Micropipette Use Activity for Students
advertisement

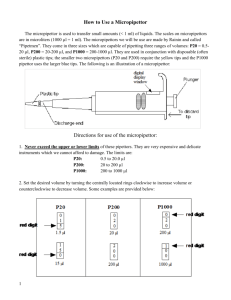
Use of Micropipettors Micropipettors are instruments used to accurately transfer small volumes (1 l to 1 ml) of solution. Because of their accuracy, ease of use, and convenience in sterile techniques they are a nearly universal lab tool. In this lab you will learn how to properly use this instrument. Use the terms and the illustration below to become familiar with the parts of a micropipettor: TERMS Barrel or shaft - the working end of the micropipettor; seat a disposable tip on the lower end of the barrel before each use. Plunger – press the plunger to withdraw liquid. Release it to expel liquid. Tip eject button - press to remove a tip from the barrel without direct handling of the disposable tip. Volume readout - displays the volume the micropipettor is currently set to deliver. Volume adjustment knob - rotate to change the digital volume setting. Image adapted from: http://www.di.uq.edu.au/sparqmicropipette On the lab bench, you will find four micropipettors; each is appropriate for a specific volume range. If you look at the dot on the plunger of each micropipettor you will see a number that represents the maximum volume, in microliters (l), that can be transferred by that micropipettor. The minimum volume appropriate for each micropipettor is typically ten percent of the maximum. The dot on the plunger is also color-coded and often matches the color of the disposable tips used with that micropipettor. The table below shows the volume range, expected accuracy, and the appropriate tips for the micropipettors that you will be using. Micropipettor P1000 P200 P20 P10 Volume Range (l) 100-1000 20-200 1-20 1-10 Disposable Tip Accuracy (l) 10.0 1.0 0.5 ± 0.5 Blue Yellow Yellow Clear Adapted from: DNA Science, 2003 by Cold Spring Harbor Laboratory Press, purchased from Carolina Biological. In most experiments, accuracy is important when transferring small volumes of liquid with a micropipettor. A researcher needs to be sure that he or she is transferring the volume desired with a reasonable degree of accuracy. The researcher can be confident of this provided two conditions are satisfied. First, the micropipettor has been calibrated and tested for accuracy (this is usually done on a yearly or semi-yearly basis). Second, the researcher must be using the micropipettor properly. Part 1: Familiarization with the micropipettor Materials: set of micropipettors Pick up a P20 and set the digital volume setting for 5 l (reading down, the setting should be "050'"). Push the plunger down and notice that at some point the plunger becomes more resistant and requires more effort to push further. This point is called the first stop. Notice that the plunger can be pushed well beyond the first stop until it reaches "the second stop". Be sure you can feel the first stop point and notice how far the plunger travels before reaching this point. Reset the micropipettor to 20 l ("200"). Again push the plunger to the first stop. You should notice now that the plunger travels further to reach the first stop than it did when the micropipettor was set for 5 I. Examine the P10, P200, and P1000 setting windows as well. The table below shows how the volume display looks for each micropipettor when it is set at its maximum volume. Micropipettor P1000 P200 P20 P10 Maximum Setting 1 0 0 2 0 0 2 0 0 1 0 0 Look at the diagram below, which shows how to read the volume of a micropipettor. Image adapted from: http://faculty.buffalostate.edu/wadswogj/courses/BIO211%20Page/Resources/micropipetting%20lab.pdf Practice determining the volume on the diagram below. Write the correct volume for each micropipette. P10 P20 P200 P1000 0 5 6 1 3 2 1 8 8 0 4 7 volume volume volume volume Take up sample with micropipette: 1. Set pipet volume by rotating the volume adjustment knob. Note: the volume setting should never be adjusted above the maximum volume specified for a particular micropipettor. Remember that the maximum volume is the volume shown on the plunger. 2. Seat a disposable tip on the micropipettor by placing the end of the barrel into a tip. To avoid contamination, use a new tip for each transfer. 3. Depress plunger to first stop and hold at that spot. 4. Place tip into the liquid to be pipetted. 5. Slowly release plunger to draw up liquid. Do not release too quickly or you will create air bubbles. 6. Wipe any excess droplets on side of container, but do not touch the tip. Release sample from micropipette: 1. Put tip into tube and touch the inner wall of tube with tip. 2. Press plunger down to the second stop and hold it there. 3. Pull micropipette out of tube and release plunger once it is out of tube. This prevents you from inadvertently drawing in liquid. 4. Eject the used tip. Place the tip over the appropriate waste container and press the tip ejector button. If the tip is difficult to eject it is likely that you are jamming the tips onto the micropipettor harder than necessary. Part 2: Transferring small volumes with a micropipettor Materials: Piece of blotting paper 2X Loading dye P20 and rack of yellow tips P10 and rack of clear tips Obtain a 5 x 5-inch piece of blotting paper and a microcentrifuge tube containing 2X loading dye. Follow the protocol above and use a P10 to spot the following volumes of dye directly onto the paper in a linear order: 1 l, 3 l, 5 l, and 7 l. Repeat using the P20 for: 10 l, 15 l, and 20 l. Repeat in a second row directly below the first. Compare your first and second rows to check your consistency. Compare the spots you made with those of other students. If the spots do not look like those of the instructor, try to determine the cause. Part 3: Transferring large volumes with a micropipettor This exercise simulates parts of a bacterial transformation or plasmid preparation for which a 100 - 1000 l micropipettor is used. It is far easier to mismeasure when using a large-volume micropipettor. If the plunger is not released slowly, an air bubble may form or solution may be drawn into the piston. Materials: 2 x 1.5 ml microcentrifuge tubes Solutions I - IV P1000 and rack of blue tips 1. Use a permanent marker to label two 1.5 ml reaction tubes E and F. 2. Use matrix below as a checklist while adding solutions to each reaction tube. Tube E F Solution I 100 l 150 l Solution II 200 l 250 l Solution III 150 l 350 l Solution IV 550 l 250 l 3. Set micropipettor to add appropriate volumes of Solutions I-IV to tubes E and F. Follow the same procedure as for small-volume pipettor. 4. A total of 1000 l of reactants was added to each tube. To check that your measurements were accurate, set the micropipettor to 1000 l and carefully withdraw solution from each tube. a. Is the tip just filled? or b. Is a small volume of fluid left in the tube? or c. After extracting all fluid, is an air space left in tip's end? (The air can be displaced and actual volume determined simply by rotating the volume adjustment to push the fluid to the very end of the tip. Then, read the volume directly.) 5. If the measurements were inaccurate, repeat the exercise to obtain a nearly perfect result. Part 4: Pipette-Mixing Activity Mixing two liquids together is an essential laboratory skill. Sometimes you turn the task over to a vortexer—a small vibrating plate. Other times, you need to create a more gentle and controlled mixing motion. Therefore, you need to be able to mix two solutions with a micropipette. When pipette-mixing, you need to balance mixing a solution completely with not mixing too vigorously. Over-zealous suction can pull solution into the barrel of the pipette (cleaning a pipette barrel is an expensive process!) This activity allows you to see and improve your pipette mixing skills for your upcoming lab activities. Materials: 1.5 ml microcentrifuge tube Blue and yellow loading dyes P-200 and rack of yellow tips 1. Add yellow loading dye. Using a P-200 micropipette, add 100 ul of yellow loading dye into a 1.5 ml microcentrifuge tube. 2. Add blue loading dye. Add 100 ul of blue loading dye into the same microcentrifuge tube as the yellow dye. Insert the pipette tip the whole way to the bottom of the tube and expel the blue dye underneath the yellow dye. 3. Homogenize dyes. In order to make mixing easier, reset your P-200 to 200 ul so that you can suck up and expel the entire contents of the tube at once. This will also allow you to check your pipetting accuracy, as you did in the previous activity. Mix the contents of the tube together until completely homogenized by slowly stirring with the pipette while sucking up and expelling the dyes in the tube. Once finished, the dye solution should be completely green with no blue or yellow swirls.