Locke et al., Diabetes AEI final accepted version
advertisement

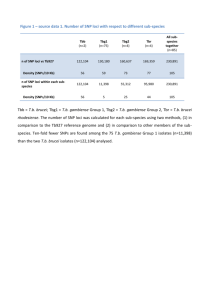
Targeted allelic expression profiling in human islets identifies cis-regulatory effects for multiple variants identified by type 2 diabetes genome-wide association studies Running title: - Type 2 diabetes GWAS genes allelic islet levels Jonathan M. Locke1, Gerald Hysenaj1, Andrew R Wood1, Michael N Weedon1, Lorna W Harries1 1 Institute of Biomedical & Clinical Science, University of Exeter Medical School, Exeter, EX2 5DW Corresponding author: Lorna W Harries Room L3/20 RILD Building University of Exeter Medical School Barrack Road Exeter EX2 5DW Tel: 01392 406749 Fax: 01392 408385 Email: l.w.harries@exeter.ac.uk Word count: 3118 Figures: 3 Tables: 1 Genome-wide association studies (GWAS) have identified variation at >65 genomic loci associated with susceptibility to type 2 diabetes, but little progress has been made to date in elucidating the molecular mechanisms behind most of these associations. Allelic expression profiling is a powerful technique for identifying cis-regulatory variants controlling gene expression, using samples heterozygous at transcribed single nucleotide polymorphisms (SNPs) to distinguish and measure levels of transcripts derived from each allele. Exonic SNPs, suitable for measuring mRNA levels and in high linkage disequilibrium (LD) with 65 lead type 2 diabetes GWAS SNPs, were identified in 18 RefSeq genes. For 14 of these genes allelic expression could be determined using RNA and DNA isolated from islets of 36 white, non-diabetic donors. A significant allelic expression imbalance (AEI) was identified for 7 genes (ANPEP, CAMK2B, HMG20A, KCNJ11, NOTCH2, SLC30A8 and WFS1). For 5 of these genes allelic expression could be determined using other linked exonic SNPs, with significant AEI confirmed in each case. This study provides one of the first pieces of evidence supporting the hypothesis that changes to cis-regulation of gene expression are involved in a large proportion of common variant associations with susceptibility to type 2 diabetes. Genome-wide association studies (GWAS) have discovered germline genetic variation associated with type 2 diabetes risk (1-4). One of the largest GWAS, involving DNA taken from individuals of European descent, and conducted by the DIAGRAM (DIAbetes Genetics Replication and Meta-analysis) consortium identified 65 loci associated with type 2 diabetes risk (1). However for most of these loci, the precise identity of the affected gene and the molecular mechanisms underpinning the altered risk are not known. Similar to other complex polygenic diseases, the lack of protein-coding variation at the majority of these loci has led many to believe that the disease-associated variation must result in changes in the level of gene expression. This is predicted to affect the amount or nature of protein produced resulting in compromised cellular function and ultimately altered disease risk. It is this hypothesis that has led many to conduct expression quantitative trait loci (eQTL) studies, where the disease-associated genotype is correlated with expression of cislinked transcripts. Whilst there have been some successes when using such an approach (5; 6), the presence of multiple known and unknown confounding factors that can affect gene expression means few disease-associated loci are robustly associated. Allelic expression profiling provides an alternative and direct way to measure the effect of cis-regulatory variation on gene expression. By measuring transcripts from each allele of a gene (using a SNP as a marker to differentiate between the two mRNA copies) the effect of trans-acting factors on gene expression is essentially removed as the output from one allele serves as a within-sample control for the other. The known tissue-specificity of gene expression regulation means that the most informative studies will measure transcript levels in the specific tissue(s) relevant to the disease. In the case of type 2 diabetes, characterisation of physiological responses (e.g. stimulus-induced insulin secretion, insulin sensitivity) suggests most loci are associated with defects in pancreatic beta cell function (2; 3; 7). Therefore there is a real need to measure gene expression in human beta cells (or whole islets as these have been shown to be a suitable proxy (8)). There have however been very few reports linking type 2 diabetes-associated variation with islet gene expression using the classical eQTL approach (9; 10). In this report we provide one of the first pieces of evidence to support the theory that for a large proportion of loci associated with predisposition to type 2 diabetes the variants affect the expression of known protein-coding genes. Furthermore by pinpointing genes with allelic expression imbalances we highlight the genes likely to be involved in disease pathogenesis. RESEARCH DESIGN AND METHODS Tissue and nucleic acid extraction Snap-frozen islets were purchased from ProCell Biotech (Newport Beach, CA, USA) where islets had been collected with ethical permission at source. Donor characteristics are presented in Supplementary Table 1. Islet purity and viability was determined by dithizone and fluorescein diacetate/propidium iodide staining, respectively. RNAlater-ICE (Life Technologies, Carlsbad, CA, USA) was used to transition the tissue to a state where RNA could be extracted using the miRVana miRNA isolation kit, as per the manufacturers’ instructions. Small amounts of genomic DNA were co-eluted upon RNA extraction. Wholegenome amplification was subsequently carried out using the REPLI-g Mini kit (Qiagen, Venlo, Netherlands). Genotyping and quantitative reverse transcriptase-PCR (qRT-PCR) To find heterozygous samples suitable for allelic expression measurements whole genome amplified genomic DNAs were first genotyped. In cases where the transcribed SNP is more common in the CEU population than the lead type 2 diabetes SNP at that locus (according to 1000 Genomes Project, Phase 1, CEU population) the lead SNP was also genotyped (by Sanger sequencing) and only samples heterozygous at both loci were included for further analysis. For allelic expression analyses total RNA was DNase-treated using the Turbo DNA-free kit (Life Technologies). DNase-treated RNA was subsequently reverse transcribed using the SuperScript VILO cDNA synthesis system (Life Technologies). For each SNP duplicate wells were used to amplify cDNA and ~5ng genomic DNA in all heterozygous samples. Following amplification wells with a cycle threshold (Ct) value >36 were excluded from our analysis. To calculate dCt values the difference in Ct value for each reporter dye (VIC/FAM), detecting each SNP allele, was determined. Subsequently, ddCt values were calculated by determining the difference between the dCt and the mean dCt of all genomic DNA samples. Genomic DNA samples should show a 1:1 allelic ratio and thus any departure from 0 illustrates unequal amplification of alleles which must be corrected for. These ddCt values were then transformed to 2-ddCt values to take into account the logarithmic nature of PCR. Mean average allelic expression measurements, determined from two independent cDNAs reverse transcribed and amplified on different days, are presented. All genotyping and allelic expression experiments were performed with TaqMan SNP Genotyping assays and TaqMan Genotyping Master Mix (both Life Technologies). Inventoried TaqMan SNP genotyping assays were used where possible (amplicon mapping 100% to exonic sequence). If not available or not meeting our design specifications custom TaqMan SNP genotyping assays were designed and purchased where possible. For the islet eQTL analysis inventoried TaqMan Gene Expression assays and TaqMan Fast Advanced Master Mix (both Life Technologies) and were used. Relative expression of target genes was normalised to the geometric mean of 5 housekeeping genes (ACTB, B2M, GUSB, HMBS, RPL11). All TaqMan assays were run on the ABI7900HT platform (Life Technologies). Assay IDs and primer/probe sequences for TaqMan SNP Genotyping and TaqMan Gene Expression assays are shown in Supplementary Table 2. Statistical analysis Paired two-tailed T-tests, comparing genomic DNA and cDNA values from the same donor, were used to determine statistical significance for allelic expression. Allelic and total gene expression values were normally distributed upon log2 transformation and thus permitted the use of parametric statistics. Pearson correlation coefficients were used to ascertain the robustness of our allelic expression measurement. RESULTS Bioinformatic analysis of type 2 diabetes GWAS loci to identify exonic SNPs for allelic expression profiling in human islets Large-scale association studies conducted by DIAGRAM, in individuals overwhelmingly of European descent, have reported 65 lead SNPs associated with susceptibility to type 2 diabetes (1). Figure 1 illustrates how these SNPs and closely correlated proxy SNPs were systematically selected for allelic expression analysis. In brief 1525 proxy SNPs (r2>0.8, CEU, 1000 Genomes Phase 1) were found. Of these SNPs (lead + proxies) 45/1590 (2.8%) map to exons of 23 human RefSeq genes. For 18 of these genes, TaqMan SNP genotyping assays could be designed to map entirely to exonic sequence, thus allowing for amplification and measurement of mature (i.e. spliced) mRNA species and normalisation of allelic expression using genomic DNA from the same individual. After exclusion of SNPs with <4 heterozygotes (rs1801282, PPARG; rs3734621, KIF6) and assays where >50% cDNA samples yielded Ct values >36 (rs2793823, ADAM30; rs7377, SRGN), indicating very low levels of gene expression, allelic expression could be determined for 14 genes in samples from 36 white, non-diabetic donors. Validation of TaqMan SNP genotyping assays as a robust method for determining allelic expression The reproducibility of the allelic expression measurement was determined by comparing values generated from independent cDNAs that were reverse transcribed and amplified by real-time PCR on different days and PCR plates (r2=0.72, p=1.8 x 10-62; Fig. 2A). To further demonstrate the robustness of this technique allelic expression values for 5 genes were compared between 2 linked SNPs residing in the same gene (r2=0.67, p=5.7 x 10-16; Fig. 2B). Identification of allelic expression imbalance at GWAS loci Significant (unadjusted p<0.05) allelic expression imbalance (AEI) was observed for 7/14 genes tested (Table 1, Fig. 3A,C,E,G,I,K,L). For 5 of the genes where significant imbalance was observed (ANPEP, KCNJ11, NOTCH2, SLC30A8, WFS1), there was at least one other exonic proxy SNP which was used for validation of our findings. For all five of the tested SNPs significant (unadjusted p<0.05) AEI in a direction consistent with the results from the first SNP was observed (Table 1, Fig. 3B,D,F,H,J). We were unable to validate our results for CAMK2B and HMG20A due to the position of the only other proxy exonic SNP on the transcript (SNP either too close to a splice site to allow for assay to be designed to measure mature mRNA, or near a sequence of low complexity precluding assay design). Islet eQTL analysis provides further evidence to support ANPEP as being the causal gene at this locus One limitation of our approach is its reliance on the presence of exonic proxy SNPs, thus preventing testing of all candidate genes at a locus. Of the 7 genes where we identified significant AEI ANPEP exhibited by far the largest imbalance, with both SNPs tested showing ~50% difference between mRNA alleles. Therefore we predicted that we had most power to see an eQTL effect at this locus versus the other 6 where far lower AEIs were observed. Using real-time PCR we measured the expression of ANPEP and four neighbouring RefSeq genes, the two immediately 5’ of ANPEP (MESP1, MESP2) and the two immediately 3’ of ANPEP (AP3S2, ARPIN) in the 36 islet samples. Consistent with our AEI findings of increased levels of ANPEP mRNA originating from the risk allele we measured 2.2-fold higher levels of ANPEP expression in individuals homozygous for the risk (G) allele of rs2007084 compared to heterozygous individuals (p=0.03) (Fig. 4A). We found no association with rs2007084 genotype for 3 of the other genes at this locus (AP3S2, p=0.61; ARPIN, p=0.11; MESP1, p=0.97) (Fig. 4B-D). Very low levels of MESP2 transcripts (all samples either mean Ct>38 or not amplified) meant analysis of MESP2 expression with respect to genotype was not possible. In summary, this eQTL analysis supports our AEI findings for ANPEP and provides further evidence that the type 2 diabetes-associated risk allele at this locus exerts its detrimental effect via increasing ANPEP expression. DISCUSSION In this study we report that half of the type 2 diabetes SNPs examined had effects on allelic expression of nearby genes, consistent with a role for such variation in cis-regulation of islet gene expression. This figure is greater than genome-wide analyses of AEI which have generally reported 4-8% of common SNPs associated with the expression of at least one transcript (11; 12). An enrichment of significant genotype effects on cis-linked gene expression has been reported for complex disease traits (13; 14) and our results suggest that effects on gene expression may play a significant role in mediating the genotype associations with type 2 diabetes. We are aware of only one other published study reporting associations between gene expression levels and multiple type 2 diabetes-associated lead SNPs. Using global microarray data measuring gene expression in islets from 63 donors Taneera et al. reported a significant (p<0.001) cis-eQTL for 5/47 (11%) type 2 diabetes-associated SNPs (10). Unfortunately none of the cis-eQTLs identified in their study could be tested in our study due to a lack of SNPs meeting our inclusion criteria (exonic and assay mapping purely to exonic sequence). A comparison of the results from their study and ours illustrates some of the advantages and disadvantages of the respective techniques. Whilst our targeted allelic expression approach identifies a higher percentage (50% versus 11%) of genes regulated by cis-acting variation our approach is limited to genes with exonic SNPs in high linkage disequilibrium (r2>0.8) with the lead type 2 diabetes-associated SNP. Indeed this dependence will also often preclude examining isoform-specific effects which may be substantial (15) and an examination of all candidate genes at a locus. The classical cis-eQTL approach is not dependent on transcribed exonic SNPs and thus can examine many more of the disease-associated loci and the expression of any of the closely-residing genes and their isoforms. However the presence of known and unknown trans-acting confounding factors, which cannot all be accounted for in regression analyses, adds significant variation to the gene expression measurements and likely contributes to the lower number of significant associations between genotype and expression. To enable a greater number of type 2 diabetes-associated loci to be examined by the more powerful allelic expression approach future studies may consider using intronic SNPs to measure allelic expression. Allelic expression measurements calculated from intronic SNPs have been shown to be highly correlated with values calculated from linked exonic SNPs (16). Whilst this is, to our knowledge, the first report of a systematic use of allelic expression measurements in human islets to identify likely causal genes at type 2 diabetes GWAS loci, there are other published studies that have reported allelic imbalance in human islets at single loci. Kulzer et al. report allelic expression imbalance for two SNPs in ARAP1 (17), one of which (rs11603334) was also tested in our study. They report significant AEI for rs11603334 in 6 individuals with a mean AEI = 12.6% whereas our study fails to identify significant AEI (p=0.54) in the 7 heterozygous individuals tested. It is difficult to understand the reasons for such disparate findings, given that allelic expression should remove much of the confounding effects of trans-acting factors, but it may be that differences in methodology (TaqMan versus MALDI-TOF) and the small sample sizes (n=6-7) explain the lack of replication. Another study by Nica et al. analysed allelic expression for type 2 diabetes GWAS implicated genes using RNA-Seq in pancreatic beta cells from 11 donors. In support of our findings they also report AEI for rs5215 (KCNJ11) and rs11558471 (SLC30A8), albeit only in four samples and one sample, respectively (8). The lack of reports detailing allelic expression imbalances for type 2 diabetes GWAS genes may be explained by the widespread use of non-targeted RNA-Seq methodologies. Whilst whole transcriptome RNA-Seq approaches provide researchers with an unparalleled resource to investigate all transcripts, its lack of targeting of specific transcripts means the sequencing depth at loci of interest is not as high as could be achieved using targeted approaches. An examination of two recent studies using RNA-Seq methods to quantify the islet transcriptome (18; 19) shows that the median RPKM (reads per kilobase per million mapped reads) for the genes at the 65 type 2 diabetes-associated GWAS loci is 8-9 RPKM. If one considers that current read lengths are ~50bp and typical human islet sequencing depths vary, but are perhaps currently ~50 million mapped reads per sample, then an average 25 reads will map to one exonic SNP. Fontanillas et al. calculate that 200 reads (equivalent to 8 samples) would give 80% power to detect AEI of 1.5, whilst to detect an AEI =1.25 >500 mapped reads (>20 samples) would be needed (assuming a liberal type 1 error rate α = 5%) (20). In our study we found an AEI ≤1.25 for 5/7 loci where AEI was significant. Therefore to replicate our findings using current non-targeted RNA-Seq technologies would likely require a number of heterozygous individuals which, given the difficulty in collecting large cohorts of human islets, would be difficult to procure in the near future. The significance of small AEIs (≤1.25) at 5 of the loci deserves discussion. Are such seemingly small imbalances of significant consequence to islet function? At one of these loci (SLC30A8) a missense SNP (rs13266634; W325R) is in complete LD (r2=1, D’=1) with the lead SNP (rs3802177) also examined for AEI. There is some evidence that functional nonsynonymous variants have co-evolved with regulatory variants so as to strengthen or diminish their functional impact (21). This would suggest that the increase in expression of the risk versus non-risk allele is not directly altering disease risk in such cases, but rather moderating the effect of the non-synonymous variant, which is primarily responsible for the molecular deficit increasing disease risk. However, with respect to SLC30A8 it has recently been reported that heterozygous loss-of-function mutations, which are likely null alleles, protect against type 2 diabetes (22). This is strong evidence that the precise levels of SLC30A8 are important for beta cell function. Given that we measured increased levels of SLC30A8 transcripts originating from the risk, versus non-risk allele, one could speculate that it is the increase in SLC30A8 levels, rather than the presence of the missense variant, which increases type 2 diabetes risk. Indeed the sole study, to date, that examined the functional effects of the missense variant on zinc transporter activity, reported only a mild attenuation in zinc ion transporter activity in cells overexpressing the risk (R) allele of W325R, compared to cells expressing the non-risk (W) allele (23). If we consider the other 6 loci where we observe significant AEI, two (ANPEP, KCNJ11) have at least one missense variant within that gene and in high LD (r2>0.8) with the lead SNP, whilst at the NOTCH2 locus there is a missense SNP in a nearby gene (ADAM30). Whether the SNP in ADAM30 is of consequence to the type 2 diabetes association is very questionable given its reported testis-specific expression (24), and the very low expression we and others (8; 19), have observed in human islets. Furthermore, small changes in expression of NOTCH2, as identified in this study, may be of significant consequence to the beta cell with experiments in mice showing that when only one copy of NOTCH2 is conditionally removed from Ngn3-positive cells (i.e. endocrine progenitors), the cells are diverted to an acinar fate (25). Therefore it may be hypothesised that carriers of NOTCH2 risk alleles have reduced beta cell mass (and hence increased type 2 diabetes susceptibility) but difficulties in accurately measuring beta cell mass in large cohorts of individuals mean this theory is currently not easily amenable to testing. With regards to ANPEP the one proxy SNP (rs17240268) resulting in a non-synonymous substitution does not affect an evolutionary well-conserved residue. Therefore this increases the likelihood that the ANPEP expression changes we identified, via allelic expression and eQTL analyses, are causally involved in the association between SNPs at this locus and risk of type 2 diabetes. The ANPEP gene encodes the protein, aminopeptidase N, a broad specificity membraneassociated peptidase. Its widespread expression and involvement in many cellular processes (e.g. proliferation, apoptosis, antigen presentation, differentiation, angiogenesis, chemotaxis) (26) makes it difficult to predict exactly how increased expression predisposes to type 2 diabetes. Indeed this may partially explain why investigators sought to proffer the neighbouring gene, AP3S2, as the causal gene at this locus, this despite the fact that the lead and all proxy SNPs, as defined in this paper, map within the ANPEP gene. The results of this study will hopefully stimulate research into the role of aminopeptidase N in the pathology of diabetes. For three genes (CAMK2B, HMG20A and WFS1) there are no non-synonymous variants in high LD (r2>0.8), strongly suggesting that at these loci, at least, alterations in gene expression are primarily involved in affecting type 2 diabetes risk. Our finding of a small AEI for WFS1 may be of significant consequence to the beta cell given that homozygous null alleles in WFS1 cause Wolfram syndrome, a rare, recessive disorder characterised by earlyonset diabetes with a non-autoimmune origin (27), and a heterozygous mutation segregates with adult-onset diabetes in a large family (28). Our most unexpected finding is perhaps the significant AEI for CAMK2B. This gene is in close proximity to GCK, the gene which encodes glucokinase, a key enzyme involved in the first rate-limiting step of glucose metabolism in the beta cell, and a gene where heterozygous inactivating mutations cause persistent, mild hyperglycaemia and homozygous inactivating mutations cause permanent neonatal diabetes (29). Characterisation of the effects of the type 2 diabetes-associated SNP at this locus on glycaemic traits show a strong association with fasting glucose (7), further implicating GCK as the causative gene. One cannot rule out however a role for variation in CAMK2B levels affecting beta cell function given that this gene encodes a calcium/calmodulin-dependent protein kinase that has been shown to be involved in coupling Ca2+ entry with insulin secretion (30-32), and that this isoform is the predominant form in pancreatic beta cells (31; 33). Little is known about HMG20A function in human islets but its involvement in neuronal differentiation (34; 35) may point towards a similar function in the neuronal-like beta cell. Whilst we have identified AEIs in genes implicated by type 2 diabetes GWAS in human islets it is not altogether clear whether altered expression of these genes in islets is the primary mechanism leading to changes in disease susceptibility. The most recent and largest study to date examining the effect of T2D GWAS variants on continuous glycaemic traits reports 12/36 loci examined showing defects in beta cell function, whilst only 4/36 were associated with insulin sensitivity measures (7). Of the 12 loci associated with defects in beta cell function we report AEI of SLC30A8 and CAMK2B at the SLC30A8 and GCK loci, respectively. At the remaining 20 loci Dimas et al. report no discernible effect of the SNPs on glycaemic traits; this includes the NOTCH2, KCNJ11 and WFS1 loci exhibiting AEI in our study. Given the raft of measures used to assay beta cell function, in particular, one might speculate that SNPs at these twenty loci impact on other cell types relevant to diabetes pathogenesis. However it is worth noting that a number of loci harbouring genes where mutations cause monogenic diabetes (HNF1A, HNF1B, KCNJ11, WFS1), and which do so through a detrimental effect on beta-cell function, are included in this list with unknown effects on glycaemic traits. Therefore one cannot rule out the possibility that the genes where we have uncovered AEI in islets, but where there is no data currently to support an effect of the type 2 diabetes associated SNP on beta cell function (either through physiological analyses of SNPs on glycaemic measures or presence of monogenic forms of diabetes), do so via an effect in this tissue. Furthermore whilst regulation of gene expression can be highly tissue-specific the findings of significant numbers of cis-eQTLs that are tissue-independent (shared across two or more tissues) mean that AEI observed in islets may be observed in other disease-relevant tissues (36-38). In summary, we report allelic expression imbalances that are consistent with type 2 diabetesassociated variation regulating the expression of cis-linked genes in human islets. For some of the genes where significant AEI was identified (e.g. SLC30A8, WFS1) there is strong evidence from human genetics that small changes in gene dosage may have significant consequences for the pancreatic beta cell. For other genes with significant AEI (e.g. ANPEP, HMG20A) their role is less well-defined and hence this study should provide a platform for further work examining the effects of carefully manipulating the expression of these genes in human islets. Author contributions. J.M.L. and L.W.H. conceived and designed the experiments. J.M.L., G.H., A.R.W. and M.N.W. conducted experiments and/or analysed data. J.M.L., G.H., A.R.W., M.N.W. and L.W.H. interpreted the results. J.M.L. wrote the manuscript. L.W.H. and M.N.W. reviewed and edited the manuscript. L.W.H. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Acknowledgments. We would like to thank Dr Tim McDonald and Professor Tim Frayling (both University of Exeter Medical School) for helpful discussions during this study. Funding. This work was supported by funding from the Medical Research Council (Grant number MR/J006777/1). Duality of interest. The authors would like to state that they have no conflicts of interest relevant to the publication of this study. References 1. Morris AP, Voight BF, Teslovich TM, Ferreira T, Segre AV, Steinthorsdottir V, Strawbridge RJ, Khan H, Grallert H, Mahajan A, Prokopenko I, Kang HM, Dina C, Esko T, Fraser RM, Kanoni S, Kumar A, Lagou V, Langenberg C, Luan J, Lindgren CM, MullerNurasyid M, Pechlivanis S, Rayner NW, Scott LJ, Wiltshire S, Yengo L, Kinnunen L, Rossin EJ, Raychaudhuri S, Johnson AD, Dimas AS, Loos RJ, Vedantam S, Chen H, Florez JC, Fox C, Liu CT, Rybin D, Couper DJ, Kao WH, Li M, Cornelis MC, Kraft P, Sun Q, van Dam RM, Stringham HM, Chines PS, Fischer K, Fontanillas P, Holmen OL, Hunt SE, Jackson AU, Kong A, Lawrence R, Meyer J, Perry JR, Platou CG, Potter S, Rehnberg E, Robertson N, Sivapalaratnam S, Stancakova A, Stirrups K, Thorleifsson G, Tikkanen E, Wood AR, Almgren P, Atalay M, Benediktsson R, Bonnycastle LL, Burtt N, Carey J, Charpentier G, Crenshaw AT, Doney AS, Dorkhan M, Edkins S, Emilsson V, Eury E, Forsen T, Gertow K, Gigante B, Grant GB, Groves CJ, Guiducci C, Herder C, Hreidarsson AB, Hui J, James A, Jonsson A, Rathmann W, Klopp N, Kravic J, Krjutskov K, Langford C, Leander K, Lindholm E, Lobbens S, Mannisto S, Mirza G, Muhleisen TW, Musk B, Parkin M, Rallidis L, Saramies J, Sennblad B, Shah S, Sigurethsson G, Silveira A, Steinbach G, Thorand B, Trakalo J, Veglia F, Wennauer R, Winckler W, Zabaneh D, Campbell H, van Duijn C, Uitterlinden AG, Hofman A, Sijbrands E, Abecasis GR, Owen KR, Zeggini E, Trip MD, Forouhi NG, Syvanen AC, Eriksson JG, Peltonen L, Nothen MM, Balkau B, Palmer CN, Lyssenko V, Tuomi T, Isomaa B, Hunter DJ, Qi L, Shuldiner AR, Roden M, Barroso I, Wilsgaard T, Beilby J, Hovingh K, Price JF, Wilson JF, Rauramaa R, Lakka TA, Lind L, Dedoussis G, Njolstad I, Pedersen NL, Khaw KT, Wareham NJ, Keinanen-Kiukaanniemi SM, Saaristo TE, KorpiHyovalti E, Saltevo J, Laakso M, Kuusisto J, Metspalu A, Collins FS, Mohlke KL, Bergman RN, Tuomilehto J, Boehm BO, Gieger C, Hveem K, Cauchi S, Froguel P, Baldassarre D, Tremoli E, Humphries SE, Saleheen D, Danesh J, Ingelsson E, Ripatti S, Salomaa V, Erbel R, Jockel KH, Moebus S, Peters A, Illig T, de Faire U, Hamsten A, Morris AD, Donnelly PJ, Frayling TM, Hattersley AT, Boerwinkle E, Melander O, Kathiresan S, Nilsson PM, Deloukas P, Thorsteinsdottir U, Groop LC, Stefansson K, Hu F, Pankow JS, Dupuis J, Meigs JB, Altshuler D, Boehnke M, McCarthy MI: Large-scale association analysis provides insights into the genetic architecture and pathophysiology of type 2 diabetes. Nat Genet 2012;44:981-990 2. Dupuis J, Langenberg C, Prokopenko I, Saxena R, Soranzo N, Jackson AU, Wheeler E, Glazer NL, Bouatia-Naji N, Gloyn AL, Lindgren CM, Magi R, Morris AP, Randall J, Johnson T, Elliott P, Rybin D, Thorleifsson G, Steinthorsdottir V, Henneman P, Grallert H, Dehghan A, Hottenga JJ, Franklin CS, Navarro P, Song K, Goel A, Perry JR, Egan JM, Lajunen T, Grarup N, Sparso T, Doney A, Voight BF, Stringham HM, Li M, Kanoni S, Shrader P, Cavalcanti-Proenca C, Kumari M, Qi L, Timpson NJ, Gieger C, Zabena C, Rocheleau G, Ingelsson E, An P, O'Connell J, Luan J, Elliott A, McCarroll SA, Payne F, Roccasecca RM, Pattou F, Sethupathy P, Ardlie K, Ariyurek Y, Balkau B, Barter P, Beilby JP, Ben-Shlomo Y, Benediktsson R, Bennett AJ, Bergmann S, Bochud M, Boerwinkle E, Bonnefond A, Bonnycastle LL, Borch-Johnsen K, Bottcher Y, Brunner E, Bumpstead SJ, Charpentier G, Chen YD, Chines P, Clarke R, Coin LJ, Cooper MN, Cornelis M, Crawford G, Crisponi L, Day IN, de Geus EJ, Delplanque J, Dina C, Erdos MR, Fedson AC, FischerRosinsky A, Forouhi NG, Fox CS, Frants R, Franzosi MG, Galan P, Goodarzi MO, Graessler J, Groves CJ, Grundy S, Gwilliam R, Gyllensten U, Hadjadj S, Hallmans G, Hammond N, Han X, Hartikainen AL, Hassanali N, Hayward C, Heath SC, Hercberg S, Herder C, Hicks AA, Hillman DR, Hingorani AD, Hofman A, Hui J, Hung J, Isomaa B, Johnson PR, Jorgensen T, Jula A, Kaakinen M, Kaprio J, Kesaniemi YA, Kivimaki M, Knight B, Koskinen S, Kovacs P, Kyvik KO, Lathrop GM, Lawlor DA, Le Bacquer O, Lecoeur C, Li Y, Lyssenko V, Mahley R, Mangino M, Manning AK, Martinez-Larrad MT, McAteer JB, McCulloch LJ, McPherson R, Meisinger C, Melzer D, Meyre D, Mitchell BD, Morken MA, Mukherjee S, Naitza S, Narisu N, Neville MJ, Oostra BA, Orru M, Pakyz R, Palmer CN, Paolisso G, Pattaro C, Pearson D, Peden JF, Pedersen NL, Perola M, Pfeiffer AF, Pichler I, Polasek O, Posthuma D, Potter SC, Pouta A, Province MA, Psaty BM, Rathmann W, Rayner NW, Rice K, Ripatti S, Rivadeneira F, Roden M, Rolandsson O, Sandbaek A, Sandhu M, Sanna S, Sayer AA, Scheet P, Scott LJ, Seedorf U, Sharp SJ, Shields B, Sigurethsson G, Sijbrands EJ, Silveira A, Simpson L, Singleton A, Smith NL, Sovio U, Swift A, Syddall H, Syvanen AC, Tanaka T, Thorand B, Tichet J, Tonjes A, Tuomi T, Uitterlinden AG, van Dijk KW, van Hoek M, Varma D, Visvikis-Siest S, Vitart V, Vogelzangs N, Waeber G, Wagner PJ, Walley A, Walters GB, Ward KL, Watkins H, Weedon MN, Wild SH, Willemsen G, Witteman JC, Yarnell JW, Zeggini E, Zelenika D, Zethelius B, Zhai G, Zhao JH, Zillikens MC, Borecki IB, Loos RJ, Meneton P, Magnusson PK, Nathan DM, Williams GH, Hattersley AT, Silander K, Salomaa V, Smith GD, Bornstein SR, Schwarz P, Spranger J, Karpe F, Shuldiner AR, Cooper C, Dedoussis GV, Serrano-Rios M, Morris AD, Lind L, Palmer LJ, Hu FB, Franks PW, Ebrahim S, Marmot M, Kao WH, Pankow JS, Sampson MJ, Kuusisto J, Laakso M, Hansen T, Pedersen O, Pramstaller PP, Wichmann HE, Illig T, Rudan I, Wright AF, Stumvoll M, Campbell H, Wilson JF, Bergman RN, Buchanan TA, Collins FS, Mohlke KL, Tuomilehto J, Valle TT, Altshuler D, Rotter JI, Siscovick DS, Penninx BW, Boomsma DI, Deloukas P, Spector TD, Frayling TM, Ferrucci L, Kong A, Thorsteinsdottir U, Stefansson K, van Duijn CM, Aulchenko YS, Cao A, Scuteri A, Schlessinger D, Uda M, Ruokonen A, Jarvelin MR, Waterworth DM, Vollenweider P, Peltonen L, Mooser V, Abecasis GR, Wareham NJ, Sladek R, Froguel P, Watanabe RM, Meigs JB, Groop L, Boehnke M, McCarthy MI, Florez JC, Barroso I: New genetic loci implicated in fasting glucose homeostasis and their impact on type 2 diabetes risk. Nat Genet 2010;42:105-116 3. Voight BF, Scott LJ, Steinthorsdottir V, Morris AP, Dina C, Welch RP, Zeggini E, Huth C, Aulchenko YS, Thorleifsson G, McCulloch LJ, Ferreira T, Grallert H, Amin N, Wu G, Willer CJ, Raychaudhuri S, McCarroll SA, Langenberg C, Hofmann OM, Dupuis J, Qi L, Segre AV, van Hoek M, Navarro P, Ardlie K, Balkau B, Benediktsson R, Bennett AJ, Blagieva R, Boerwinkle E, Bonnycastle LL, Bengtsson Bostrom K, Bravenboer B, Bumpstead S, Burtt NP, Charpentier G, Chines PS, Cornelis M, Couper DJ, Crawford G, Doney AS, Elliott KS, Elliott AL, Erdos MR, Fox CS, Franklin CS, Ganser M, Gieger C, Grarup N, Green T, Griffin S, Groves CJ, Guiducci C, Hadjadj S, Hassanali N, Herder C, Isomaa B, Jackson AU, Johnson PR, Jorgensen T, Kao WH, Klopp N, Kong A, Kraft P, Kuusisto J, Lauritzen T, Li M, Lieverse A, Lindgren CM, Lyssenko V, Marre M, Meitinger T, Midthjell K, Morken MA, Narisu N, Nilsson P, Owen KR, Payne F, Perry JR, Petersen AK, Platou C, Proenca C, Prokopenko I, Rathmann W, Rayner NW, Robertson NR, Rocheleau G, Roden M, Sampson MJ, Saxena R, Shields BM, Shrader P, Sigurdsson G, Sparso T, Strassburger K, Stringham HM, Sun Q, Swift AJ, Thorand B, Tichet J, Tuomi T, van Dam RM, van Haeften TW, van Herpt T, van Vliet-Ostaptchouk JV, Walters GB, Weedon MN, Wijmenga C, Witteman J, Bergman RN, Cauchi S, Collins FS, Gloyn AL, Gyllensten U, Hansen T, Hide WA, Hitman GA, Hofman A, Hunter DJ, Hveem K, Laakso M, Mohlke KL, Morris AD, Palmer CN, Pramstaller PP, Rudan I, Sijbrands E, Stein LD, Tuomilehto J, Uitterlinden A, Walker M, Wareham NJ, Watanabe RM, Abecasis GR, Boehm BO, Campbell H, Daly MJ, Hattersley AT, Hu FB, Meigs JB, Pankow JS, Pedersen O, Wichmann HE, Barroso I, Florez JC, Frayling TM, Groop L, Sladek R, Thorsteinsdottir U, Wilson JF, Illig T, Froguel P, van Duijn CM, Stefansson K, Altshuler D, Boehnke M, McCarthy MI: Twelve type 2 diabetes susceptibility loci identified through large-scale association analysis. Nat Genet 2010;42:579589 4. Kooner JS, Saleheen D, Sim X, Sehmi J, Zhang W, Frossard P, Been LF, Chia KS, Dimas AS, Hassanali N, Jafar T, Jowett JB, Li X, Radha V, Rees SD, Takeuchi F, Young R, Aung T, Basit A, Chidambaram M, Das D, Grundberg E, Hedman AK, Hydrie ZI, Islam M, Khor CC, Kowlessur S, Kristensen MM, Liju S, Lim WY, Matthews DR, Liu J, Morris AP, Nica AC, Pinidiyapathirage JM, Prokopenko I, Rasheed A, Samuel M, Shah N, Shera AS, Small KS, Suo C, Wickremasinghe AR, Wong TY, Yang M, Zhang F, Abecasis GR, Barnett AH, Caulfield M, Deloukas P, Frayling TM, Froguel P, Kato N, Katulanda P, Kelly MA, Liang J, Mohan V, Sanghera DK, Scott J, Seielstad M, Zimmet PZ, Elliott P, Teo YY, McCarthy MI, Danesh J, Tai ES, Chambers JC: Genome-wide association study in individuals of South Asian ancestry identifies six new type 2 diabetes susceptibility loci. Nat Genet 2011;43:984989 5. Musunuru K, Strong A, Frank-Kamenetsky M, Lee NE, Ahfeldt T, Sachs KV, Li X, Li H, Kuperwasser N, Ruda VM, Pirruccello JP, Muchmore B, Prokunina-Olsson L, Hall JL, Schadt EE, Morales CR, Lund-Katz S, Phillips MC, Wong J, Cantley W, Racie T, Ejebe KG, Orho-Melander M, Melander O, Koteliansky V, Fitzgerald K, Krauss RM, Cowan CA, Kathiresan S, Rader DJ: From noncoding variant to phenotype via SORT1 at the 1p13 cholesterol locus. Nature 2010;466:714-719 6. van der Harst P, Zhang W, Mateo Leach I, Rendon A, Verweij N, Sehmi J, Paul DS, Elling U, Allayee H, Li X, Radhakrishnan A, Tan ST, Voss K, Weichenberger CX, Albers CA, AlHussani A, Asselbergs FW, Ciullo M, Danjou F, Dina C, Esko T, Evans DM, Franke L, Gogele M, Hartiala J, Hersch M, Holm H, Hottenga JJ, Kanoni S, Kleber ME, Lagou V, Langenberg C, Lopez LM, Lyytikainen LP, Melander O, Murgia F, Nolte IM, O'Reilly PF, Padmanabhan S, Parsa A, Pirastu N, Porcu E, Portas L, Prokopenko I, Ried JS, Shin SY, Tang CS, Teumer A, Traglia M, Ulivi S, Westra HJ, Yang J, Zhao JH, Anni F, Abdellaoui A, Attwood A, Balkau B, Bandinelli S, Bastardot F, Benyamin B, Boehm BO, Cookson WO, Das D, de Bakker PI, de Boer RA, de Geus EJ, de Moor MH, Dimitriou M, Domingues FS, Doring A, Engstrom G, Eyjolfsson GI, Ferrucci L, Fischer K, Galanello R, Garner SF, Genser B, Gibson QD, Girotto G, Gudbjartsson DF, Harris SE, Hartikainen AL, Hastie CE, Hedblad B, Illig T, Jolley J, Kahonen M, Kema IP, Kemp JP, Liang L, Lloyd-Jones H, Loos RJ, Meacham S, Medland SE, Meisinger C, Memari Y, Mihailov E, Miller K, Moffatt MF, Nauck M, Novatchkova M, Nutile T, Olafsson I, Onundarson PT, Parracciani D, Penninx BW, Perseu L, Piga A, Pistis G, Pouta A, Puc U, Raitakari O, Ring SM, Robino A, Ruggiero D, Ruokonen A, Saint-Pierre A, Sala C, Salumets A, Sambrook J, Schepers H, Schmidt CO, Sillje HH, Sladek R, Smit JH, Starr JM, Stephens J, Sulem P, Tanaka T, Thorsteinsdottir U, Tragante V, van Gilst WH, van Pelt LJ, van Veldhuisen DJ, Volker U, Whitfield JB, Willemsen G, Winkelmann BR, Wirnsberger G, Algra A, Cucca F, d'Adamo AP, Danesh J, Deary IJ, Dominiczak AF, Elliott P, Fortina P, Froguel P, Gasparini P, Greinacher A, Hazen SL, Jarvelin MR, Khaw KT, Lehtimaki T, Maerz W, Martin NG, Metspalu A, Mitchell BD, Montgomery GW, Moore C, Navis G, Pirastu M, Pramstaller PP, Ramirez-Solis R, Schadt E, Scott J, Shuldiner AR, Smith GD, Smith JG, Snieder H, Sorice R, Spector TD, Stefansson K, Stumvoll M, Tang WH, Toniolo D, Tonjes A, Visscher PM, Vollenweider P, Wareham NJ, Wolffenbuttel BH, Boomsma DI, Beckmann JS, Dedoussis GV, Deloukas P, Ferreira MA, Sanna S, Uda M, Hicks AA, Penninger JM, Gieger C, Kooner JS, Ouwehand WH, Soranzo N, Chambers JC: Seventy-five genetic loci influencing the human red blood cell. Nature 2012;492:369-375 7. Dimas AS, Lagou V, Barker A, Knowles JW, Magi R, Hivert MF, Benazzo A, Rybin D, Jackson AU, Stringham HM, Song C, Fischer-Rosinsky A, Boesgaard TW, Grarup N, Abbasi FA, Assimes TL, Hao K, Yang X, Lecoeur C, Barroso I, Bonnycastle LL, Bottcher Y, Bumpstead S, Chines PS, Erdos MR, Graessler J, Kovacs P, Morken MA, Narisu N, Payne F, Stancakova A, Swift AJ, Tonjes A, Bornstein SR, Cauchi S, Froguel P, Meyre D, Schwarz PE, Haring HU, Smith U, Boehnke M, Bergman RN, Collins FS, Mohlke KL, Tuomilehto J, Quertemous T, Lind L, Hansen T, Pedersen O, Walker M, Pfeiffer AF, Spranger J, Stumvoll M, Meigs JB, Wareham NJ, Kuusisto J, Laakso M, Langenberg C, Dupuis J, Watanabe RM, Florez JC, Ingelsson E, McCarthy MI, Prokopenko I: Impact of type 2 diabetes susceptibility variants on quantitative glycemic traits reveals mechanistic heterogeneity. Diabetes 2014;63:2158-2171 8. Nica AC, Ongen H, Irminger JC, Bosco D, Berney T, Antonarakis SE, Halban PA, Dermitzakis ET: Cell-type, allelic, and genetic signatures in the human pancreatic beta cell transcriptome. Genome Res 2013;23:1554-1562 9. Lyssenko V, Lupi R, Marchetti P, Del Guerra S, Orho-Melander M, Almgren P, Sjogren M, Ling C, Eriksson KF, Lethagen AL, Mancarella R, Berglund G, Tuomi T, Nilsson P, Del Prato S, Groop L: Mechanisms by which common variants in the TCF7L2 gene increase risk of type 2 diabetes. J Clin Invest 2007;117:2155-2163 10. Taneera J, Lang S, Sharma A, Fadista J, Zhou Y, Ahlqvist E, Jonsson A, Lyssenko V, Vikman P, Hansson O, Parikh H, Korsgren O, Soni A, Krus U, Zhang E, Jing XJ, Esguerra JL, Wollheim CB, Salehi A, Rosengren A, Renstrom E, Groop L: A systems genetics approach identifies genes and pathways for type 2 diabetes in human islets. Cell Metab 2012;16:122-134 11. Ge B, Pokholok DK, Kwan T, Grundberg E, Morcos L, Verlaan DJ, Le J, Koka V, Lam KC, Gagne V, Dias J, Hoberman R, Montpetit A, Joly MM, Harvey EJ, Sinnett D, Beaulieu P, Hamon R, Graziani A, Dewar K, Harmsen E, Majewski J, Goring HH, Naumova AK, Blanchette M, Gunderson KL, Pastinen T: Global patterns of cis variation in human cells revealed by high-density allelic expression analysis. Nat Genet 2009;41:1216-1222 12. Heap GA, Yang JH, Downes K, Healy BC, Hunt KA, Bockett N, Franke L, Dubois PC, Mein CA, Dobson RJ, Albert TJ, Rodesch MJ, Clayton DG, Todd JA, van Heel DA, Plagnol V: Genome-wide analysis of allelic expression imbalance in human primary cells by highthroughput transcriptome resequencing. Hum Mol Genet 2010;19:122-134 13. Nicolae DL, Gamazon E, Zhang W, Duan S, Dolan ME, Cox NJ: Trait-associated SNPs are more likely to be eQTLs: annotation to enhance discovery from GWAS. PLoS Genet 2010;6:e1000888 14. Zhong H, Beaulaurier J, Lum PY, Molony C, Yang X, Macneil DJ, Weingarth DT, Zhang B, Greenawalt D, Dobrin R, Hao K, Woo S, Fabre-Suver C, Qian S, Tota MR, Keller MP, Kendziorski CM, Yandell BS, Castro V, Attie AD, Kaplan LM, Schadt EE: Liver and adipose expression associated SNPs are enriched for association to type 2 diabetes. PLoS Genet 2010;6:e1000932 15. Kwan T, Benovoy D, Dias C, Gurd S, Provencher C, Beaulieu P, Hudson TJ, Sladek R, Majewski J: Genome-wide analysis of transcript isoform variation in humans. Nat Genet 2008;40:225-231 16. Lalonde E, Ha KC, Wang Z, Bemmo A, Kleinman CL, Kwan T, Pastinen T, Majewski J: RNA sequencing reveals the role of splicing polymorphisms in regulating human gene expression. Genome Res 2011;21:545-554 17. Kulzer JR, Stitzel ML, Morken MA, Huyghe JR, Fuchsberger C, Kuusisto J, Laakso M, Boehnke M, Collins FS, Mohlke KL: A common functional regulatory variant at a type 2 diabetes locus upregulates ARAP1 expression in the pancreatic beta cell. Am J Hum Genet 2014;94:186-197 18. Cnop M, Abdulkarim B, Bottu G, Cunha DA, Igoillo-Esteve M, Masini M, Turatsinze JV, Griebel T, Villate O, Santin I, Bugliani M, Ladriere L, Marselli L, McCarthy MI, Marchetti P, Sammeth M, Eizirik DL: RNA sequencing identifies dysregulation of the human pancreatic islet transcriptome by the saturated Fatty Acid palmitate. Diabetes 2014;63:19781993 19. Eizirik DL, Sammeth M, Bouckenooghe T, Bottu G, Sisino G, Igoillo-Esteve M, Ortis F, Santin I, Colli ML, Barthson J, Bouwens L, Hughes L, Gregory L, Lunter G, Marselli L, Marchetti P, McCarthy MI, Cnop M: The human pancreatic islet transcriptome: expression of candidate genes for type 1 diabetes and the impact of pro-inflammatory cytokines. PLoS Genet 2012;8:e1002552 20. Fontanillas P, Landry CR, Wittkopp PJ, Russ C, Gruber JD, Nusbaum C, Hartl DL: Key considerations for measuring allelic expression on a genomic scale using high-throughput sequencing. Mol Ecol 2010;19 Suppl 1:212-227 21. Montgomery SB, Lappalainen T, Gutierrez-Arcelus M, Dermitzakis ET: Rare and common regulatory variation in population-scale sequenced human genomes. PLoS Genet 2011;7:e1002144 22. Flannick J, Thorleifsson G, Beer NL, Jacobs SB, Grarup N, Burtt NP, Mahajan A, Fuchsberger C, Atzmon G, Benediktsson R, Blangero J, Bowden DW, Brandslund I, Brosnan J, Burslem F, Chambers J, Cho YS, Christensen C, Douglas DA, Duggirala R, Dymek Z, Farjoun Y, Fennell T, Fontanillas P, Forsen T, Gabriel S, Glaser B, Gudbjartsson DF, Hanis C, Hansen T, Hreidarsson AB, Hveem K, Ingelsson E, Isomaa B, Johansson S, Jorgensen T, Jorgensen ME, Kathiresan S, Kong A, Kooner J, Kravic J, Laakso M, Lee JY, Lind L, Lindgren CM, Linneberg A, Masson G, Meitinger T, Mohlke KL, Molven A, Morris AP, Potluri S, Rauramaa R, Ribel-Madsen R, Richard AM, Rolph T, Salomaa V, Segre AV, Skarstrand H, Steinthorsdottir V, Stringham HM, Sulem P, Tai ES, Teo YY, Teslovich T, Thorsteinsdottir U, Trimmer JK, Tuomi T, Tuomilehto J, Vaziri-Sani F, Voight BF, Wilson JG, Boehnke M, McCarthy MI, Njolstad PR, Pedersen O, Groop L, Cox DR, Stefansson K, Altshuler D: Loss-of-function mutations in SLC30A8 protect against type 2 diabetes. Nat Genet 2014;46:357-363 23. Nicolson TJ, Bellomo EA, Wijesekara N, Loder MK, Baldwin JM, Gyulkhandanyan AV, Koshkin V, Tarasov AI, Carzaniga R, Kronenberger K, Taneja TK, da Silva Xavier G, Libert S, Froguel P, Scharfmann R, Stetsyuk V, Ravassard P, Parker H, Gribble FM, Reimann F, Sladek R, Hughes SJ, Johnson PR, Masseboeuf M, Burcelin R, Baldwin SA, Liu M, LaraLemus R, Arvan P, Schuit FC, Wheeler MB, Chimienti F, Rutter GA: Insulin storage and glucose homeostasis in mice null for the granule zinc transporter ZnT8 and studies of the type 2 diabetes-associated variants. Diabetes 2009;58:2070-2083 24. Cerretti DP, DuBose RF, Black RA, Nelson N: Isolation of two novel metalloproteinasedisintegrin (ADAM) cDNAs that show testis-specific gene expression. Biochem Biophys Res Commun 1999;263:810-815 25. Cras-Meneur C, Li L, Kopan R, Permutt MA: Presenilins, Notch dose control the fate of pancreatic endocrine progenitors during a narrow developmental window. Genes Dev 2009;23:2088-2101 26. Mina-Osorio P: The moonlighting enzyme CD13: old and new functions to target. Trends Mol Med 2008;14:361-371 27. Hofmann S, Philbrook C, Gerbitz KD, Bauer MF: Wolfram syndrome: structural and functional analyses of mutant and wild-type wolframin, the WFS1 gene product. Hum Mol Genet 2003;12:2003-2012 28. Bonnycastle LL, Chines PS, Hara T, Huyghe JR, Swift AJ, Heikinheimo P, Mahadevan J, Peltonen S, Huopio H, Nuutila P, Narisu N, Goldfeder RL, Stitzel ML, Lu S, Boehnke M, Urano F, Collins FS, Laakso M: Autosomal dominant diabetes arising from a Wolfram syndrome 1 mutation. Diabetes 2013;62:3943-3950 29. Osbak KK, Colclough K, Saint-Martin C, Beer NL, Bellanne-Chantelot C, Ellard S, Gloyn AL: Update on mutations in glucokinase (GCK), which cause maturity-onset diabetes of the young, permanent neonatal diabetes, and hyperinsulinemic hypoglycemia. Hum Mutat 2009;30:1512-1526 30. Dadi PK, Vierra NC, Ustione A, Piston DW, Colbran RJ, Jacobson DA: Inhibition of Pancreatic beta-Cell Ca2+/Calmodulin-dependent Protein Kinase II Reduces Glucosestimulated Calcium Influx and Insulin Secretion, Impairing Glucose Tolerance. J Biol Chem 2014;289:12435-12445 31. Tabuchi H, Yamamoto H, Matsumoto K, Ebihara K, Takeuchi Y, Fukunaga K, Hiraoka H, Sasaki Y, Shichiri M, Miyamoto E: Regulation of insulin secretion by overexpression of Ca2+/calmodulin-dependent protein kinase II in insulinoma MIN6 cells. Endocrinology 2000;141:2350-2360 32. Wang Z, Ramanadham S, Ma ZA, Bao S, Mancuso DJ, Gross RW, Turk J: Group VIA phospholipase A2 forms a signaling complex with the calcium/calmodulin-dependent protein kinase IIbeta expressed in pancreatic islet beta-cells. J Biol Chem 2005;280:6840-6849 33. Rochlitz H, Voigt A, Lankat-Buttgereit B, Goke B, Heimberg H, Nauck MA, Schiemann U, Schatz H, Pfeiffer AF: Cloning and quantitative determination of the human Ca2+/calmodulin-dependent protein kinase II (CaMK II) isoforms in human beta cells. Diabetologia 2000;43:465-473 34. Wynder C, Hakimi MA, Epstein JA, Shilatifard A, Shiekhattar R: Recruitment of MLL by HMG-domain protein iBRAF promotes neural differentiation. Nat Cell Biol 2005;7:11131117 35. Ceballos-Chavez M, Rivero S, Garcia-Gutierrez P, Rodriguez-Paredes M, GarciaDominguez M, Bhattacharya S, Reyes JC: Control of neuronal differentiation by sumoylation of BRAF35, a subunit of the LSD1-CoREST histone demethylase complex. Proc Natl Acad Sci U S A 2012;109:8085-8090 36. Nica AC, Parts L, Glass D, Nisbet J, Barrett A, Sekowska M, Travers M, Potter S, Grundberg E, Small K, Hedman AK, Bataille V, Tzenova Bell J, Surdulescu G, Dimas AS, Ingle C, Nestle FO, di Meglio P, Min JL, Wilk A, Hammond CJ, Hassanali N, Yang TP, Montgomery SB, O'Rahilly S, Lindgren CM, Zondervan KT, Soranzo N, Barroso I, Durbin R, Ahmadi K, Deloukas P, McCarthy MI, Dermitzakis ET, Spector TD: The architecture of gene regulatory variation across multiple human tissues: the MuTHER study. PLoS Genet 2011;7:e1002003 37. Emilsson V, Thorleifsson G, Zhang B, Leonardson AS, Zink F, Zhu J, Carlson S, Helgason A, Walters GB, Gunnarsdottir S, Mouy M, Steinthorsdottir V, Eiriksdottir GH, Bjornsdottir G, Reynisdottir I, Gudbjartsson D, Helgadottir A, Jonasdottir A, Styrkarsdottir U, Gretarsdottir S, Magnusson KP, Stefansson H, Fossdal R, Kristjansson K, Gislason HG, Stefansson T, Leifsson BG, Thorsteinsdottir U, Lamb JR, Gulcher JR, Reitman ML, Kong A, Schadt EE, Stefansson K: Genetics of gene expression and its effect on disease. Nature 2008;452:423-428 38. Dimas AS, Deutsch S, Stranger BE, Montgomery SB, Borel C, Attar-Cohen H, Ingle C, Beazley C, Gutierrez Arcelus M, Sekowska M, Gagnebin M, Nisbett J, Deloukas P, Dermitzakis ET, Antonarakis SE: Common regulatory variation impacts gene expression in a cell type-dependent manner. Science 2009;325:1246-1250 65 lead SNPs published by DIAGRAM consortium 1 Proxy SNPs identified using r2>0.8, Phase 1 1000 Genomes Project, CEU population 1590 SNPs (lead + proxy SNPs) SNP annotation using SeattleSeq Annotation 138 45 exonic SNPs in 23 RefSeq genes TaqMan SNP genotyping assay maps 100% to exonic sequence 36 exonic SNPs in 18 RefSeq genes. One SNP assay per gene tested 36 genomic DNAs genotyped 16 SNPs with ≥5 heterozygotes Allelic expression measured using human islet RNA from 36 donors 14 assays where expression could be robustly detected (Ct values <36) Figure 1 – Systematic identification of exonic SNPs suitable for allelic expression analysis in human islets. A B 2 -2 1.5 1.5 1 0.5 0 -0.5 R² = 0.67 R² = 0.72 0 -1 -1.5 -2 log2 risk/non-risk allele expression Run 1 2 log2 risk/non-risk allele expression SNP 2 log2 risk/non-risk allele expression Run 2 2 1 0.5 0 -2 0 2 -0.5 -1 -1.5 log2 risk/non-risk allele expression SNP 1 Figure 2 – TaqMan SNP Genotyping assays as a robust method to determine allelic expression. A: Correlation between allelic expression measurements determined from independent cDNAs reverse transcribed and amplified on different days. B: Correlation between allelic expression measurements calculated from SNPs in high linkage disequilibrium with each other and residing within the same gene. A C B 1.5 1 2 D 1 0.4 0.5 0.2 0 0 0 -0.5 -0.2 -0.5 -1 -0.4 G H 1.5 1 0.5 0.5 0 log2 risk/non-risk allelic expression -0.5 E F 0.4 0.5 0.4 0.4 0.2 0 0.2 -0.5 0 -1 -0.2 0 -0.2 -0.4 -0.6 I 0.6 0.2 0 -0.2 -0.4 -0.6 -1.5 -0.4 -0.8 J K L 0.4 0.8 0.4 0.6 0.2 0.6 0.2 0.4 0.4 0 0.2 -0.2 0 -0.4 -0.8 -0.2 -0.6 -0.2 -1 -0.4 -0.8 -0.4 0 -0.2 -0.4 -0.6 0.2 0 Figure 3 – Significant allelic expression imbalance (AEI) identified for SNPs in 7 genes. A: rs17240240 (ANPEP), B: rs41276922 (ANPEP), C: rs5219 (KCNJ11), D: rs5215 (KCNJ11), E: rs699779 (NOTCH2), F: rs835575 (NOTCH2), G: rs11558471 (SLC30A8), H: rs3802177 (SLC30A8), I: rs1046320 (WFS1), J: rs1801206 (WFS1), K: rs1065359 (CAMK2B), L: rs952472 (HMG20A). White circles = genomic DNA; white squares = cDNA. Dotted lines show paired genomic DNA:cDNA samples. log2 relative expression A B C D * Number of risk alleles Figure 4 –rs2007084 genotype is significantly associated with (A) ANPEP expression but not expression of neighbouring genes (B) AP3S2, (C) ARPIN, (D) MESP1 in human islet tissue (n=36). *p<0.05 (T-test). Table 1 – Allelic expression results for 14 type 2 diabetes GWAS implicated genes DIAGRAM Transcribed SNP (gene) lead SNP p Adjusted* Linkage Mean Unadjusted lead allelic value p value transcribed expression SNP r2 (D’) rs10923931 rs699779 (NOTCH2) 0.99 (1.00) 0.79 4.8 x 10-5 9.1 x 10-4 rs5215 rs5219 (KCNJ11) 1.00 (1.00) 61.43 1.3 x 10-4 1.2 x 10-3 rs2007084 rs17240240 (ANPEP) 0.86 (0.95) 1.50 7.4 x 10-4 4.7 x 10-3 rs7177055 rs952472 (HMG20A) 0.91 (0.99) 1.14 2.1 x 10-3 6.7 x 10-3 rs10278336 rs1065359 (CAMK2B) 0.99 (1.00) 0.82 2.6 x 10-3 7.1 x 10-3 rs3802177 rs11558471 (SLC30A8) 0.97 (1.00) 1.12 6.2 x 10-3 1.5 x 10-2 rs4458523 rs1046320 (WFS1) 0.95 (0.99) 0.92 6.9 x 10-3 1.5 x 10-2 rs12427353 rs55834942 (HNF1A) 0.95 (0.99) 0.92 0.10 0.15 rs10203174 rs7578597 (THADA) 0.91 (0.98) 1.04 0.19 0.26 rs10401969 rs72999033 (HAPLN4) 0.90 (1.00) 1.13 0.27 0.34 rs7202877 rs13337017 (BCAR1) 0.93 (0.97) 1.07 0.29 0.34 rs516946 rs750625 (ANK1) 0.96 (0.99) 0.94 0.45 0.50 rs1552224 rs11603334 (ARAP1) 1.00 (1.00) 1.05 0.54 0.57 rs5215 rs11024271 (NCR3LG1) 0.88 (0.96) 0.97 0.65 0.65 rs2007084 rs41276922 (ANPEP) 0.83 (0.95) 1.51 1.6 x 10-3 6.7 x 10-3 rs10923931 rs835575 (NOTCH2) 0.99 (1.00) 0.70 1.9 x 10-3 6.7 x 10-3 rs5215 rs5215 (KCNJ11) 1.00 (1.00) 1.12 1.6 x 10-2 3.0 x 10-2 rs4458523 rs1801206 (WFS1) 0.93 (0.99) 0.93 3.1 x 10-2 5.4 x 10-2 rs3802177 rs3802177 (SLC30A8) 1.00 (1.00) 1.05 3.9 x 10-2 6.2 x 10-2 Validation SNPs exhibiting significant AEI (unadjusted p<0.05) shown in bold.*Adjusted using Benjamini-Hochberg false discovery rate