Solvent KIE of hydrolysis - Chemistry Courses: About
advertisement

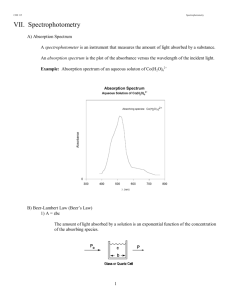
Kinetic Isotope Effects On the Hydrolysis of Vinyl Ethers Purpose: To investigate the mechanism of vinyl ether hydrolysis using KIE Experimental overview Acid-catalyzed hydrolysis of a vinyl ether is conducted using HCl and DCl The kinetic data is determined by following the reaction with UV spectroscopy The reaction is run under various acid concentrations to determine the pseudo-first order rate constant Procedures: Titration of stock acid solutions Your team will need to determine the acid concentration of the stock HCl solution. (The molarity of the stock DCl solution will be given in the lab.) The solution is approximately 0.1 M, but you need to determine the concentrations accurately. Write your own procedure (a good source would be a general chemistry labbook) and show it to your AI before proceeding. Make a series of acid reaction solutions for your experiment You should make four dilute HCl reaction solutions from the stock solution using a volumetric flask. Be as precise as possible! Fill in this table: Reaction Solution A B C D Total volume (mL) 10.00 10.00 10.00 10.00 Stock solution added (mL) 1.00 2.00 3.00 4.00 Molarity of Stock Molarity of Reaction solution The DCl reaction solutions E-H have been made for you in a way similar to that above. You will be given the concentration of the stock solution of DCl; calculate the molarities of the reaction solutions. Reaction Solution E F G H Total volume (mL) 10.00 10.00 10.00 10.00 Stock solution added (mL) 1.00 2.00 3.00 4.00 Molarity of Stock Molarity of Reaction solution Make a stock solution of ethyl vinyl ether (EVE) The EVE stock should be made by dissolving EVE in methanol (50% by volume.) How much should you make? (Consider your experiment—don’t generate an undo amount of waste!) Check with the AI with your calculations before proceeding. Kinetics experiment Set up the spectrometer to obtain kinetic data at 225nm. To do this, select “Basic Application” then “Absorbance”, and enter a wavelength of 225 nm. You are going to do kinetics the old fashioned way—you will need a stop watch, paper and pen. To a quartz cuvette, add 3.00 mL of acid solution A. Inject 0.02 mL of EVE solution into the cuvette, cover and shake quickly, and insert into the spectrometer. Have one team member keep time and another record data. At short time intervals (every few seconds, call out the absorbance so that it can be recorded. Obtain data until there is no discernible change in the absorbance. (CONSIDER THIS: The only problem with collecting more data than needed is that it will use time. Collecting too little data will require the experiment to be repeated.) Repeat the experiment for solutions B through H. Put your data into an Excel sheets, and use Excel to work up your data. Results and Discussion: To determine the KIE for this reaction, you will need a rate constant with H3O+. Calculate the first order rate constant for solutions A-D. Review your class notes as a reminder of how to do this. You will then take an average of these data for the rate constant with protonated acid. Do the same with Solutions E-H for deuterated acid. Consider: Which figure(s) and table(s) would be necessary in your formal reports/presentations? Your main purpose in this lab is to propose a mechanism that has some experimental support. You will probably want to propose two or more mechanisms, or (ideally) a range of mechanisms using MOFJ, and show how your experimental data is consistent or inconsistent with each mechanism.