Protocol for HIV-1 near full-length genome amplification
advertisement

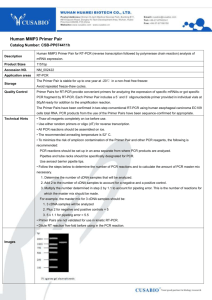
Protocol for HIV-1 near full-length genome amplification and sequencing from plasma RNA 1. RNA extraction 2. Amplification in four overlapping fragments by RT-PCR+nested PCR 3. Checking PCR amplification 4. Purification of PCR products 5. Sequencing 1. RNA Extraction RNA is extracted from 1 ml plasma with NucliSENS® kit (bioMérieux, France), following the manufacturer’s instructions. 2. Amplification in four overlapping fragments by RT-PCR and nested PCR Amplification of the HIV-1 genome is done in four overlapping fragments (A, B, C, and D, with sizes of 2 kb, 3 kb, 2.9 kb, and 1.8 kb, respectively) by RT-PCR followed by nested PCR using the following reaction mixtures RT-PCR H2O (DNAse, RNAse free) Buffer BioTaq DNA Polymerase, 10X MgCl2 (50 mM) dNTP mix (20 mM) (GE Healthcare) Primer sense (20 µM) Primer antisense (20µM) rRNasin RNase inhibitor (40 U/µl) (Promega) SuperScript III RT (200 U/µl) (Invitrogen) BioTaq DNA polymerase (5 U/µl) (Bioline) Pfu DNA polymerase (3 U/µl) (Stratagene) Premix Vol. RNA sample Final Vol. Nested PCR X1 14.775 µl 2.5 µl 1 µl 0.25 µl 0.5 µl 0.5 µl 0.125 µl X ___ H2O (DNAse, RNAse free) Buffer BioTaq DNA Polymerase, 10X MgCl2 (50 mM) dNTP mix (20 mM) (GE Healthcare) Primer sense (20 µM) Primer antisense (20 µM) X1 37.9 µl 5 µl 2 µl 0.5 µl 1 µl 1 µl 0.05 µl 0.25 µl 0.5 µl 20 µl BioTaq DNA polymerase (5U/µl) (Bioline) Pfu DNA polymerase (3U/µl) (Stratagene) Premix Vol. 5 µl 25 µl RT-PCR product Final Vol. 2 µl 50 µl 0.05 µl 0.1 µl 48 µl X ___ PCR primers RT-PCR Fragment A B C D Sense Antisense Nested PCR Sense Antisense 5’R1 PROS2b 3’HIV-OS3 gp41-nefOS-2 PROA Pol OA1 gp120-OA Nef-AS-4 5’RU5S PRTS 3’HIV-OS gp120S PRTA 5’HIV-NA gp41-AS 3’Nef-3 Primers’ sequences and their positions in the HXB2 reference isolate are shown in table 1. Thermocycling profiles RT-PCR: RT: 55ºC, 30min Denaturation: 94ºC, 3min 35 PCR cycles: - Denaturation: 94ºC, 30sec - Annealing: 55ºC, 30sec - Elongation: 72ºC, 3 min Final elongation: 72ºC, 7 min Hold 4ºC Nested-PCR: Denaturation: 94ºC, 3min 35 PCR cycles: - Denaturation: 94ºC, 30sec - Annealing: 55ºC, 30 sec - Elongation: 72ºC, 3 min Final elongation: 72ºC, 7 min Hold 4ºC Although the lengths of amplified fragment are different, the four PCRs can be performed together with the same thermocycling profile, using 3 min in the elongation step. If done separately, 2 min elongation can be used for fragments A and D, and 3 min elongation for fragments B and C. 3. Checking amplification and fragment length Check the amplification and the length of the amplified fragments by electrophoresis in a 1% agarose gel. 4. Enzymatic purification of PCR products Reaction mix: 1 µl PCR product 1 µl shrimp alkaline phosphatase (SAP) (1 U/µl) (GE Healthcare) 0.1 µl exonuclease I (10 U/µl) (GE Healthcare) 4.9 SAP buffer 5x (dilute 1:2 10x buffer) Incubate at 37ºC, 15 minutes, followed by enzyme inactivation at 80ºC, 15 minutes. 5. Sequencing Sequencing reaction mix: 5.9 µl H2O (DNAse free) 1.6 µl primer (2 µM) 1.0 µl BigDye terminator cycle sequencing kit v3.1 (Applied Biosystems) 1.5 µl BigDye buffer 1 µl PCR product (previously purified) Thermocycling profile: 94ºC, 2 min; 25 cycles: 94ºC, 30 sec; 50ºC, 15 sec; 60ºC, 4 min. Sequencing primers, with their sequences and positions in the HXB2 reference strain, are listed in table 1. The table includes alternative sequencing primers in case that good quality electropherograms are not obtained with first-choice primers. In case that the sequence is illegible in some fragment, due to length polymorphisms, end-point dilution RT-PCR of the corresponding fragment can be done (RT-PCR/nested PCR is done with serial 1:2 RNA dilutions, and sequencing is done with the PCR product obtained with the highest dilution for which amplification is positive). There are different programs to assemble and edit electropherograms. The program we use is Lasergene’s Seqman (DNASTAR). References 1. Thomson MM, Delgado E, Herrero I, et al. (2002). Diversity of mosaic structures and common ancestry of human immunodeficiency virus type 1 BF intersubtype recombinant viruses from Argentina revealed by analysis of near full-length genome sequences. J Gen Virol; 83:107-119. 2: Delgado E, Thomson MM, Villahermosa ML, et al. (2002). Identification of a newly characterized HIV-1 BG intersubtype circulating recombinant form in Galicia, Spain, which exhibits a pseudotype-like virion structure. J Acquir Immune Defic Syndr; 29:536-543. 3. Sierra M, Thomson MM, Ríos M, et al. (2005). The analysis of near full-length genome sequences of human immunodeficiency virus type 1 BF intersubtype recombinant viruses from Chile, Venezuela and Spain reveals their relationship to diverse lineages of recombinant viruses related to CRF12_BF. Infect Genet Evol; 5:209-217.