Quantum Mechanics Writing Sample
advertisement

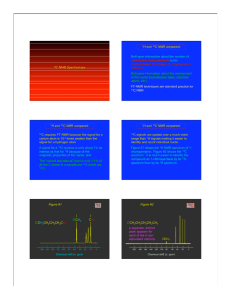
Michael Steury Dr. Dan Smith Quantum Mechanics 26 November 2009 Two Dimensional Analysis of 2’, 3’-Di-O-Acetyladenosine and 2’, 3’-Iso-OPropyladenosine Abstract: Two-dimensional NMR techniques DEPT, COSY, and HETCOR were used in addition to 1H and 13C spectra to assign proton peak locations on the molecules 2’, 3’-Di-OAcetyladenosine and 2’, 3’-Iso-O-Propyladenosine. The 1H assignments (in ppm) for 2’, 3’-Di-O-Acetyladenosine are: A, 1.98; B, 2.18; C, 3.77; D, 4.18; E, 5.57; F, 5.88; G, 6.18; H, 7.22; I, 8.18; J, 8.22; K, 5.90; L, 3.77. The 13C assignments (in ppm) are: 1, 19; 2, 21; 3, 67; 4, 76; 5, 78; 6, 90; 7, 93; 8, 125; 9, 145; 10, 154; 11, 157; 12, 161; 13, 174; 14, 175. The 1H assignments (in ppm) for 2’, 3’-Iso-O-Propyladenosine are: A, 1.42; B, 1.53; C, 3.56; D, 3.56; E, 4.20; F, 5.00; G, 5.32; H, 6.12; I, 7.46; J, 8.15; K, 8.42; L, 6.08. The 13C assignments (in ppm) are: 1, 26; 2, 27; 3, 62; 4, 82; 5, 84; 6, 87; 7, 90; 8, 113; 9, 119; 10, 140; 11, 148; 12, 153; 13, 156. Introduction: Using nuclear magnetic resonance (NMR) techniques the molecules 2’, 3’-Di-OAcetyladenosine and 2’, 3’-Iso-O-Propyladenosine were analyzed to identify 1H and 13C peak locations and their location in their respective molecules. NMR techniques provide a very sensitive probe of the electron distribution of a molecule, allowing for the distinguishing between inequivalent nuclei. The individual spins, such as from nearby 1H, can couple to generate multiplet splitting patterns, these patterns can then be used to determine structures of small organic molecules as well as a non-destructive imaging technique for materials or living tissues. Two-dimensional NMR with Fourier transform techniques allow for the structural determination of larger molecules as well as providing helpful information in assigning complex splitting patterns.1 Materials and Methods: Both 2’, 3’-Di-O-Acetyladenosine and 2’, 3’-Iso-O-Propyladenosine were dissolved in a combination of the solvents CDCL3-d and DMSO-d that was recommended by the Aldrich Spectroscopy Manual.2 2’, 3’-Di-O-Acetyladenosine was in a 1:1 mixture of solvents while 2’, 3’-Iso-O-Propyladenosine was in a ~1:3 mixture of CDCL3-d: DMSO-d4. The solute was slowly added to the solvents until no more solute could be dissolved in the solution, 160 mg of 2’, 3’-Di-O-Acetyladenosine and 70 mg of 2’, 3’Iso-O-Propyladenosine were added to meet this condition. 1H, 13C, COSY, DEPT, and HETCOR data was collected using a Varian Anaspect EM360 NMR Spectrometer (60 MHz) instrument and also using a Bruker Ultrashield 400 NMR (400 MHz) from Valparaiso University. Results and Discussion: The data was approached first with the goal of identifying the 13C peaks. Relying heavily on data from the DEPT (see attached spectra 4 and 8) and knowledge of chemical shifts due to oxygen and nitrogen, the carbon peaks could be indentified as seen on Figures 1 for 2’, 3’-Iso-O-Propyladenosine and Figure 2 for 2’, 3’-Di-OAcetyladenosine. Due to instrumental complications preventing the construction of HETCOR plot; identifying the hydrogen peaks was based upon different coupling patterns observed in the COSY and splitting patterns in the 1H spectra. These values are also displayed on Figures 1 and 2. The large singlet at 3.42 ppm is due to water contamination brought downfield through interaction with the DMSO-d solvent. In 2’, 3’-Di-O-Acetyladenosine the water peak coalesces with the CH2 peak on carbon 3. The explanation for the multiplet formed at 3.60 ppm in 2’, 3’-Iso-O-Propyladenosine (unresolved in 2’, 3’Di-O-Acetyladenosine) is that the two hydrogen’s on carbon 3 are sterically inequivalent. This leads to the two hydrogen’s splitting each other, being split by the hydrogen on carbon 6, and also split through long range coupling from carbon 5 (carbon 4 in 2’, 3’-Di-O-Acetyladenosine). This creates a doublet of doublets of doublets when placed on top of each other forms the visible multiplet. The alcohol peak for both compounds is found to coalesce with the peak from the hydrogen on carbon 4 (carbon 5 for 2’, 3’-Di-O-Acetyladenosine). This matches with the known chemical shift for alcohol groups and with integrations from the spectra.3 Due to the lack of a HETCOR plot for both molecules it was impossible to connect the data from the 13C plots (DEPT, 13C) with the data from the 1H experiments (COSY, 1H). This led to a higher level of uncertainty in the assignments both for hydrogen and carbon. The main limitations come from hydrogen’s and carbons that are a part of the aromatic rings. These carbons and hydrogen’s have very similar chemical shifts and splitting patterns. Because of COSY’s ability to detect long-range coupling and DEPT inability to clearly distinguish between carbon atoms there is a higher relative error in these assignments. Conclusion: Using the two-dimensional NMR techniques of 1H, 13C, COSY, and DEPT on both 60 and 400 MHz instruments all of the proton and carbon peaks were assigned (see Figures 1 and 2). The CH2 group for both compounds was determined to sterically inequivalent creating a doublet of doublets of doublets from short and long-range coupling. The assignments were limited by the instrumental complications preventing the acquisition of a HETCOR spectrum, generating uncertainties in the aromatic region. Reference: (1) Engel, T. Quantum Chemistry & Spectroscopy; Kaveney, D.; DuPont, C.; Folchetti, N.; Peasron Education, Inc., Upper Saddle River, New Jersey, 2006; 2ed., 17, p 407. (2) Pouchert, C.; Campbell, J. The Aldrich Library of NMR Spectra; Aldrich Chemical Co., Milwaukee, Wisconsin, 1983 (3) Engel, T. Quantum Chemistry & Spectroscopy; Kaveney, D.; DuPont, C.; Folchetti, N.; Peasron Education, Inc., Upper Saddle River, New Jersey, 2006; 2ed., 17, p 413.