Handling of animal by-products and products made
advertisement

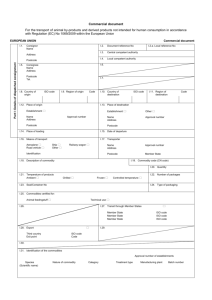
Informationsdokument Komparativ medicin Handling of animal by-products and products made thereof This document summarizes the requirements set for usage, import and export of animal by-products and products made thereof. Background All products from the animal kingdom not intended for human consumption and not yet processed as manufactured products, are defined as animal by-products (often abbreviated abp)1. Within research activities this pertains to, for example, proteins (over 10 kDA), certain antibodies2, cell cultures, and serum that have an animal origin. Also other tissues as well as whole or parts of dead animals (e.g. organs from laboratory animals) are included. All animal products are not, however, covered by this regulation3. Examples of what is not covered: - CE-certified products are considered as non-contagious and are exempted from the rules (the company has responsibility for protection against contagion) - Use of dead wild animals, or parts of wild animals, that are not suspected to be infected with diseases that can be transmitted to humans or animals - Eggs, embryos and sperm intended for breeding. Users of animal by-products for research purposes In order to use animal by-products and products manufactured thereof for research and diagnostic purposes, one must be granted a permit for this from the Swedish Board of Agriculture4. One purpose for this, among other reasons, is to ensure traceability. Manufactured products are those products that have resulted from processing animal by-products. Karolinska Institutet is registered as a facility for handling of animal by-products for research purposes (not diagnostics) and has a permit for the use of these. The permit is available by e-mailing Comparative Medicine at km@ki.se. For those who want to acquire animal by-products for research and educational purposes the following must be observed 1. No import permit is required for import from the EU as well as Norway, Iceland and Switzerland. The transporter must, however, be approved for transporting animal byproducts. 2. For import from countries outside the EU, Norway, Iceland and Switzerland, an import permit is required. For both types of import there must be a trade document in original, issued by the sender, together with the transport. The reason for this is e.g. to ensure the traceability. The trade document is to be saved by you, the recipient, for at least two years. Note that this requirement applies both to national and international transports. Karolinska Institutet has a permit for import of samples for research and education from the following sender and origin countries: USA, Canada, Taiwan, Japan, Singapore and Australia. A time-limited copy of the permit for import can be sent to you upon request; e-mail Comparative Medicine at km@ki.se 1 Regulated by Regulation EG No 1069/2009 as well as the Swedish Board of Agriculture's Regulation SJVFS 2011:21. 2 Affinity-cleaned antibodies are not considered as animal by-products and are not regarded as animal by-products. 3 Read more on what is not covered by the regulations (Article 2 EG No 1069/2009). 4 Research with animal byproducts that have been produced within an animal facility are not covered by this if the usage is contained within the animal facility. Original lagras elektroniskt i Centuri, utskrift utan kvalitetsansvarigs signatur är ogiltig. Utfärdad av Godkänd av Isabel Dellacasa Björn Vennström Dok.nr-version KI161-1 Gäller från 2012-09-05 Sida 1 av 2 Karolinska Institutet must also keep a continuous register of these imports which upon request are to be shown to the Swedish Board of Agriculture. A copy of the received trade document is therefore to be sent to Comparative Medicine, km@ki.se, after receipt of the shipment. For those who want to export or send animal by-products for research or educational purposes the following must be observed A trade document in original, issued by you, has to be included with the shipment. The recipient of the products must save the trade document for two years. This applies to animal by-products for both national and international transports. Other rules may be applicable for recipients in lands outside the EU, which must be determined before exporting. Note that the recipient can refuse to accept the product if the trade document is lacking. Only the standardized trade documents downloaded from the Swedish Board of Agriculture's website can be used. Contact Comparative Medicine by e-mail at km@ki.se if you need help with this. A copy of the completed trade document shall then be sent to km@ki.se. During transport a label must be attached to the container or vehicle stating: a) Which category of material the shipment belongs to (category 1, 2 or 3) b) An easily legible text on the container or vehicle with the text: “For research and diagnostic purposes” EG 1069/2009 (replaces EG 1774/2002). Regulation laying down health rules as regards animal by-products and derived products not intended for human consumption SFS 2006:805. Law on fodder and animal by-products SFS 2006:814. Regulation on fodder and animal by-products SJVFS 2011:21/ SJVFS 2006:84. The Swedish Board of Agriculture’s regulation on handling animal by-products and import of other products, except for food for human consumption that can spread infectious diseases to animals and humans SJVFS 2007:21. Official control of fodder and animal by-products More information and classification of the different categories is available through this link (Swedish only): http://www.jordbruksverket.se/amnesomraden/djur/djurprodukter/vadaranimaliskabiprodukter.4.67e843d911ff9f551db80002182.html Original lagras elektroniskt i Centuri, utskrift utan kvalitetsansvarigs signatur är ogiltig. Gäller från 2012-09-05 Dok.nr-version KI161-1 Sida 2 av 2