AP Chem Syllabus 2015-16
advertisement

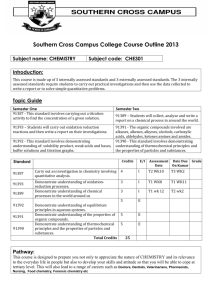
2015-16 AP Chemistry Syllabus LIN Instructor – Ms. Danielle Lin M. Ed. danielle.lin@rentonschools.us Available for help via email or from 6:50am-7:20am, 2:03pm-2:45pm Welcome to your uber-rigorous AP Chemistry course. My name is Danielle Lin and I will be your instructor. I hold a Bachelor of Science in biochemistry from Western Washington University and a Master in Education from Seattle Pacific University. I have worked in academic research toward creating single-chain human hemoglobin for use as an acute oxygen carrier. I have also worked in industry conducting clinical trials for an immunocuetical specifically created to teach the body to recognize prostate cancer. I have taught biology, AP biology, life science, 9th grade science, physical science, physics and chemistry. I am excited to teach AP Chemistry and for you to grow beyond what you feel is your potential. I look forward to our focused and intent work together. If you have any questions or concerns for me please feel free to contact me directly via email: Danielle.lin@rentonschools.us or phone at 425-204-4251. I am also available before and after school for individual consultations. I look forward to a great year!! Overall Grading Philosophy The goal of the AP Chemistry class is to aid in student preparation for the AP Chemistry exam. Homework, lab work, practice exams, sample AP exam question, etc will be assigned different point values based on difficulty. Students will be given a grade for the class and I will make a positive adjustment to student grades based on their performance on the AP exam. Any student who receives a 3 on the exam will achieve a minimum of a B grade for the class overall, any student who receives a 4 or 5 on the AP exam will be given an A for the class overall. Homework Homework is assigned and is to be submitted through Webassign.net. Students are required to sign up and pay $10.50 for the year to access homework problems and submit their work. This is the same website used by most universities and provides students with exposure to a system commonly used at most colleges. Homework will be assigned each day and is due the next school day. Login and setup information will be provided on day one. The lab fee for this class is $20. Labs Sixteen total labs will be performed through the year. Students need to be able to submit their lab notebook for review after each lab. The lab notebook needs to be neat, organized, easy to read, and thorough. Failure to meet this standard will result in grade reduction. Tardy Policy This class adheres strictly to the schoolwide policy on truancy. Students who arrive up to 5 minutes and 59 seconds after the period begins will be marked tardy. Students who are 6 minutes or later after the period begins will be marked absent. Missed Days Every day represents a great educational opportunity for students to learn and succeed in my classroom. Although there will be no points docked for absences it is the student’s responsibility to make up any work. Homework is available from WebAssign, if you are absent I expect that homework is still turned in unless otherwise arranged. Notes from a particular day are best obtained from fellow students. If you still have questions I am available both before and after school to help. In-Class Behavior & Norms This is a college level course, likely the hardest course you will take in high school. I expect full commitment to the class and for each student to aid fellow classmates when appropriate to raise the intellectual level of the class as a whole. Since this is a lab class with many chemicals, food and drink are not allowed. Plagiarism is not allowed, please see Hazen student handbook for rules. 2015-16 AP Chemistry Syllabus LIN This is the pacing guide for this AP Chemistry Course **NOTE there are 30 WEEKS of school between now and the AP EXAM Textbook: CHEMISTRY: THE CENTRAL SCIENCE-Brown and LeMay 9th Edition 1. Review Chapters 1-3, plus chapter 4. (4 weeks) This includes: Brown and LeMay Chapters 1-4: Chapter 1:Matter and Measurement Chapter 2: Atoms, Molecules and Ions Chapter 3: Stoichiometry, Calculations with Chemical Formulas and Equations Chapter 4: Aqueous Reactions and Solution Stoichiometry Chapters 1-3 are MAINLY a review of the following first year Chemistry topics: a. Types of Mattere. Balanced Equations elements, compounds, f. Stoichiometry homogeneous/heterog g. States of Matter eneous mixtures h. Gas Laws b. Atomic structure i. Periodic Properties c. Naming j. Electron compounds/writing Configurations formulas k. Ionic/Covalent d. Mole Concept bonding Topics in Chapter 4 “Aqueous Reactions and Solution Stoichiometry are MAINLY new and require more instruction. LAB: Scientific Measurements LAB: Lab Practical—Titration of an Unknown Acid MID FIRST QUARTER EXAM 2. Chapter 5: Thermochemistry LAB: Separation of the Components of a Mixture LAB: Thermochemistry and Hess’s Law 3. Chapter 6: Electronic Structure of Atoms LAB: The Atomic Structure of Hydrogen LAB: Empirical Formula of an Oxide by Formation 4. Chapter 7: Periodic Properties of the Elements LAB: Gravimetric Determination of Chloride END OF FIRST QUARTER EXAM 5. Chapter 8: Basic concepts of Chemical Bonding LAB: Precipitation and Analysis of the Silver Group 6. Chapter 9: Molecular Geometry and Bonding Theories LAB: Spectrophotometric Iron Analysis (2 weeks) (1 week) (1 week) (2 weeks) (2 weeks) 7. Chapter 10: Gases (2 weeks) LAB: Determination of Molecular Weight by the Dumas Method MID SECOND QUARTER EXAM 2015-16 AP Chemistry Syllabus LIN 8. Chapter 11: Intermolecular Forces, Liquids and Solids (2 weeks) LAB: Determination of the Calcium content of Water by EDTA 9. Chapter 13: Properties of Solutions (NOTE: Chapter 12 “Modern Materials” is not covered LAB: Preparation of Aspirin and Oil of Wintergreen (2 weeks) END OF SECOND QUARTER EXAM 10. Chapter 14: Chemical Kinetics (3 weeks) LAB: Rates of Reactions II: Rate and Order of Hydrogen Peroxide Decomposition 11. Chapter 15: Chemical Equilibrium (2 weeks) LAB: Equilibrium Constant for FeSCN2+ Formation 12. Chapter 16: Acid-Base Equilibria (3 weeks) LAB: pH Titration of an Unknown Weak Base MID THIRD QUARTER EXAM 13. Chapter 17: Additional Aspects of Aqueous Equilibria (2 weeks) 14. Chapter 19: Chemical Thermodynamics (2 weeks) (NOTE: Chapter 18 “Chemistry of the Environment” is not covered) 15. Chapter 20: Electrochemistry (2 weeks) LAB: Electrochemical Cells END OF THIRD QUARTER EXAM 16. Review for AP Exam (2 weeks) “MOCK” AP EXAM NOTE: Chapters 21 “Nuclear Chemistry”, Chapter 24 “Chemistry of Coordination Compounds” and Chapter 25 “The Chemistry of Life: Organic and Biological Chemistry” are covered as extra credit assignments during school holidays. 2015-16 AP Chemistry Syllabus LIN 2015-16 AP Chemistry Syllabus LIN 2015-16 AP Chemistry Syllabus LIN The undersigned have read and understand the 2015-16 AP Chemistry Syllabus: Student Name______________________________________________________ Student Signature___________________________________________________ Date_____________________________________________________________ Parent Name_____________________________________________________ Parent Signature__________________________________________________ Parent Email_____________________________________________________ Parent Phone_____________________________________________________ Date_____________________________________________________________ What is your goal (…in life, or if that is too big picture for the 5 years after High School)? How does AP Chem fit into the big picture of your goal? Why did you sign up for AP Chemistry? What do you feel is your “potential” in chemistry?