/ www.chemicalelements.com/ hsc.csu.edu.au/chemistry/options/shipwrecks/2729/ch962Dec2_03.htm www.corrosion-doctors.org/Aircraft/galvseri-table.htm www.corrosion-club.com/galvseries.htm These were accurate at the time of publication. Version 2 19 Board Endorsed October 2006 – Amended December 2013 Resources Resources required for delivering this course include: Well-equipped laboratories Laboratory assistant Suitably qualified teaching staff Textbooks and scientific journals Data logging equipment and software Internet access Proposed Evaluation Procedures The course will be evaluated formally with student surveys, enrolment data and course completion data from the Year 12 Study. Feedback may also be sought from teachers and other interested parties (for example, former students engaged in tertiary study and staff at tertiary institutions). See Appendix for sample surveys. Version 2 20 Board Endorsed October 2006 – Amended December 2013 Introductory Chemistry Value: 1.0 Prerequisites Nil Specific Unit Goals This unit should enable students to: demonstrate a basic understanding of the atom, its composition and structure and explain the properties, behaviour and uses of matter based on this knowledge; select suitable purification techniques to separate mixtures into their components; carry out basic chemical experiments and measure, observe, report, discuss the results of such investigations; use material and equipment with due care and consideration for safety; write ionic formulas, covalent formulas and chemical equations, recognise scientific endeavour in chemistry over time and how it impacts on everyday life; understand the concepts of chemical bonding, and relate these concepts to the structure and properties of matter; use concepts of quantitative chemistry to solve stoichiometric problems. Content Matter and Materials general classification of matter – homogeneous/heterogeneous elements, compounds, and mixtures metals and non-metals review of physical and chemical properties, changes and means of separation Atomic Theory basic kinetic theory of matter brief historical overview up to and including Bohr structure of the nuclear atom atomic number, mass number, isotopes periodic table groups, periods and valence (simple compounds) Chemical Symbols, Formulae and Equations word equations chemical symbols and formulae molecular and structural (constitutional) formula balanced chemical equation writing, using common reaction types Bonding and Properties metallic bonding ionic bonding molecular covalent bonding including Lewis diagrams giant covalent lattice and covalent layer lattice Version 2 21 Board Endorsed October 2006 – Amended December 2013 Molecules and Intermolecular Forces shapes of molecules and polarity molecular and structural formulae intermolecular forces (dispersion forces, dipole – dipole interaction, hydrogen bonding) relationship between structure, properties and bond types The Mole Avogadro’s number relative atomic mass (including isotopes) molar mass (elements and compounds) percentage composition empirical and molecular formulae stoichiometry limiting reactant Reactions in Water relevant terms – soluble, insoluble, solute, solvent, precipitate, suspension types of solution solubility rules solubility curves solutions and dilution Teaching and Learning Strategies These may include the use of: practical work to provide concrete experiences to help students in the use of chemical formulae, equations and the nature of bonds; visual material including model kits to understand the nature and properties of different bond types and chemical formulae; chemical data books to assist identification of compounds according to their properties; quantitative gravimetric analysis to enhance the ideas and relevance of constant proportions of mass in compounds, solutions, reactants and products; preparation of solutions of given concentrations using both solutes and other solutions; time line(s) to provide a historical context to many of the concepts involved; quiz type exercises to emphasise the learning of chemical formulae and equation writing; homework to reinforce concepts covered plus more challenging questions to extend more able students; tutorials to support students. Version 2 22 Board Endorsed October 2006 – Amended December 2013 Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Molymod Modelling Kits. Suggested Texts: James, M. et al. 1991, Chemical connections VCE chemistry Book 1, (Ch 1-7 inclusive, Ch 14, Ch 16), Jacaranda Press, Melbourne. Sharwood, J. (ed) et al. 2000, Nelson chemistry (VCE Units 1 and 2), Nelson, Melbourne. Teacher Resources Aylward, G. and Findlay, T. 1994, SI chemical data, John Wiley & Sons, Milton, QLD Brown, K. 1991, MOLES, a survival guide for GCSE science, Cambridge University Press, Cambridge. Journal Articles Relevant articles from ChemMatters and Chemistry Review. Audio visual Material Chemical Bonding Series 1997, Video Education Australasia. The Atom 1989, World of Chemistry 2, Educational Film Center, University of Maryland. The Mole 1989, World of Chemistry 1, Educational Film Center University of Maryland The Mole 1994, Classroom Video French’s Forest Sydney. Electron Arrangement and Bonding 1984, Concepts in Science- Chemistry 1, TV Ontario Educational Communications Authority. The Mole Concept 1984, Concepts in Science- Chemistry 1, TV Ontario Educational Communications Authority. Atomic Theory and Chemistry 1990, Britannica. Version 2 23 Board Endorsed October 2006 – Amended December 2013 Web sites WebElements™ periodic table. Winters, M. http://www.webelements.com/ Atomic Structure Timeline http://www.watertown.k12.wi.us/HS/Staff/Buescher/atomtime.asp Atomic Structure http://web.jjay.cuny.edu/~acarpi/NSC/3-atoms.htm The Writing of Formulas http://members.aol.com/profchm/formwrit.html Writing Formulas and Naming Compounds http://members.aol.com/profchm/naming.html States of Matter http://www.chem.purdue.edu/gchelp/atoms/states.html Chemical Bonding Information http://users.senet.com.au/~rowanb/chem/chembond.htm Chemical Bonding. Carpi, Anthony http://www.visionlearning.com/library/module_viewer.php?mid=55 Intermolecular Forces http://chemed.chem.purdue.edu/genchem/topicreview/bp/intermol/intermol.html The Mole Table of Contents http://dbhs.wvusd.k12.ca.us/webdocs/Mole/Mole.html Writing Equations: Precipitation Reactions http://www.ausetute.com.au/ppteeqtn.html Solubility of Solutes and Aqueous Solutions http://www.chem.lsu.edu/lucid/tutorials/solubility/Solubility.html These were accurate at the time of publication. Version 2 24 Board Endorsed October 2006 – Amended December 2013 Introduction to Chemistry Value: 0.5 Prerequisites Nil Specific Unit Goals This unit should enable students to: demonstrate a basic understanding of the atom its composition and structure and explain the properties, behaviour and uses of matter based on this knowledge; select suitable purification techniques to separate mixtures into their components; carry out basic chemical experiments and measure, observe, report, discuss the results of such investigations; use material and equipment with due care and consideration for safety; write ionic formulas, covalent formulas and chemical equations, recognise scientific endeavour in chemistry over time and how it impacts on everyday life; understand the concepts of chemical bonding, and relate these concepts to the structure and properties of matter. Content Matter and Materials general classification of matter – homogeneous/heterogeneous elements, compounds, and mixtures review of physical and chemical properties, changes and means of separation Atomic Theory basic kinetic theory of matter brief historical overview up to and including Bohr structure of the nuclear atom atomic number, mass number, isotopes periodic table groups, periods and valence (simple compounds) Chemical Symbols, Formulae and Equations word equations chemical symbols and formulae molecular and structural (constitutional) formula balanced chemical equation writing, using common reaction types Bonding and Properties metallic bonding and alloys ionic bonding molecular covalent bonding including Lewis diagrams giant covalent lattice and covalent layer lattice Molecules and Intermolecular Forces shapes of molecules and polarity molecular and structural formulae intermolecular forces (dispersion forces, dipole – dipole interaction, hydrogen bonding) relationship between structure, properties and bond types Version 2 25 Board Endorsed October 2006 – Amended December 2013 Teaching and Learning Strategies These may include the use of: practical work to provide concrete experiences to help students in the use of chemical formulae, equations and the nature of bonds; visual material including model kits to understand the nature and properties of different bond types and chemical formulae; chemical data books to assist identification of compounds according to their properties; time line(s) to provide a historical context to many of the concepts involved; quiz type exercises to emphasise the learning of chemical formulae and equation writing; homework to reinforce concepts covered plus more challenging questions to extend more able students; tutorials to support students. Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Version 2 26 Board Endorsed October 2006 – Amended December 2013 Specific Unit Resources Molymod Modelling Kits. Suggested Texts: James, M. et al. 1991, Chemical connections VCE chemistry Book 1, (Ch 1-7 inclusive, Ch 14, Ch 16), Jacaranda Press, Melbourne. Sharwood, J. (ed) et al. 2000, Nelson chemistry (VCE Units 1 and 2), Nelson, Melbourne. Teacher Resources Aylward, G. and Findlay, T. 1994, SI chemical data, John Wiley & Sons, Milton, QLD ` Brown, K. 1991, MOLES, a survival guide for GCSE science, Cambridge University Press, Cambridge UK. Journal Articles Relevant articles from ChemMatters and Chemistry Review Audio visual Material Chemical Bonding Series (video), 1997 Video Education Australasia The Atom-World of Chemistry 2 (video), 1989 Educational Film Center, University of Maryland Electron Arrangement and Bonding- Concepts in Science- Chemistry 1 (video), 1984 TV Ontario Educational Communications Authority Atomic Theory and Chemistry (video), 1990, Britannica Web sites WebElements™ periodic table. Winters, M. http://www.webelements.com/ Atomic Structure Timeline http://www.watertown.k12.wi.us/HS/Staff/Buescher/atomtime.asp Atomic Structure http://web.jjay.cuny.edu/~acarpi/NSC/3-atoms.htm The Writing of Formulas http://members.aol.com/profchm/formwrit.html Writing Formulas and Naming Compounds http://members.aol.com/profchm/naming.html States of Matter http://www.chem.purdue.edu/gchelp/atoms/states.html Chemical Bonding Information http://users.senet.com.au/~rowanb/chem/chembond.htm Chemical Bonding. Carpi, Anthony Version 2 27 Board Endorsed October 2006 – Amended December 2013 http://www.visionlearning.com/library/module_viewer.php?mid=55 Intermolecular Forces http://chemed.chem.purdue.edu/genchem/topicreview/bp/intermol/intermol.html These were accurate at the time of publication. Version 2 28 Board Endorsed October 2006 – Amended December 2013 Moles, Water, Reactions & Acids and Redox Value: 1.0 This unit combines The Mole, Water and Reactions in Solution (0.5) with Acids and Redox (0.5) Prerequisites Introductory Chemistry Specific Unit Goals This unit should enable students to: carry out basic chemical experiments and measure, observe, report, discuss the results of such investigations; use material and equipment with due care and consideration for safety; write ionic formulas, covalent formulas and chemical equations, recognise scientific endeavour in chemistry over time and how it impacts on everyday life; use concepts of quantitative chemistry to solve stoichiometric problems. investigate reactions of acids and bases in water using safe and accurate laboratory procedures, while becoming aware of the importance and complexity of these reactions in the management of the environment; observe, collect and interpret data relating to redox reactions and electrochemistry particularly with respect to processes of economic importance (such as corrosion and its prevention); solve problems cooperatively and communicate findings clearly in a variety of ways using appropriate scientific language. Content The Mole Avogadro’s number relative atomic mass (including isotopes) molar mass (elements and compounds) percentage composition empirical and molecular formulae stoichiometry limiting reactant Reactions in Water relevant terms – soluble, insoluble, solute, solvent, precipitate, suspension types of solution solubility rules solubility curves solutions and dilution Acids and Bases definitions, chemical properties Arrhenius, Bronsted-Lowry theories polyprotic acids conjugate acid/base pairs neutralisation using titration dissociation, ionisation Version 2 29 Board Endorsed October 2006 – Amended December 2013 strong and weak acids & bases indicators, pH (simple explanation with strong acids and bases only) Redox oxidation/reduction reactions oxidation number oxidising and reducing agents introduction to redox equations including half equations electrochemical series corrosion : process and protection the galvanic cell, batteries, emf Teaching and Learning Strategies These may include the use of: practical work to provide concrete experiences to help students in the use of chemical formulae, equations and the nature of bonds; chemical data books to assist identification of compounds according to their properties; quantitative gravimetric analysis to enhance the ideas and relevance of constant proportions of mass in compounds, solutions, reactants and products; preparation of solutions of given concentrations using both solutes and other solutions; quiz type exercises to emphasise the learning of chemical formulae and equation writing; homework to reinforce concepts covered plus more challenging questions to extend more able students; tutorials to support students. volumetric analysis; detailed practical analysis of corrosion accompanied by chemical reactions and explanations; audio visual material to stimulate class discussion about the importance of redox reactions in nature, and the way reactions may be controlled by technology to further production in industry; sessions of problem solving within small groups complemented by students demonstrating how to work out the solution in front of the class; the library as a source for more in-depth research of the relevance to the environment of the topics studied in this unit; molecular model kits and laboratory experimental investigations carried out by individuals or small groups with occasional teacher/student demonstrations; excursions and visiting speakers complemented by library research (including information retrieval using CD-ROMs and the internet); interactive lessons using audiovisual aids, seminars and oral presentations by students. Assessment Refer to page 13. Version 2 30 Board Endorsed October 2006 – Amended December 2013 Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Suggested Texts: James, M. et al. 1991, Chemical connections VCE chemistry Book 1, (Ch 1-7 inclusive, Ch 14, Ch 16), Jacaranda Press, Melbourne. Sharwood, J. (ed) et al. 2000, Nelson chemistry (VCE Units 1 and 2), Nelson, Melbourne. Commons, C. et al. 1999, Chemistry 1, 3rd ed, Heinemann, Melbourne. Irwin, D. et al. 2002, Chemistry contexts book 2, Longman Sciences, Melbourne. Smith, R. 2005, Conquering chemistry, 4th ed, McGraw-Hill, Australia. Teacher Resources Aylward, G. and Findlay, T. 1994, SI chemical data, John Wiley & Sons, Milton, QLD Brown, K. 1991, MOLES, a survival guide for GCSE science, Cambridge University Press, Cambridge. Books Aylward, G. and Findlay, T. 1994, SI chemical data, 3rd ed, John Wiley & Sons, Milton. Bucat, R.(Ed) 1984, Elements of chemistry, Vol 1, 2 and teachers' guides, Australian Academy of Science ,Canberra. de Vreeze, D. and McMicking, K. 1998, Instant lessons in chemistry Bks 2, 3, Blake Education, Sydney. Gogarty, K. et al. 2006, Practical experiments in chemistry 1 & 2, Blake Education, Victoria. Gribben, P. 1999, Interpreting data in chemistry Bk 2, Blake Education, Victoria. Jones, J. 2002, Captivating chemistry: the acidic environment, University of Sydney & UTS, Sydney. McMurry, J. 1994, Fundamentals of organic chemistry, Brookes/Cole, US. Version 2 31 Board Endorsed October 2006 – Amended December 2013 Journal Articles Relevant articles from ChemMatters and Chemistry Review Relevant articles from ChemMatters and Chemistry Review Audio visual Material The Mole World of Chemistry 1 (video), 1989, Educational Film Center University of Maryland The Mole (updated version) (video), 1994, Classroom Video French’s Forest Sydney The Mole Concept- Concepts in Science- Chemistry 1 (video), 1984, TV Ontario Educational Communications Authority Oxidation And Reduction World of Chemistry 2 1989, Educational Film Center University of Maryland, US. Petroleum: A Vital Resource 1993, Brittanica Centre. Carbon Chemistry 1992, Classroom Video French’s Forest NSW. Carbon the Compromiser World of Chemistry 2 1989, Educational Film Center University of Maryland, US. Addition Polymers-Investigating Polymers Series 1998, Video Education Australasia. Condensation Polymers-Investigating Polymers Series 1998, Video Education Australasia. An Introduction to Oxidation and Reduction 1997, Learning Essentials. Corrosion Reactions 1997, Learning Essentials. Web sites WebElements™ periodic table. Winters, M. http://www.webelements.com/ The Mole Table of Contents http://dbhs.wvusd.k12.ca.us/webdocs/Mole/Mole.html Writing Equations: Precipitation Reactions http://www.ausetute.com.au/ppteeqtn.html Solubility of Solutes and Aqueous Solutions http://www.chem.lsu.edu/lucid/tutorials/solubility/Solubility.html http://www.science.org.au/nova/037/037sit.htm http://www.webelements.com http://www.chemicalelements.com/ http://hsc.csu.edu.au/chemistry/options/shipwrecks/2729/ch962Dec2_03.htm http://corrosion-club.com/galvseries.htm. http://www.unisanet.unisa.edu.au/08365/h&pacba.htm#acid_base_top http://library.nudgee.com/acid_and_bases.htm These were accurate at the time of publication. Version 2 32 Board Endorsed October 2006 – Amended December 2013 The Mole, Water & Reactions in Solution Value: 0.5 Prerequisites Nil Specific Unit Goals This unit should enable students to: carry out basic chemical experiments and measure, observe, report, discuss the results of such investigations; use material and equipment with due care and consideration for safety; write ionic formulas, covalent formulas and chemical equations, recognise scientific endeavour in chemistry over time and how it impacts on everyday life; use concepts of quantitative chemistry to solve stoichiometric problems. Content The Mole Avogadro’s number relative atomic mass (including isotopes) molar mass (elements and compounds) percentage composition empirical and molecular formulae stoichiometry limiting reactant Reactions in Water relevant terms – soluble, insoluble, solute, solvent, precipitate, suspension types of solution solubility rules solubility curves solutions and dilution Teaching and Learning Strategies These may include the use of: practical work to provide concrete experiences to help students in the use of chemical formulae, equations and the nature of bonds; chemical data books to assist identification of compounds according to their properties; quantitative gravimetric analysis to enhance the ideas and relevance of constant proportions of mass in compounds, solutions, reactants and products; preparation of solutions of given concentrations using both solutes and other solutions; quiz type exercises to emphasise the learning of chemical formulae and equation writing; homework to reinforce concepts covered plus more challenging questions to extend more able students; tutorials to support students. Assessment Version 2 33 Board Endorsed October 2006 – Amended December 2013 Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Suggested Texts: James, M. et al. 1991, Chemical connections VCE chemistry Book 1, (Ch 1-7 inclusive, Ch 14, Ch 16), Jacaranda Press, Melbourne. Sharwood, J. (ed) et al. 2000, Nelson chemistry (VCE Units 1 and 2), Nelson, Melbourne. Teacher Resources Aylward, G. and Findlay, T. 1994, SI chemical data, John Wiley & Sons, Milton, QLD Brown, K. 1991, MOLES, a survival guide for GCSE science, Cambridge University Press, Cambridge. Journal Articles Relevant articles from ChemMatters and Chemistry Review Audio visual Material The Mole World of Chemistry 1 (video), 1989, Educational Film Center University of Maryland The Mole (updated version) (video), 1994, Classroom Video French’s Forest Sydney The Mole Concept- Concepts in Science- Chemistry 1 (video), 1984, TV Ontario Educational Communications Authority Version 2 34 Board Endorsed October 2006 – Amended December 2013 Web sites WebElements™ periodic table. Winters, M. http://www.webelements.com/ The Mole Table of Contents http://dbhs.wvusd.k12.ca.us/webdocs/Mole/Mole.html Writing Equations: Precipitation Reactions http://www.ausetute.com.au/ppteeqtn.html Solubility of Solutes and Aqueous Solutions http://www.chem.lsu.edu/lucid/tutorials/solubility/Solubility.html These were accurate at the time of publication. Version 2 35 Board Endorsed October 2006 – Amended December 2013 Acids, Redox and Organic Chemistry Value: 1.0 Prerequisites Introductory Chemistry Specific Unit Goals This unit should enable students to: investigate reactions of acids and bases in water using safe and accurate laboratory procedures, while becoming aware of the importance and complexity of these reactions in the management of the environment; observe, collect and interpret data relating to redox reactions and electrochemistry particularly with respect to processes of economic importance (such as corrosion and its prevention); understand how carbon chemistry is based on the unique structure of the carbon atom; use practical investigations to appreciate that carbon compounds have certain common structures which determine their properties, function and classification into major groups; research the impact of organic substances, especially polymers, in an environmental, social and economic context; solve problems cooperatively and communicate findings clearly in a variety of ways using appropriate scientific language. Content Acids and Bases definitions, chemical properties Arrhenius, Bronsted-Lowry theories polyprotic acids conjugate acid/base pairs neutralisation using titration dissociation, ionisation strong and weak acids & bases indicators, pH (simple explanation with strong acids and bases only) Redox oxidation/reduction reactions oxidation number oxidising and reducing agents introduction to redox equations including half equations electrochemical series corrosion : process and protection the galvanic cell, batteries, emf Organic Chemistry and Polymers sources of organic compounds (fossil fuels - natural gas, petroleum, coal, living organisms) the carbon atom alkanes, alkenes, alkynes, alicyclics, aromatics and isomers systematic nomenclature and structural formulae functional group chemistry : Version 2 36 Board Endorsed October 2006 – Amended December 2013 haloalkanes alkanols alkanals and alkanones alkanoic acids alkanoates (salts & esters) alkanamines and alkanamides natural and synthetic polymers thermoplastics and thermosets addition and condensation polymerisation cross linking manufacturing techniques for plastics importance to society environmental concerns regarding use Teaching and Learning Strategies These may include the use of: volumetric analysis; detailed practical analysis of corrosion accompanied by chemical reactions and explanations; audio visual material to stimulate class discussion about the importance of redox reactions in nature, and the way reactions may be controlled by technology to further production in industry; sessions of problem solving within small groups complemented by students demonstrating how to work out the solution in front of the class; the library as a source for more in-depth research of the relevance to the environment of the topics studied in this unit; molecular model kits and laboratory experimental investigations carried out by individuals or small groups with occasional teacher/student demonstrations; excursions and visiting speakers complemented by library research (including information retrieval using CD-ROMs and the internet); interactive lessons using audiovisual aids, seminars and oral presentations by students. Assessment Refer to page 13. Version 2 37 Board Endorsed October 2006 – Amended December 2013 Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Suggested texts: Commons, C. et al. 1999, Chemistry 1, 3rd ed, Heinemann, Melbourne. Irwin, D. et al. 2002, Chemistry contexts book 2, Longman Sciences, Melbourne. James, M. et al. 1991, Chemical connections VCE Chemistry Book 1, Ch. 8, 9, 17, 20. The Jacaranda Press, Sydney. Sharwood, J. (ed) et al. 2000, Nelson chemistry (VCE units 1 & 2), Nelson, Melbourne. Smith, R. 2005, Conquering chemistry, 4th ed, McGraw-Hill, Australia. Books Aylward, G. and Findlay, T. 1994, SI chemical data, 3rd ed, John Wiley & Sons, Milton. Bucat, R.(Ed) 1984, Elements of chemistry, Vol 1, 2 and teachers' guides, Australian Academy of Science ,Canberra. de Vreeze, D. and McMicking, K. 1998, Instant lessons in chemistry Bks 2, 3, Blake Education, Sydney. Gogarty, K. et al. 2006, Practical experiments in chemistry 1 & 2, Blake Education, Victoria. Gribben, P. 1999, Interpreting data in chemistry Bk 2, Blake Education, Victoria. Jones, J. 2002, Captivating chemistry: the acidic environment, University of Sydney & UTS, Sydney. McMurry, J. 1994, Fundamentals of organic chemistry, Brookes/Cole, US. Journal Articles Relevant articles from ChemMatters and Chemistry Review Audio visual Material Oxidation And Reduction World of Chemistry 2 1989, Educational Film Center University of Maryland, US. Petroleum: A Vital Resource 1993, Brittanica Centre. Carbon Chemistry 1992, Classroom Video French’s Forest NSW. Carbon the Compromiser World of Chemistry 2 1989, Educational Film Center University of Maryland, US. Addition Polymers-Investigating Polymers Series 1998, Video Education Australasia. Version 2 38 Board Endorsed October 2006 – Amended December 2013 Condensation Polymers-Investigating Polymers Series 1998, Video Education Australasia. An Introduction to Oxidation and Reduction 1997, Learning Essentials. Corrosion Reactions 1997, Learning Essentials. Web sites http://www.science.org.au/nova/037/037sit.htm http://www.webelements.com http://www.chemicalelements.com/ http://hsc.csu.edu.au/chemistry/options/shipwrecks/2729/ch962Dec2_03.htm http://corrosion-club.com/galvseries.htm. http://www.unisanet.unisa.edu.au/08365/h&pacba.htm#acid_base_top http://library.nudgee.com/acid_and_bases.htm These were accurate at the time of publication. Version 2 39 Board Endorsed October 2006 – Amended December 2013 Acids and Redox Value: 0.5 Prerequisites Introductory Chemistry Specific Unit Goals This unit should enable students to: investigate reactions of acids and bases in water using safe and accurate laboratory procedures, while becoming aware of the importance and complexity of these reactions in the management of the environment; observe, collect and interpret data relating to redox reactions and electrochemistry particularly with respect to processes of economic importance (such as corrosion and its prevention); solve problems cooperatively and communicate findings clearly in a variety of ways using appropriate scientific language. Content Acids and Bases definitions, chemical properties Arrhenius, Bronsted-Lowry theories polyprotic acids conjugate acid/base pairs neutralisation using titration dissociation, ionisation strong and weak acids & bases indicators, pH (simple explanation with strong acids and bases only) Redox oxidation/reduction reactions oxidation number oxidising and reducing agents introduction to redox equations including half equations electrochemical series corrosion : process and protection the galvanic cell, batteries, emf Version 2 40 Board Endorsed October 2006 – Amended December 2013 Teaching and Learning Strategies These may include the use of: volumetric analysis; detailed practical analysis of corrosion accompanied by chemical reactions and explanations; audio visual material to stimulate class discussion about the importance of redox reactions in nature, and the way reactions may be controlled by technology to further production in industry; sessions of problem solving within small groups complemented by students demonstrating how to work out the solution in front of the class; the library as a source for more in-depth research of the relevance to the environment of the topics studied in this unit; molecular model kits and laboratory experimental investigations carried out by individuals or small groups with occasional teacher/student demonstrations; excursions and visiting speakers complemented by library research (including information retrieval using CD-ROMs and the internet); interactive lessons using audiovisual aids, seminars and oral presentations by students. Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Suggested texts: Commons, C. et al. 1999, Chemistry 1, 3rd ed, Heinemann, Melbourne. Irwin, D. et al. 2002, Chemistry contexts book 2, Longman Sciences, Melbourne. James, M. et al. 1991, Chemical connections VCE Chemistry Book 1, Ch. 8, 9, 17, 20. The Jacaranda Press, Sydney. Sharwood, J. (ed) et al. 2000, Nelson chemistry (VCE units 1 & 2), Nelson, Melbourne. Smith, R. 2005, Conquering chemistry, 4th ed, McGraw-Hill, Australia. Version 2 41 Board Endorsed October 2006 – Amended December 2013 Books Aylward, G. and Findlay, T. 1994, SI chemical data, 3rd ed, John Wiley & Sons, Milton. Bucat, R.(Ed) 1984, Elements of chemistry, Vol 1, 2 and teachers' guides, Australian Academy of Science ,Canberra. de Vreeze, D. and McMicking, K. 1998, Instant lessons in chemistry Bks 2, 3, Blake Education, Sydney. Gogarty, K. et al. 2006, Practical experiments in chemistry 1 & 2, Blake Education, Victoria. Gribben, P. 1999, Interpreting data in chemistry Bk 2, Blake Education, Victoria. Jones, J. 2002, Captivating chemistry: the acidic environment, University of Sydney & UTS, Sydney. McMurry, J. 1994, Fundamentals of organic chemistry, Brookes/Cole, US. Journal Articles Relevant articles from ChemMatters and Chemistry Review Audio visual Material Oxidation And Reduction World of Chemistry 2 1989, Educational Film Center University of Maryland, US. Petroleum: A Vital Resource 1993, Brittanica Centre. Carbon Chemistry 1992, Classroom Video French’s Forest NSW. Carbon the Compromiser World of Chemistry 2 1989, Educational Film Center University of Maryland, US. Addition Polymers-Investigating Polymers Series 1998, Video Education Australasia. Condensation Polymers-Investigating Polymers Series 1998, Video Education Australasia. An Introduction to Oxidation and Reduction 1997, Learning Essentials. Corrosion Reactions 1997, Learning Essentials. Web sites http://www.science.org.au/nova/037/037sit.htm http://www.webelements.com http://www.chemicalelements.com/ http://hsc.csu.edu.au/chemistry/options/shipwrecks/2729/ch962Dec2_03.htm http://corrosion-club.com/galvseries.htm. http://www.unisanet.unisa.edu.au/08365/h&pacba.htm#acid_base_top http://library.nudgee.com/acid_and_bases.htm These were accurate at the time of publication. Version 2 42 Board Endorsed October 2006 – Amended December 2013 Organic Chemistry Value: 0.5 Prerequisites Introductory Chemistry Specific Unit Goals This unit should enable students to: understand how carbon chemistry is based on the unique structure of the carbon atom; use practical investigations to appreciate that carbon compounds have certain common structures which determine their properties, function and classification into major groups; research the impact of organic substances, especially polymers, in an environmental, social and economic context; solve problems cooperatively and communicate findings clearly in a variety of ways using appropriate scientific language. Content Organic Chemistry and Polymers sources of organic compounds (fossil fuels - natural gas, petroleum, coal, living organisms) the carbon atom alkanes, alkenes, alkynes, alicyclics, aromatics and isomers systematic nomenclature and structural formulae functional group chemistry : haloalkanes alkanols alkanals and alkanones alkanoic acids alkanoates (salts & esters) alkanamines and alkanamides natural and synthetic polymers thermoplastics and thermosets addition and condensation polymerisation cross linking manufacturing techniques for plastics importance to society environmental concerns regarding use Version 2 43 Board Endorsed October 2006 – Amended December 2013 Teaching and Learning Strategies These may include the use of: sessions of problem solving within small groups complemented by students demonstrating how to work out the solution in front of the class; the library as a source for more in-depth research of the relevance to the environment of the topics studied in this unit; molecular model kits and laboratory experimental investigations carried out by individuals or small groups with occasional teacher/student demonstrations; excursions and visiting speakers complemented by library research (including information retrieval using CD-ROMs and the internet); interactive lessons using audiovisual aids, seminars and oral presentations by students. Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Suggested texts: Commons, C. et al. 1999, Chemistry 1, 3rd ed, Heinemann, Melbourne. Irwin, D. et al. 2002, Chemistry contexts book 2, Longman Sciences, Melbourne. James, M. et al. 1991, Chemical connections VCE Chemistry Book 1, Ch. 8, 9, 17, 20. The Jacaranda Press, Sydney. Sharwood, J. (ed) et al. 2000, Nelson chemistry (VCE units 1 & 2), Nelson, Melbourne. Smith, R. 2005, Conquering chemistry, 4th ed, McGraw-Hill, Australia. Books Aylward, G. and Findlay, T. 1994, SI chemical data, 3rd ed, John Wiley & Sons, Milton. Bucat, R.(Ed) 1984, Elements of chemistry, Vol 1, 2 and teachers' guides, Australian Academy of Science ,Canberra. de Vreeze, D. and McMicking, K. 1998, Instant lessons in chemistry Bks 2, 3, Blake Education, Sydney. Gogarty, K. et al. 2006, Practical experiments in chemistry 1 & 2, Blake Education, Victoria. Gribben, P. 1999, Interpreting data in chemistry Bk 2, Blake Education, Victoria. Version 2 44 Board Endorsed October 2006 – Amended December 2013 Jones, J. 2002, Captivating chemistry: the acidic environment, University of Sydney & UTS, Sydney. McMurry, J. 1994, Fundamentals of organic chemistry, Brookes/Cole, US. Journal Articles Relevant articles from ChemMatters and Chemistry Review Audio visual Material Oxidation And Reduction World of Chemistry 2 1989, Educational Film Center University of Maryland, US. Petroleum: A Vital Resource 1993, Brittanica Centre. Carbon Chemistry 1992, Classroom Video French’s Forest NSW. Carbon the Compromiser World of Chemistry 2 1989, Educational Film Center University of Maryland, US. Addition Polymers-Investigating Polymers Series 1998, Video Education Australasia. Condensation Polymers-Investigating Polymers Series 1998, Video Education Australasia. An Introduction to Oxidation and Reduction 1997, Learning Essentials. Corrosion Reactions 1997, Learning Essentials. Web sites http://www.science.org.au/nova/037/037sit.htm http://www.webelements.com http://www.chemicalelements.com/ http://hsc.csu.edu.au/chemistry/options/shipwrecks/2729/ch962Dec2_03.htm http://corrosion-club.com/galvseries.htm. http://www.unisanet.unisa.edu.au/08365/h&pacba.htm#acid_base_top http://library.nudgee.com/acid_and_bases.htm These were accurate at the time of publication. Version 2 45 Board Endorsed October 2006 – Amended December 2013 Physical Chemistry Value: 1.0 Prerequisites Minor in Chemistry Specific Unit Goals This unit should enable students to: demonstrate an understanding of atomic structure including quantum numbers and the gradual filling of orbitals determine the position of elements in the periodic table; use practical investigations to observe the common properties and trends of elements within "groups" and "blocks" in the periodic table with special focus on the transition elements; use scientific investigation to understand the inter relationships of factors affecting the behaviour of gases, qualitatively and, where possible, quantitatively; organise and process data collected by careful measurements so as to enhance their understanding of energy changes in chemical reactions; solve computational problems relating to thermochemical equations; investigate the principles of equilibrium as they relate to chemistry in living systems and/or industrial processes; understand the dynamic nature of chemical reactions and predict the impact of imposed changes; demonstrate effective research and communicate findings with clarity. Content Gases kinetic molecular theory gas laws (Gay-Lussac, Charles, Boyle, General Gas Equation, Ideal Gas Equation and Dalton’s Law of Partial Pressures) molar volume, Avogadro's hypothesis, real and ideal gases Modern Atomic Theory models from Dalton to quantum mechanical organisation of, and trends in the periodic table, eg ionisation energy, atomic radius, ionic radius, melting point, electronegativity etc. transition elements and their properties Thermochemistry definitions and conversions energy changes in chemical reactions ΔHr, ΔHc, ΔHf, ΔHat, ΔHv and bond energies quantitative aspects of energy input and output enthalpy diagrams calorimetry and Hess's law sources of chemical energy for fuel Version 2 46 Board Endorsed October 2006 – Amended December 2013 Reaction Kinetics and Equilibrium activation energy reaction rates, catalysis, activated complex concepts of equilibrium Le Chatelier's principle, the equilibrium constant, Kc, Kw, Ksp strong and weak acids, pH, Ka, Kb and buffers acid base titrations, indicator theory applications in industry and living systems Teaching and Learning Strategies These may include the use of: discharge tubes and spectrometers to demonstrate atomic finger prints; practical investigations that show the properties of elements in a group and period, with particular emphasis on transition elements; specialised equipment to demonstrate the laws of Boyle, Charles and Gay-Lussac; calorimetric exercises demonstrating quantitative aspects of energy changes including Hess's Law; electronic temperature probe and computer assisted graphing of results; small group work to solve thermodynamic problems combined with student demonstration of problem solving to the class; a written research assignment in the form of an in-class or polished essay or a written item in line with AST expectations; practical activities examining Le Chatelier’s Principle. Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Version 2 47 Board Endorsed October 2006 – Amended December 2013 Specific Unit Resources Suggested Texts: Bucat, R (Ed) 1984, Elements of chemistry vol I & II, Australian Academy of Science, Canberra. James, M. et al. 1991 & 1992, Chemical connection, VCE chemistry, Book 1(chapter 19) & Book 2 (chapters 13,14,24,25,26), Jacaranda Press, Melbourne. Smith, R. 2005, Conquering chemistry, 3rd ed, McGraw-Hill, Rydalmere NSW (chapters 13,14,15,16,19,23,25) . Wiecek, C. 1989, Chemistry for senior students, The Jacaranda Press, Sydney. Journal Articles Relevant articles from Chemistry Review, Phillip Allan Updates, Oxfordshire. Relevant articles from ChemMatters, American Chemical Society, Washington. Audio visual Material Acid Rain and Kw Chemistry Connection Series #40 1997, (video) Classroom. Chemistry of the Environment (video) Classroom Video. Hess’s Law and Enthalpy Communication-Chemistry Connection Series #8 1997 (video) Classroom Chemical Equilibrium 1984, Concepts in Science Chemistry 1 TV Ontario. Chemical Energy, Classroom Video. Equilibrium- the Concepts (DVD) 2003, VEA Australasia. Equilibrium 2- Applying the Concepts (DVD) 2003, VEA Australasia. CD ROMS D.Smith, S. Monteath, M. Gould, (CD ROM), Chemistry in Use Book 1 Teacher Resource The McGraw Hill Companies 2006 Version 2 48 Board Endorsed October 2006 – Amended December 2013 Biochemistry Value: 1.0 Prerequisites Minor in Chemistry Specific Unit Goals This unit should enable students to: outline the relationship between cell functions and structure, understand the structure of biomolecules, their identification and the relationship between structure and function; carry out simple experiments which demonstrate the control and management of chemical reactions; use scientific investigations to understand the complexity of processes in living organisms at a molecular level, both qualitatively and, where possible, quantitatively; demonstrate safe handling of materials and equipment and work effectively as an individual or in a team; gain an appreciation of the ethical issues involved in genetic engineering, and an understanding of the chemical processes involved. Content Introduction to Biochemistry Cell structure and function Biomolecules Revision of functional groups Monomers and polymers Saccharides - monosaccharides, open and ring structure - disaccharides, bonding between rings - polysaccharides – chains and cross-linking Lipids – fatty acids, triglycerides, phosphoglycerides, lipid bi-layers, and simple steroid structure Amino acids Proteins and their structure - primary, secondary, tertiary and quaternary Enzymes - proteins in action factors influencing activity Nucleic acids – RNA - three types, structure and function DNA - structure and function Protein synthesis Cellular Respiration Emphasis should be on inputs and outputs, and location in cell of process, rather than the chemical detail of each reaction Glycolysis Krebs cycle Electron transfer chain Alternative pathways (anaerobic) Coupled reactions, energy outcomes Role of ATP, NADH, FADH2 and Coenzyme-A Version 2 49 Board Endorsed October 2006 – Amended December 2013 Photosynthesis Light and dark reactions The photo-systems, the Calvin Cycle Genetic Engineering DNA sequencing - the Sanger technique and its modern derivatives Electrophoresis Gene splicing Benefits of genetic engineering and ethical issues. Case studies of some Australian examples, e.g. cotton, canola, human insulin. Teaching and Learning Strategies These may include the use of: interactive lessons incorporating audiovisual aids, discussion and debate, group work etc; practical investigations with an emphasis on the analysis and interpretation of data, and the presentation of results; use of model building, e.g. Molymod kits, to gain an understanding of molecular structure; peer tutoring / student presentations / student as teacher; research with the aim to supplement learning outcomes in practical activities; a detailed written/oral presentation of a biochemical process studied; visit to CSIRO Green Machine and/or ANU and/or UC to see DNA sequencing and other techniques which cannot be carried out in a college laboratory. Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Version 2 50 Board Endorsed October 2006 – Amended December 2013 Specific Unit Resources Molymod Modelling Kits Suggested Text Harwood R, 2002, Biochemistry, Cambridge University Press, Cambridge. Teacher Resources Aldridge, S. 1994, Biochemistry for advanced biology, Cambridge University Press, Cambridge. Green, N.P.D. et al. 1984, Biological science I chapters 5&6, Cambridge University Press,UK. James, M.1992, Chemical connections book 2 , The Jacaranda Press, Sydney. Selinger, B. 1990, Chemistry in the market place, 4th ed. Harcourt Brace Jovanovich, Marrickville. Tooley, P. 1975, Experiments in applied chemistry, John Murray, London. Audio visual Material Cellular Respiration – Concepts In Science Biology 1984, TV Ontario Educational Communications Authority. Photosynthesis - Concepts In Science Biology 1984, TV Ontario Educational Communications Authority. Protein Synthesis Concepts In Science Biology 1984, TV Ontario Educational Communications. Genetic Engineering Education Kit, 1992, CSIRO. ABC video on DNA 2005. CD ROMS DNA Interactive DVD (PAL version), 2003, Howard Hughes Medical Institute, Cold Springs Harbour Laboratory and Windfall Films Ltd. Version 2 51 Board Endorsed October 2006 – Amended December 2013 Biochemical Structures and Functions Value: 0.5 Prerequisites Minor in Chemistry Specific Unit Goals This unit should enable students to: outline the relationship between cell functions and structure, understand the structure of biomolecules, their identification and the relationship between structure and function; carry out simple experiments which demonstrate the control and management of chemical reactions; use scientific investigations to understand the complexity of processes in living organisms at a molecular level, both qualitatively and, where possible, quantitatively; demonstrate safe handling of materials and equipment and work effectively as an individual or in a team. Content Introduction to Biochemistry Cell structure and function Biomolecules Revision of functional groups Monomers and polymers Saccharides - monosaccharides, open and ring structure - disaccharides, bonding between rings - polysaccharides – chains and cross-linking. Lipids - fatty acids, triglycerides, phosphoglycerides, lipid bi-layers, and simple steroid structure. Amino acids Proteins and their structure, - primary, secondary, tertiary and quaternary Enzymes - proteins in action factors influencing activity. Nucleic acids – RNA - three types, structure and function DNA - structure and function Protein synthesis Teaching and Learning Strategies These may include the use of: interactive lessons incorporating audiovisual aids, discussion and debate, group work etc; practical investigations with an emphasis on the analysis and interpretation of data, and the presentation of results; use of model building, e.g. molymod kits, to gain an understanding of molecular structure; peer tutoring / student presentations / student as teacher; research with the aim to supplement learning outcomes in practical activities; Version 2 52 Board Endorsed October 2006 – Amended December 2013 Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Molymod Modelling Kit Suggested Text Harwood, R, 2002, Biochemistry, Cambridge University Press, Cambridge, Teacher Resources Aldridge, S. 1994, Biochemistry for advanced biology, Cambridge University Press, Cambridge. Green, N. et al. 1984, Biological science I chapters 5&6, Cambridge University Press , UK. James, M. 1992, Chemical connections book 2, Jacaranda Press, Sydney. Selinger, B. 1990, Chemistry in the market place, 4th ed. Harcourt Brace Jovanovich, Marrickville. Tooley, P. 1975, Experiments in applied chemistry, John Murray, London. Audio visual Material ABC video on DNA 2005. CD ROMS DNA Interactive DVD (PAL version), 2003, Howard Hughes Medical Institute, Cold Springs Harbour Laboratory and Windfall Films Ltd Version 2 53 Board Endorsed October 2006 – Amended December 2013 Biochemical Processes & Applications Value: 0.5 Prerequisites Minor in Chemistry Specific Unit Goals This unit should enable students to: outline the relationship between cell functions and structure, carry out simple experiments which demonstrate the control and management of chemical reactions; demonstrate safe handling of materials and equipment and work effectively as an individual or in a team; use information technology to facilitate the analysis and interpretation of results obtained in practical activities; gain an appreciation of the ethical issues involved in genetic engineering, and an understanding of the chemical processes involved. Content Cellular Respiration Emphasis should be on inputs and outputs, and location in cell of process, rather than the chemical detail of each reaction Glycolysis Krebs cycle Electron transfer chain Alternative pathways (anaerobic) Coupled reactions, energy outcomes Role of ATP, NADH, FADH2 and Coenzyme-A Photosynthesis Light and dark reactions The photo-systems, the Calvin Cycle Genetic Engineering DNA sequencing - the Sanger technique and its modern derivatives Electrophoresis Gene splicing Benefits of genetic engineering and ethical issues. Case studies of some Australian examples, e.g. cotton, canola, human insulin. Version 2 54 Board Endorsed October 2006 – Amended December 2013 Teaching and Learning Strategies These may include the use of: interactive lessons incorporating audiovisual aids, discussion and debate, group work etc; practical investigations with an emphasis on the analysis and interpretation of data, and the presentation of results; peer tutoring / student presentations / student as teacher; research with the aim to supplement learning outcomes in practical activities; a detailed written/oral presentation of a biochemical process studied; Visit to CSIRO Green Machine and/or ANU and/or UC to see DNA sequencing and other techniques which cannot be carried out in college laboratory. Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Books Aldridge, S. 1994, Biochemistry for advanced biology, Cambridge University Press, Cambridge. Green, N. et al. 1984, Biological science I chapters 5&6, Cambridge University Press, UK. James, M. 1992, Chemical connections book 2, The Jacaranda Press, Sydney. Selinger, B. 1990, Chemistry in the market place, 4th ed, Harcourt Brace Jovanovich, Marrickville. Tooley, P. 1975, Experiments in applied chemistry, John Murray, London. Audio visual Material Cellular Respiration – Concepts In Science Biology, 1984, TV Ontario Educational Communications Authority Photosynthesis – Concepts In Science Biology, 1984, TV Ontario Educational Communications Authority. Protein Synthesis – Concepts In Science Biology, 1984, TV Ontario Educational Communications. Genetic Engineering Education Kit, 1992, CSIRO. ABC video on DNA 2005 CD ROMS Version 2 55 Board Endorsed October 2006 – Amended December 2013 DNA Interactive DVD (PAL version) 2003, Howard Hughes Medical Institute, Cold Spring Harbour Laboratory and Windfall Films Ltd. Version 2 56 Board Endorsed October 2006 – Amended December 2013 Industrial Chemistry Value: 1.0 Prerequisites Minor in Chemistry Specific Unit Goals This unit should enable students to: design and carry out scientific investigations relating to industrial processes, and report on these investigations; record and analyse experimental data and use this data to further their experimental investigations; research, analyse, synthesise, evaluate and communicate relevant concepts and their applications in industry; organise and manipulate concepts and data to solve problems relevant to industrial chemistry Content Preparative chemistry Comparison between laboratory and industrial scale Emphasis on yield, purity, by products and error Stoichiometry and energy relationships extended Considerations for chemical industry Siting of industry Economic viability Environmental implications Overview of reactivity of metals and significance to their method of extraction Extraction of metals and electrolysis in industry Steel Copper Aluminium The Chlor-Alkali Industry Manufacture of Sulfuric acid Ammonia Nitric acid Petrochemicals especially ethene and its derivatives, including polymers. Choice of any one of the following: Food industry Ceramics Pharmaceuticals Agricultural Cosmetics Version 2 57 Board Endorsed October 2006 – Amended December 2013 Teaching and Learning Strategies Teaching and Learning Strategies may include the use of: experimental work simulating industrial processes and analytical methods; significant individual or small group research and experimentation, using a diary method of keeping track of student progress in their projects, with demonstration seminars; an excursion to relevant mining and/or other industrial sites and local factories; interactive lessons incorporating audiovisual aids, discussion and debate, group work etc. Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Books James, M. and Stokes, R. 1991 & 1992, Chemical connections VCE chemistry book 2, Jacaranda Press, Sydney. Selinger, B. 1998, Chemistry in the market place, 5th Ed. Harcourt Brace Jovanovich, Marrickville. Tooley, P. 1975, Experiments in applied chemistry, John Murray, London. CD ROMS Haber Process Advanced Version 1.1, 2001, O’Brien, M. Web sites www.newbyte.com These were accurate at the time of publication. Version 2 58 Board Endorsed October 2006 – Amended December 2013 Foundations of Industrial Chemistry Value: 0.5 Prerequisites Minor in Chemistry Specific Unit Goals This unit should enable students to: design and carry out scientific investigations relating to industrial processes, and report on these investigations; record and analyse experimental data and use this data to further their experimental investigations; research, analyse, synthesise, evaluate and communicate relevant concepts and their applications in industry; organise and manipulate concepts and data to solve problems relevant to industrial chemistry; Content Preparative chemistry Comparison between laboratory and industrial scale Emphasis on yield, purity, by products and error Stoichiometry, energy relationships extended Considerations for chemical industry Siting of industry Economic viability Environmental implications Overview of reactivity of metals and significance to their method of extraction Extraction of metals and electrolysis in industry Steel Copper Aluminium The Chlor-Alkali Industry Teaching and Learning Strategies Teaching and Learning Strategies may include the use of: experimental work simulating industrial processes and analytical methods; significant individual or small group research and experimentation, using a diary method of keeping track of student progress in their projects, with demonstration seminars; an excursion to relevant mining and/or other industrial sites and local factories; interactive lessons incorporating audiovisual aids, discussion and debate, group work etc. Assessment Refer to page 13. Version 2 59 Board Endorsed October 2006 – Amended December 2013 Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Books James, M. and Stokes, R., 1992, Chemical connections book 2 The Jacaranda Press, (particularly Chapters 6,7,8,12,17,18) Sydney. Selinger, B. 1990, Chemistry in the market place, 4th ed, Harcourt Brace Jovanovich, Marrickville. Tooley, P. 1975, Experiments in applied chemistry, John Murray, London. Version 2 60 Board Endorsed October 2006 – Amended December 2013 Investigation of Industrial Processes Value: 0.5 Prerequisites Minor in Chemistry Specific Unit Goals This unit should enable students to: design and carry out scientific investigations relating to industrial processes, and report on these investigations; record and analyse experimental data and use this data to further their experimental investigations; research, analyse, synthesise, evaluate and communicate relevant concepts and their applications in industry; organise and manipulate concepts and data to solve problems relevant to industrial chemistry; Content Manufacture of: Sulphuric acid Ammonia Nitric acid Petrochemicals especially ethene and its derivatives, including polymers. Choice of any one of the following: Food industry Ceramics Pharmaceuticals Agricultural Cosmetics Teaching and Learning Strategies Teaching and Learning Strategies may include the use of: experimental work simulating industrial processes and analytical methods; significant individual or small group research and experimentation, using a diary method of keeping track of student progress in their projects, with demonstration seminars; an excursion to relevant mining and/or other industrial sites and local factories; interactive lessons incorporating audiovisual aids, discussion and debate, group work. Assessment Refer to page 13. Version 2 61 Board Endorsed October 2006 – Amended December 2013 Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Books James, M. and Stokes, R. 1992, Chemical connections book 2 (particularly Chapters 6,7,8,12,17,18), The Jacaranda Press, Sydney. Selinger, B. 1990, Chemistry in the market place, 4th ed, Harcourt Brace Jovanovich, Marrickville. Tooley, P. 1975, Experiments in applied chemistry, John Murray, London. Version 2 62 Board Endorsed October 2006 – Amended December 2013 Environmental Chemistry Value: 1.0 Prerequisites Introductory Chemistry Specific Unit Goals This unit should enable students to: recognise the cyclic nature of matter and the fact that human interference causes changes to accelerate and that these changes are often cumulative, unpredictable and sometimes irreversible; analyse and evaluate conditions that affect an environment by trials and data collection from selected areas; understand that the management of resources and the environment demands a multidisciplinary approach and that knowledge of chemistry often plays an integral role in finding solutions to problems; carry out research to increase understanding of the complexities of the environment and use the findings to explain observed phenomena. Content The Environment definition components: land, air, water and the biomass cycling of matter in nature management of the environment Resources renewable and non-renewable properties: physical and chemical management of resources coal chemistry Pollution land, air and water Greenhouse, Acid Rain, Ozone Depletion waste management, recycling “clean” technologies Relevant Environmental Incidents e.g. Bhopal, Coode Is, Minamata, Chernobyl, Exxon Valdez, North Parkes Topics For Investigation (examples only) fertilizer, salinity, pH trials using suitable plant material and different soil types, water quality monitoring, industrial waste clean up, testing biodegradable material, use and misuse of surfactants, optimum conditions for composting organic material, energy and fuel efficiency, phosphates in water, heavy metals etc Version 2 63 Board Endorsed October 2006 – Amended December 2013 Teaching and Learning Strategies These may include the use of: interactive lessons incorporating audiovisual aids (with emphasis on the use of videos on current issues) to generate discussion and debate; collection and analysis of newspaper articles relevant to environmental issues; excursions to the local environment (eg Dick’s Creek) and industrial sites to witness chemical and management procedures first hand (eg Lower Molonglo Water Treatment Plant); a wide range of experiments and other investigations relating to a relevant topic and culminating in a major report; use of visiting experts eg from Waterwatch Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Books Australian Academy of Science 1994, Environmental science, Australian Academy of Science, Canberra. James, M. et al. 1991, Chemical connections book 1, Ch. 12, 14, 15, 18; book 2, Ch. 4, 6, 14, 22, Jacaranda Press, Sydney. Laidler, G. 1991, Environmental chemistry an Australian perspective, Longman Cheshire, South Melbourne. Audio visual Material Against Nature 1998, ABC Inside Story. Battle For The Planet 1989, Air Pollution. Rachel Carson’s Silent Spring 1993, WGBH Educational Foundation Peace River Films. The Blue Revolution 1989, BRNK & HTV. Ocean Planet 1989, BRNK & HTV. Curse of an Ancient Land – SALT (DVD) 2004, The Earth Resources Foundation, University of Sydney. Web Pages Version 2 64 Board Endorsed October 2006 – Amended December 2013 http://www.dfat.gov.au/geo/australia.aus_environ.html www.environment.gov.au www.environment.gov.au/ssg/bpem/database.html http://farrer.riv.csu.edu.au/ASGAP/weeds3.html www.greenhouse.crc.org.au www.science.org.au/nova/010/010box02.htm www.geocities.com/RainForest/5161/lab2.htm http://earthtrends.wri.org/ www.measurement.gov.au/index www.waterwatch.org.au/ http://www.landcareonline.com/ http://www.greenpeace.org.au/climate/solutions/renewables/biomass.html Version 2 65 Board Endorsed October 2006 – Amended December 2013 Principles of Environmental Chemistry Value: 0.5 Prerequisites Introductory Chemistry Specific Unit Goals This unit should enable students to: recognise the cyclic nature of matter and the fact that human interference causes changes to accelerate and that these changes are often cumulative, unpredictable and sometimes irreversible; analyse and evaluate conditions that affect an environment by trials and data collection from selected areas; understand that the management of resources and the environment demands a multidisciplinary approach and that knowledge of chemistry often plays an integral role in finding solutions to problems; carry out research to increase understanding of the complexities of the environment and use the findings to explain observed phenomena. Content The Environment definition components: land, air, water and the biomass cycling of matter in nature management of the environment Resources renewable and non-renewable properties: physical and chemical management of resources coal chemistry Pollution land, air and water Greenhouse, Acid Rain, Ozone Depletion waste management, recycling “clean” technologies Relevant Environmental Incidents e.g. Bhopal, Coode Is, Minamata, Chernobyl, Exxon Valdez, North Parkes Version 2 66 Board Endorsed October 2006 – Amended December 2013 Teaching and Learning Strategies These may include the use of: interactive lessons incorporating audiovisual aids (with emphasis on the use of videos on current issues) to generate discussion and debate; collection and analysis of newspaper articles relevant to environmental issues; excursions to the local environment (eg Dick’s Creek) and industrial sites to witness chemical and management procedures first hand (eg Lower Molonglo Water Treatment Plant); a wide range of experiments and other investigations relating to a relevant topic and culminating in a major report; use of visiting experts eg from Waterwatch Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Books Australian Academy of Science,1994, Environmental science, Australian Academy of Science, Canberra. James, M. et al. 1991, Chemical connections book 1, Ch. 12, 14, 15, 18. book 2, Ch. 4, 6, 14, 22, The Jacaranda Press, Sydney. Laidler, G. 1991, Environmental chemistry an australian perspective, Longman Cheshire, South Melbourne. Audio visual Material Against Nature 1998, ABC Inside Story. Battle For The Planet 1989, Air Pollution. Rachel Carson’s Silent Spring 1993, WGBH Educational Foundation Peace River Films. The Blue Revolution 1989, BRNK & HTV. Ocean Planet 1989, BRNK & HTV. Version 2 67 Board Endorsed October 2006 – Amended December 2013 Web Pages http://www.dfat.gov.au/geo/australia.aus_environ.html www.environment.gov.au www.environment.gov.au/ssg/bpem/database.html http://farrer.riv.csu.edu.au/ASGAP/weeds3.html www.greenhouse.crc.org.au www.science.org.au/nova/010/010box02.htm www.geocities.com/RainForest/5161/lab2.htm http://earthtrends.wri.org/ www.measurement.gov.au/index www.waterwatch.org.au/ http://www.landcareonline.com/ http://www.greenpeace.org.au/climate/solutions/renewables/biomass.html Version 2 68 Board Endorsed October 2006 – Amended December 2013 Investigation of Environmental Incidents Value: 0.5 Prerequisites Introductory Chemistry Specific Unit Goals This unit should enable students to: analyse and evaluate conditions that affect an environment by trials and data collection from selected areas; discuss that the management of resources and the environment demands a multidisciplinary approach and that knowledge of chemistry often plays an integral role in finding solutions to problems; carry out research to increase understanding of the complexities of the environment and use the findings to explain observed phenomena. Content Topics For Investigation (examples only) fertilizer, salinity, pH trials using suitable plant material and different soil types, water quality monitoring, industrial waste clean up, testing biodegradable material, use and misuse of surfactants, optimum conditions for composting organic material, energy and fuel efficiency, phosphates in water, heavy metals etc Teaching and Learning Strategies These may include the use of: interactive lessons incorporating audiovisual aids (with emphasis on the use of videos on current issues) to generate discussion and debate; collection and analysis of newspaper articles relevant to environmental issues; excursions to the local environment (eg Dick’s Creek) and industrial sites to witness chemical and management procedures first hand (eg Lower Molonglo Water Treatment Plant); a wide range of experiments and other investigations relating to a relevant topic and culminating in a major report; use of visiting experts eg from Waterwatch. Assessment Refer to page 13. Version 2 69 Board Endorsed October 2006 – Amended December 2013 Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Books Australian Academy of Science,1994, Environmental science, Australian Academy of Science, Canberra. James, M. et al. 1991, Chemical connections book 1, Ch. 12, 14, 15, 18; book 2, Ch. 4, 6, 14, 22, Jacaranda Press, Sydney. Laidler, G. 1991, Environmental chemistry an Australian perspective, Longman Cheshire, South Melbourne. Audio visual Material Against Nature 1998, ABC Inside Story. Battle For The Planet 1989, Air Pollution. Rachel Carson’s Silent Spring 1993, WGBH Educational Foundation Peace River Films. The Blue Revolution 1989, BRNK & HTV. Ocean Planet 1989, BRNK & HTV. Version 2 70 Board Endorsed October 2006 – Amended December 2013 Analytical Chemistry Value: 1.0 Prerequisites Minor in Chemistry Specific Unit Goals This unit should enable students to: understand the roles of analysis and the development of modern techniques; carry out both qualitative and quantitative analysis; understand advanced instrumentation using tertiary and commercial facilities; discuss the issues surrounding quality control and food testing; carry out research to further understand the complexities analytical chemistry and use the findings to explain observed phenomena; apply analytical techniques to examine problems for consumers and improve environmental safety Content Basic separation techniques paper and liquid chromatography electrophoresis Quantitative analysis accuracy and precision gravimetric techniques redox volumetric analysis back titrations Qualitative analysis analysis of ionic species by solubility and precipitation using various cations Basic spectroscopic techniques flame photometry spectroscopy redox volumetric analysis back titrations colorimetry IR and UV Advanced instrumentation fluorometry, AAS, HPLC, GC, NMR Version 2 71 Board Endorsed October 2006 – Amended December 2013 Teaching and Learning Strategies These may include the use of: collection of data to investigate identification of substances and detect errors and statistical trends; interactive lessons incorporating methods of analysis and detection; experimental investigations with an emphasis on the analysis and interpretation of raw data, and the presentation of results in the scientific method; use of analytical equipment , to gain an understanding of techniques used in industrial laboratories; use of data loggers and interpreting graphical data; group work to encourage team data collection; research and use second hand data to formulate an understanding of advanced analytical techniques; peer tutoring / student presentations / student as teacher; research with the aim to supplement learning outcomes in practical activities; a major research project on advanced analytical instrumentation and social/ethical implications for health and environment. Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Books Commons, C. et al. 1995, Chemistry 2, Rigby Heinemann, Melbourne. Sharwood, J. et al. 2000, Nelson chemistry, Nelson Thomson Learning, Victoria. McCarthy, A. 2002, Methods of analysis and detection, Cambridge University Press, Bichester. Version 2 72 Board Endorsed October 2006 – Amended December 2013 Resources A wide range of laboratory equipment and analytical equipment is viewed at institutions within the ACT. These include : Version 2 ANU University of Canberra Therapeutic Goods Administration CSIRO 73 Board Endorsed October 2006 – Amended December 2013 Introduction to Chemical Analysis Value: 0.5 Prerequisites Minor in Chemistry Specific Unit Goals This unit should enable students to: understand the roles of analysis; carry out both qualitative and quantitative analysis; understand advanced instrumentation using tertiary and commercial facilities; Content Basic separation techniques paper and liquid chromatography electrophoresis Quantitative analysis accuracy and precision gravimetric techniques redox volumetric analysis back titrations Qualitative analysis analysis of ionic species by solubility and precipitation using various cations Basic spectroscopic techniques flame photometry spectroscopy redox volumetric analysis back titrations Teaching and Learning Strategies These may include the use of: collection of data to investigate identification of substances and detect errors and statistical trends; interactive lessons incorporating methods of analysis and detection; experimental investigations with an emphasis on the analysis and interpretation of raw data, and the presentation of results in the scientific method; use of analytical equipment , to gain an understanding of techniques used in industrial laboratories; use of data loggers and interpreting graphical data; group work to encourage team data collection; research and use second hand data to formulate an understanding of advanced analytical techniques; Version 2 74 Board Endorsed October 2006 – Amended December 2013 peer tutoring / student presentations / student as teacher; research with the aim to supplement learning outcomes in practical activities. Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Books Commons, C. et al. 1995, Chemistry 2, Rigby Heinemann, Melbourne. Sharwood, J. et al. 2000, Nelson chemistry, Nelson Thomson Learning, Victoria. McCarthy, A. 2002, Methods of analysis and detection, Cambridge University Press, Bichester. Version 2 75 Board Endorsed October 2006 – Amended December 2013 Advanced Analytical Processes Value: 0.5 Prerequisites Minor in Chemistry Specific Unit Goals This unit should enable students to: discuss the issues surrounding quality control and food testing; carry out research to further understand the complexities analytical chemistry and use the findings to explain observed phenomena; apply analytical techniques to examine problems for consumers and improve environmental safety Content Basic spectroscopic techniques colorimetry IR and UV Advanced instrumentation fluorometry, AAS, HPLC, GC, NMR A major research project on an advanced analytical instrumentation and social/ethical implications for health and environment Teaching and Learning Strategies These may include the use of: collection of data to investigate identification of substances and detect errors and statistical trends; interactive lessons incorporating methods of analysis and detection; experimental investigations with an emphasis on the analysis and interpretation of raw data, and the presentation of results in the scientific method; use of analytical equipment , to gain an understanding of techniques used in industrial laboratories; use of data loggers and interpreting graphical data; group work to encourage team data collection; research and use second hand data to formulate an understanding of advanced analytical techniques; peer tutoring / student presentations / student as teacher; research with the aim to supplement learning outcomes in practical activities. Version 2 76 Board Endorsed October 2006 – Amended December 2013 Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Books Commons, C. et al. 1995, Chemistry 2, Rigby Heinemann, Melbourne. Sharwood, J. et al. 2000, Nelson chemistry, Nelson Thomson Learning, Victoria. McCarthy, A. 2002, Methods of analysis and detection, Cambridge University Press, Bichester. Version 2 77 Board Endorsed October 2006 – Amended December 2013 Forensic Chemistry Value: 1.0 Prerequisites Minor in Chemistry Specific Unit Goals This unit should enable students to: understand the role of the forensic chemist in helping crime investigators solve crimes demonstrate an understanding of the processes of chemical analysis of evidence and the security involved in maintaining the chain of custody design and carry out scientific investigations relating to chemical analysis of inorganic evidence materials demonstrate the safe handing of materials and equipment and work effectively as an individual or in a team understand the instrumentation used in forensic science laboratories Content In this unit, students will study the analysis of evidence as carried out in the forensic laboratory. The role of the forensic chemist Locard’s principle of forensic science need for accuracy, often only small samples, prevention of contamination collection, transport and storage of evidence chain of custody issues overview of the techniques and instruments used in the forensic laboratory destructive and non-destructive techniques qualitative and quantitative analysis Microscopy instruments available including the comparison and scanning electron microscopes use in comparison of trace evidence such as soils, paint layers, hairs and fibres Spectroscopy using light of different wavelengths to compare samples of soils, fibres, oils atomic emission spectroscopy (AES) Chemical analysis that involves cations and anions qualitative and quantitative analysis precipitation and solubility analysis volumetric and gravimetric techniques flame tests atomic absorption spectroscopy (AAS) mass spectrometry (MS) chromatography Analysis of soil samples mineral and organic matter analysis comparisons using microscopy and UV light, measurements of pH and density Comparison of glass samples Version 2 78 Board Endorsed October 2006 – Amended December 2013 chemical composition of different glass types density and refractive index measurements Hydrocarbons the different functional groups tests to distinguish the different groups infrared spectroscopy (IR) used to identify classes of drugs high performance liquid chromatography (HPLC) Proteins and amino acids chemical tests to identify the presence of proteins (Biurets) electrophoresis DNA profiling Analysis of body fluids the post mortem effect of drugs and poisons on the human body recognition and measurement of these chemicals to help identify the cause of death Arson and explosives study of fire and explosions chemicals that commonly used to accelerate a fire and how they are detected and identified chemicals and devices used to cause explosions and how they can be identified and lead investigators to a possible suspect Teaching and Learning Strategies These may include the use of: practical work to give experience and understanding in the analysis of unknown substances audio visual material to stimulate class discussion about how chemical analysis is used to solve crimes library and internet as a resource for an in-depth research of a modern chemical analysis technique used in the forensic laboratory collection and analysis of a newspaper article reporting a recent crime where chemical analysis was crucial in the solving of the crime critical analysis of a recent television episode in which chemical analysis of evidence contributed to the solving of the crime excursions and/or visiting speakers to enhance the student’s understanding of real life work in a modern forensic laboratory solving a hypothetical crime where the students are given chemical evidence that requires analysis Assessment Refer to page 13. Version 2 79 Board Endorsed October 2006 – Amended December 2013 Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Suggested Texts: Smith, R, 2005, Conquering chemistry HSC course, (Ch 13-15), McGraw-Hill Australia Pty Ltd, Sydney. Thickett, G. 2000, Chemistry pathways 1 and 2, (Forensic Chemistry supplement) Macmillan, Melbourne. Teacher Resources Saferstein, R. 2001, Criminalistics an introduction to forensic science, Prentice Hall, N J US. Lyle, D. 2004, Forensics for dummies, Wiley Publishing Inc, Indianapolis US. Lane, B. 1993, The encyclopaedia of forensic science, Hodder Headline PLC, London. Evans, C. 1996, The casebook of forensic detection, John Wiley & Sons, US. Owen, D. 2002, Police lab, New Burlington Books, London UK. Brown, M. and Wilson, P. 1992, Justice and nightmares, NSW University Press, Sydney. Genge, N. 2004, The forensic case book, Random House Australia, Sydney. Jolley, J. and Powrie, J. 2000, Forensic science for high schools book 1, Emerald City Press, Glebe, NSW. Powrie, J. and Jolley, J. 2000, Forensic science for high schools book 2, Emerald City Press, Glebe NSW. Jolley, J. and Powrie, J. 2003, Forensic science for high schools book 3, Learning Essentials, Malvern Vic. Powrie, J. and Jolley, J. 2003, Forensic science for high schools book 4, Learning Essentials, Malvern Vic. Jolley, J. Forensic chemistry books 1 & 2, Blake Education (to be published in 2007), Sydney. Version 2 80 Board Endorsed October 2006 – Amended December 2013 Journal Articles Relevant articles from ChemMatters, Chemistry Review and The Australian Journal of Forensic Sciences. Audio visual Material A Case of Murder, 1997, Learning Essentials Clayton South, Victoria The New Detective Series: Learning Essentials Clayton South, Victoria Infallible Witness, Shreds of Evidence, Double Helix Video Education Australia video(s) coming on Forensic Chemistry in 2007 Web sites Reddy’s Forensic Page, most forensic applications covered, Chamakura, Reddy http://www.forensicpage.com/ National Institute of Forensic Science (Australia) home page. excellent resources and links http://www.nifs.com.au/ Zeno’s Forensic Site, covers most aspect forensic chemistry, Geradts, Dr Zeno http://forensic.to/forensic.html Home page of National Association of High school Teachers of Forensic Science (USA) http://www.hstofs.org/ Website that gives a large number of forensic sites suitable for teachers and students http://www.shambles.net/pages/learning/ScienceP/forensic/ These were accurate at the time of publication. Resources Australian Federal Police Weston Canberra. University of Canberra. Canberra Institute of Technology (Bruce campus). Version 2 81 Board Endorsed October 2006 – Amended December 2013 Principles & Processes of Forensics Value: 0.5 Prerequisites Minor in Chemistry Specific Unit Goals This unit should enable students to: understand the role of the forensic chemist in helping crime investigators solve crimes demonstrate an understanding of the processes of chemical analysis of evidence and the security involved in maintaining the chain of custody design and carry out scientific investigations relating to chemical analysis of inorganic evidence materials demonstrate the safe handing of materials and equipment and work effectively as an individual or in a team understand the instrumentation used in forensic science laboratories Content In this unit, students will study the analysis of evidence as carried out in the forensic laboratory The role of the forensic chemist Locard’s principle of forensic science need for accuracy, often only small samples, prevention of contamination collection, transport and storage of evidence chain of custody issues overview of the techniques and instruments used in the forensic laboratory destructive and non-destructive techniques qualitative and quantitative analysis Microscopy instruments available including the comparison and scanning electron microscopes use in comparison of trace evidence such as soils, paint layers, hairs and fibres Spectroscopy using light of different wavelengths to compare samples of soils, fibres, oils atomic emission spectroscopy (AES) Chemical analysis that involves cations and anions qualitative and quantitative analysis precipitation and solubility analysis volumetric and gravimetric techniques flame tests atomic absorption spectroscopy (AAS) Analysis of soil samples mineral and organic matter analysis comparisons using microscopy and UV light, measurements of pH and density Comparison of glass samples chemical composition of different glass types density and refractive index measurements Version 2 82 Board Endorsed October 2006 – Amended December 2013 Teaching and Learning Strategies These may include the use of: practical work to give experience and understanding in the analysis of unknown substances; audio visual material to stimulate class discussion about how chemical analysis is used to solve crimes; library and internet as a resource for an in-depth research of a modern chemical analysis technique used in the forensic laboratory; collection and analysis of a newspaper article reporting a recent crime where chemical analysis was crucial in the solving of the crime; critical analysis of a recent television episode in which chemical analysis of evidence contributed to the solving of the crime; excursions and/or visiting speakers to enhance the student’s understanding of real life work in a modern forensic laboratory; solving a hypothetical crime where the students are given chemical evidence that requires analysis. Assessment Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Suggested Texts: Smith, R, 2005, Conquering chemistry HSC Course, (Ch 13-15), McGraw-Hill Australia Pty Ltd, Sydney. Thickett, G, 2000, Chemistry pathways 1 and 2, (Forensic Chemistry supplement) Macmillan, Melbourne. Teacher Resources Saferstein, R. 2001, Criminalistics an introduction to forensic science, Prentice Hall, N J US. Lyle, D. 2004, Forensics for dummies, Wiley Publishing Inc, Indianapolis US. Lane, B. 1993, The encyclopaedia of forensic science, Hodder Headline PLC, London. Evans, C. 1996, The casebook of forensic detection, John Wiley & Sons, US. Owen, D. 2002, Police lab, New Burlington Books, London UK. Brown, M. and Wilson, P. 1992, Justice and nightmares, NSW University Press, Sydney. Version 2 83 Board Endorsed October 2006 – Amended December 2013 Genge, N. 2004, The forensic case book, Random House Australia, Sydney. Jolley, J. and Powrie, J. 2000, Forensic science for high schools book 1, Emerald City Press, Glebe, NSW. Powrie, J. and Jolley, J. 2000, Forensic science for high schools book 2, Emerald City Press, Glebe NSW. Jolley, J. and Powrie, J. 2003, Forensic science for high schools book 3, Learning Essentials, Malvern Vic. Powrie, J. and Jolley, J. 2003, Forensic science for high schools book 4, Learning Essentials, Malvern Vic. Jolley, J. Forensic chemistry books 1 & 2, Blake Education (to be published in 2007), Sydney. Jolley, J. and Powrie, J. 2003, Forensic science for high schools book 3, Learning Essentials, Malvern Vic. Powrie, J. and Jolley, J. 2003, Forensic science for high schools book 4, Learning Essentials, Malvern Vic. Jolley, J. Forensic chemistry books 1 & 2, Blake Education (to be published in 2007), Sydney. Journal Articles Relevant articles from ChemMatters, Chemistry Review and The Australian Journal of Forensic Sciences. Audio visual Material Learning Essentials video on fibres and DNA. Video Education Australia video(s) coming on Forensic Chemistry in 2007. Web sites Reddy’s Forensic Page, most forensic applications covered, Chamakura, Reddy http://www.forensicpage.com/ National Institute of Forensic Science (Australia) home page. excellent resources and links http://www.nifs.com.au/ Zeno’s Forensic Site, covers most aspect forensic chemistry, Geradts, Dr Zeno http://forensic.to/forensic.html Home page of National Association of High School Teachers of Forensic Science (USA) http://www.hstofs.org/ Website that gives a large number of forensic sites suitable for teachers and students http://www.shambles.net/pages/learning/ScienceP/forensic/ These were accurate at the time of publication. Version 2 84 Board Endorsed October 2006 – Amended December 2013 Resources Australian Federal Police Weston Canberra. University of Canberra. Canberra Institute of Technology (Bruce campus). Version 2 85 Board Endorsed October 2006 – Amended December 2013 Electrochemistry Value 0.5 Prerequisites Minor in Chemistry Specific Unit Goals This unit should enable students to: further develop knowledge of redox reactions, including corrosion and its electrochemical nature and prevention techniques; develop an understanding of theoretical and practical issues in the design of devices that produce electricity from chemical reactions. develop an awareness of industrial electrolytic processes in production of metals (e.g. aluminium, halogens (e.g. chlorine) and sodium hydroxide. develop an appreciation of some of the issues encountered within the chemical industry, particularly that part of it based on chlorine production. Content Review of : Oxidation/Reduction reactions & redox equations Electrochemical cells & the electrochemical series Primary & secondary cells: design features of those currently available and being developed. Fuel cells: current designs, future applications. Issues in developing a hydrogen economy. Electrolytic cells: prediction of products at electrodes, application of Faraday’s Laws Industrial electrolytic processes: e.g. production of aluminium, chlorine, sodium hydroxide The chlorine-based chemical industry: its products and environmental impacts. Teaching and Learning Strategies Teaching and learning strategies may include the use of detailed practical analysis of electrochemical cells accompanied by chemical reactions and explanations; audio visual material to stimulate class discussion about the importance of redox reactions in technologies of great importance to the future; the library as a source for more in-depth research into the environmental impact of the topics studied in this unit; excursions and visiting speakers complemented by library research (including information retrieval using CD-ROMs and the internet); interactive lessons incorporating audiovisual aids, discussion and debate and oral presentations by students. electronic probe data logging with computer-assisted graphing of results; Assessment Version 2 86 Board Endorsed October 2006 – Amended December 2013 Refer to page 13. Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members RESOURCES Aylward, G. and. Findlay, T, 1994, SI chemical data, John Wiley & Sons, Milton QLD. Budavari, S (ed) 1996 The Merck Index – An encyclopaedia of chemicals, drugs & botanicals, 12th ed, Merck & Co, Whitehouse Station NJ US. Hill, G. and Holman, J. 1989, Chemistry in context, Nelson, Melbourne. Selinger, B. 1998, Chemistry in the market place, 5th ed. Harcourt Brace Jovanovich, Marrickville. Sharwood, .J. et al. 2000, Nelson chemistry VCE units 3&4, Nelson, Melbourne. Weast, R. (ed) 1979-1980, CRC handbook of chemistry & physics, 60th ed, The Chemical Rubber Company, Cleveland US. Journals Choice Magazine New Scientist Videos Primary Cells, Secondary Cells and Fuel Cells 1997, (videorecording), Marcom Projects, Loganholme Queensland. Salt of the Earth 1990, (videorecording)/David Suzuki CBC Television, Toronto, Canada. The Making of Aluminium 1993, (Advanced Version) (videorecording) Video Education Australasia, Bendigo, Victoria. The Chlor-Alkali Industry 1995, (videorecording) Granada, London UK. Version 2 87 Board Endorsed October 2006 – Amended December 2013 Extended Research Value: 0.5 Prerequisites Students undertaking this unit should have demonstrated a high degree of interest in, and aptitude for, Chemistry with at least a minor in the subject. Specific Unit Goals This unit should enable students to: demonstrate a high degree of initiative in the planning and organisation of a major piece of work in a field of chemistry; work independently and negotiate appropriate parameters for the study undertaken, including the emphasis that should be placed on the impact of chemistry on the environment and the ensuing social and economic consequences; collect and process data showing a high level of analytical and interpretive skills, while taking responsibility for the safe handling of experimental material and equipment when investigating the chemistry related to their topic; clearly communicate an understanding of the chemical concepts underpinning the topic. Content This unit enables students to study a topic of interest to them in greater depth than is possible in the other units. Negotiated content should reflect the goals of the unit. Some suggested approaches are: an original/creative exercise based on a student’s initiative and research; a review of current research in an area of interest. This should involve not only library research but also discussions/interviews with scientists working in that area; conduct a series of experiments in an area of interest, combined with library research and culminating in a paper analysing and discussing the results. Teaching and Learning Strategies Teaching strategies may include the use of: mentoring student progress through review of weekly work goals; facilitating communication with scientists in the community and the use of their facilities; negotiating the scope for the study, deadlines for work, and assessment weighting; developing seminar presentation skills to include the use of IT and audio visual techniques to maximise effective communication to an audience. Assessment Refer to page 13. Version 2 88 Board Endorsed October 2006 – Amended December 2013 Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners Specific Unit Resources Books Budavari, S (Ed) 1996, The Merck Index – an encyclopaedia of chemicals, drugs & botanicals, 12th ed, Merck & Co, Whitehouse Station NJ US. Weast, R. (Ed) CRC handbook of chemistry & physics – a ready reference book of chemical & physical data, The Chemical Rubber Company, Cleveland Ohio US. Tooley, P. 1975, Experiments in applied chemistry, John Murray, London.Teacher Resource Wessex Project Version 2 89 Board Endorsed October 2006 – Amended December 2013 Biochemical Structures & Analysis Value: 1.0 This unit combines Biochemical Structures 0.5 and Introduction to Chemical Analysis 0.5 Prerequisites Minor in Chemistry Specific Unit Goals This unit should enable students to: outline the relationship between cell functions and structure, understand the structure of biomolecules, their identification and the relationship between structure and function; carry out simple experiments which demonstrate the control and management of chemical reactions; use scientific investigations to understand the complexity of processes in living organisms at a molecular level, both qualitatively and, where possible, quantitatively; demonstrate safe handling of materials and equipment and work effectively as an individual or in a team. understand the roles of analysis; carry out both qualitative and quantitative analysis; understand advanced instrumentation using tertiary and commercial facilities; Content Introduction to Biochemistry Cell structure and function Biomolecules Revision of functional groups Monomers and polymers Saccharides - monosaccharides, open and ring structure - disaccharides, bonding between rings - polysaccharides – chains and cross-linking. Lipids - fatty acids, triglycerides, phosphoglycerides, lipid bi-layers, and simple steroid structure. Amino acids Proteins and their structure, - primary, secondary, tertiary and quaternary Enzymes - proteins in action factors influencing activity. Nucleic acids – RNA - three types, structure and function DNA - structure and function Protein synthesis Basic separation techniques paper and liquid chromatography electrophoresis Quantitative analysis Version 2 90 Board Endorsed October 2006 – Amended December 2013 accuracy and precision gravimetric techniques redox volumetric analysis back titrations Qualitative analysis analysis of ionic species by solubility and precipitation using various cations Basic spectroscopic techniques flame photometry spectroscopy redox volumetric analysis back titrations Teaching and Learning Strategies These may include the use of: interactive lessons incorporating audiovisual aids, discussion and debate, group work etc; practical investigations with an emphasis on the analysis and interpretation of data, and the presentation of results; use of model building, e.g. molymod kits, to gain an understanding of molecular structure; peer tutoring / student presentations / student as teacher; research with the aim to supplement learning outcomes in practical activities; collection of data to investigate identification of substances and detect errors and statistical trends; interactive lessons incorporating methods of analysis and detection; experimental investigations with an emphasis on the analysis and interpretation of raw data, and the presentation of results in the scientific method; use of analytical equipment , to gain an understanding of techniques used in industrial laboratories; use of data loggers and interpreting graphical data; group work to encourage team data collection; research and use second hand data to formulate an understanding of advanced analytical techniques; peer tutoring / student presentations / student as teacher; research with the aim to supplement learning outcomes in practical activities. Assessment Refer to page 13. Version 2 91 Board Endorsed October 2006 – Amended December 2013 Student Capabilities Evidence could be in: Student Capabilities Goals Content Teaching Assessment creative and critical thinkers enterprising problem-solvers skilled and empathetic communicators informed and ethical decision-makers environmentally and culturally aware citizens confident and capable users of technologies independent and self-managing learners collaborative team members Specific Unit Resources Molymod Modelling Kit Books Commons, C. et al. 1995, Chemistry 2, Rigby Heinemann, Melbourne. Sharwood, J. et al. 2000, Nelson chemistry, Nelson Thomson Learning, Victoria. McCarthy, A. 2002, Methods of analysis and detection, Cambridge University Press, Bichester. Suggested Text Harwood, R, 2002, Biochemistry, Cambridge University Press, Cambridge, Teacher Resources Aldridge, S. 1994, Biochemistry for advanced biology, Cambridge University Press, Cambridge. Green, N. et al. 1984, Biological science I chapters 5&6, Cambridge University Press , UK. James, M. 1992, Chemical connections book 2, Jacaranda Press, Sydney. Selinger, B. 1990, Chemistry in the market place, 4th ed. Harcourt Brace Jovanovich, Marrickville. Tooley, P. 1975, Experiments in applied chemistry, John Murray, London. Audio visual Material ABC video on DNA 2005. CD ROMS DNA Interactive DVD (PAL version), 2003, Howard Hughes Medical Institute, Cold Springs Harbour Laboratory and Windfall Films Ltd Version 2 92 Board Endorsed October 2006 – Amended December 2013 Appendix A - Australian Curriculum Achievement Standards Chemistry Units 1 and 2 (T) A B Chemistry concepts, models and applications Chemistry inquiry skills For the chemical systems studied, the student: analyses how structure, bond strength and energy transfers and transformations are interrelated in chemical systems analyses how a range of factors affect atomic or molecular interactions and change the structure and properties of systems explains the theories and model/s used to explain the system and the aspects of the system they include applies theories and models of systems and processes to explain phenomena, interpret complex problems, and make reasoned, plausible predictions in unfamiliar contexts For the chemical science contexts studied, the student: analyses the roles of collaboration, debate and review, and technologies, in the development of chemical science theories and models evaluates how chemical science has been used in concert with other sciences to meet diverse needs and inform decision making, and how these applications are influenced by interacting social, economic and ethical factors For the chemical systems studied, the student explains how structure, bonding and energy transfers and transformations are interrelated in chemical systems explains how a range of factors change the structure and properties of chemical systems describes the theories and model/s used to explain the system applies theories and models of systems and processes to explain phenomena, interpret problems, and make plausible predictions in unfamiliar contexts For the chemical science contexts studied, the student: explains the role of collaboration, debate and review, and technologies, in the development of chemical science theories and models explains how chemical science has been used to meet diverse needs and inform decision making, and how these applications are influenced by social, economic and ethical factors For the chemical science contexts studied, the student: designs, conducts and improves safe, ethical investigations that efficiently collect valid, reliable data in response to a complex question or problem analyses data sets to explain causal and correlational relationships, the reliability of the data, and sources of error justifies their selection of data as evidence, analyses evidence with reference to models and/or theories, and develops evidence-based conclusions that identify limitations evaluates processes and claims, and provides an evidence-based critique and discussion of improvements or alternatives selects, constructs and uses appropriate representations to describe complex relationships and solve complex and unfamiliar problems communicates effectively and accurately in a range of modes, styles and genres for specific audiences and purposes Version 2 For the chemical science contexts studied, the student: designs, conducts and improves safe, ethical investigations that collect valid, reliable data in response to a question or problem analyses data sets to identify causal and correlational relationships, anomalies, and sources of error selects appropriate data as evidence, interprets evidence with reference to models and/or theories, and provides evidence for conclusions evaluates processes and claims, provides a critique with reference to evidence, and identifies possible improvements or alternatives selects, constructs and uses appropriate representations to describe complex relationships and solve unfamiliar problems communicates clearly and accurately in a range of modes, styles and genres for specific audiences and purposes 93 Board Endorsed October 2006 – Amended December 2013 Chemistry Units 1 and 2 (continued) (T) C D E Chemistry concepts, models and applications Chemistry inquiry skills For the chemical systems studied, the student: describes how structure, bonding and energy transfers are related in chemical systems describes how some factors change the structure and properties of chemical systems describes a theory or model used to explain the system applies theories or models of systems and processes to explain phenomena, interpret problems, and make plausible predictions in familiar contexts For the chemical science contexts studied, the student: describes the role of collaboration and review, and technologies, in the development of chemical science theories or models discusses how chemical science has been used to meet needs and inform decision making, and some social, economic or ethical implications of these applications For the chemical systems studied, the student: describes structure and bonding in substances describes how some factors affect chemical systems identifies aspects of a theory or model related to the system describes phenomena, interprets simple problems, and makes simple predictions in familiar contexts For the chemical science contexts studied, the student: describes the role of communication and new evidence in developing chemical science knowledge describes ways in which chemical science has been used in society to meet needs, and identifies some implications of these applications For the chemical systems studied, the student: identifies observable properties of substances identifies observable changes to chemical systems identifies aspects of a theory or model related to parts of the system describes phenomena and makes simple predictions in familiar, simple contexts For the chemical science contexts studied, the student: identifies that chemical science knowledge has changed over time identifies ways in which chemical science has been used in society to meet needs For the chemical science contexts studied, the student: designs and conducts safe, ethical investigations that collect valid data in response to a question or problem analyses data to identify relationships, anomalies, and sources of error selects data to demonstrate relationships linked to chemical science knowledge, and provides conclusions based on data evaluates processes and claims, and suggests improvements or alternatives selects, constructs and uses appropriate representations to describe relationships and solve problems communicates clearly in a range of modes, styles and genres for specific purposes Version 2 For the chemical science contexts studied, the student: plans and conducts safe, ethical investigations to collect data in response to a question or problem analyses data to identify trends and anomalies selects data to demonstrate trends, and presents simple conclusions based on data considers processes and claims from a personal perspective constructs and uses simple representations to describe relationships and solve simple problems communicates in a range of modes and genres For the chemical science contexts studied, the student: follows a procedure to conduct safe, ethical investigations to collect data identifies trends in data selects data to demonstrate trends considers claims from a personal perspective constructs and uses simple representations to describe phenomena communicates in a range of modes 94 Board Endorsed October 2006 – Amended December 2013 Chemistry Units 3 and 4 (T) A B Chemistry concepts, models and applications Chemistry inquiry skills For the chemical systems studied, the student: analyses how a range of interrelated factors affect atomic and molecular interactions and change the structure, properties and dynamics of chemical systems analyses how interactions between matter and energy in complex chemical systems can be designed, monitored and controlled to produce desired outcomes explains the theories and model/s used to explain the system, the supporting evidence, and their limitations and assumptions applies theories and models of systems and processes to explain phenomena, critically analyse complex problems, and make reasoned, plausible predictions in unfamiliar contexts For the chemical science contexts studied, the student: analyses the roles of collaboration, debate and review, and technologies, in the development of chemical science theories and models evaluates how chemical science has been used in concert with other sciences to meet diverse needs and inform decision making, and how these applications are influenced by interacting social, economic and ethical factors For the chemical systems studied, the student: explains how a range of interrelated factors change the structure, properties and dynamics of chemical systems explains how interactions between matter and energy in chemical systems can be designed, monitored and controlled to produce desired outcomes describes the theories and model/s used to explain the system, some supporting evidence, and their limitations applies theories and models of systems and processes to explain phenomena, analyse problems, and make plausible predictions in unfamiliar contexts For the chemical science contexts studied, the student: explains the roles of collaboration, debate and review, and technologies, in the development of chemical science theories and models explains how chemical science has been used to meet diverse needs and inform decision making, and how these applications are influenced by social, economic and ethical factors For the chemical science contexts studied, the student: designs, conducts and improves safe, ethical investigations that efficiently collect valid, reliable data in response to a complex question or problem analyses data sets to explain causal and correlational relationships, the reliability of the data, and sources of error justifies their selection of data as evidence, analyses evidence with reference to models and/or theories, and develops evidence-based conclusions that identify limitations evaluates processes and claims, and provides an evidence-based critique and discussion of improvements or alternatives selects, constructs and uses appropriate representations to describe complex relationships and solve complex and unfamiliar problems communicates effectively and accurately in a range of modes, styles and genres for specific audiences and purposes Version 2 For the chemical science contexts studied, the student: designs, conducts and improves safe, ethical investigations that collect valid, reliable data in response to a question or problem analyses data sets to identify causal and correlational relationships, anomalies, and sources of error selects appropriate data as evidence, interprets evidence with reference to models and/or theories, and provides evidence for conclusions evaluates processes and claims, provides a critique with reference to evidence, and identifies possible improvements or alternatives selects, constructs and uses appropriate representations to describe complex relationships and solve unfamiliar problems communicates clearly and accurately in a range of modes, styles and genres for specific audiences and purposes 95 Board Endorsed October 2006 – Amended December 2013 Chemistry Units 3 and 4 (continued) (T) C D E Chemistry concepts, models and applications Chemistry inquiry skills For the chemical systems studied, the student: explains how a range of factors change the structure, properties and dynamics of chemical systems describes how chemical systems are controlled and monitored to produce desired outcomes describes key aspects of a theory or model used to explain system processes, and the phenomena to which those processes can be applied applies theories or models of systems and processes to explain phenomena, interpret problems, and make plausible predictions in some unfamiliar contexts For the chemical science contexts studied, the student: describes the roles of collaboration and review, and technologies, in the development of chemical science theories or models discusses how chemical science has been used to meet needs and inform decision making, and some social, economic or ethical implications of these applications For the chemical systems studied, the student: describes how some factors affect the properties of chemical systems describes how chemical systems are manipulated to produce desired outcomes describes key aspects of a theory or model used to explain a system process describes phenomena, interprets simple problems, and makes predictions in familiar contexts For the chemical science contexts studied, the student: describes the roles of communication and new evidence in developing chemical science knowledge describes ways in which chemical science has been used in society to meet needs, and identifies some implications of these applications For the chemical systems studied, the student: describes changes to chemical systems describes how chemical systems are used to produce desired outcomes identifies aspects of a theory or model related to a system process describes phenomena and makes simple predictions in familiar contexts For the chemical science contexts studied, the student: identifies that chemical science knowledge has changed over time identifies ways in which chemical science has been used in society to meet needs For the chemical science contexts studied, the student: designs and conducts safe, ethical investigations that collect valid data in response to a question or problem analyses data to identify relationships, anomalies, and sources of error selects data to demonstrate relationships linked to chemical science knowledge, and provides conclusions based on data evaluates processes and claims, and suggests improvements or alternatives selects , constructs and uses appropriate representations to describe relationships and solve problems communicates clearly in a range of modes, styles and genres for specific purposes Version 2 For the chemical science contexts studied, the student: plans and conducts safe, ethical investigations to collect data in response to a question or problem analyses data to identify trends and anomalies selects data to demonstrate trends, and presents simple conclusions based on data considers processes and claims from a personal perspective constructs and uses simple representations to describe relationships and solve simple problems communicates in a range of modes and genres For the chemical science contexts studied, the student: follows a procedure to conduct safe, ethical investigations to collect data identifies trends in data selects data to demonstrate trends considers claims from a personal perspective constructs and uses simple representations to describe phenomena communicates in a range of modes 96