LAWRENCE TECHNOLOGICAL UNIVERSITY, DECEMBER 2012
advertisement

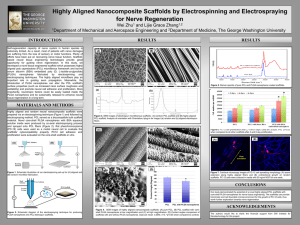
LAWRENCE TECHNOLOGICAL UNIVERSITY, DECEMBER 2012 1 Surface Modification of Electrospun PCL Fibers for Enhanced Cell Adhesion and Proliferation Ahmad Arabi, Emily Boggs, Manan Patel, LTU Biomedical Engineering Department ABSTRACT: Polycaprolactone (PCL) is gaining popularity in the field of tissue engineering due to its non-toxic degradation byproducts and low-cost manufacturing method. However, it is also a markedly hydrophobic material, leading to suboptimal cell-material interactions. Our study aims to chemically modify electrospun PCL fibers to promote cell attachment, proliferation and matrix formation. Our research examines a two-step technique used to modify the surface of PCL to increase cell adhesion and proliferation. One method immobilizes arginine-glycine-aspartic acid (RGD) peptides by introducing functional groups on the PCL surface. The other method utilizes sodium hydroxide (NaOH) treatment to create surface nanotopography with improved hydrophilicity. Changes in the PCL fiber surface topography, hydrophilicity and chemistry will be evaluated using scanning electron microscopy (SEM), contact angle measurement, x-ray photoelectron spectroscopy (XPS), and infrared spectroscopy (IR). Human periodontal ligament fibroblast (HPDLF) cells will be seeded on the PCL fibers and their growth and proliferation will be characterized by DAPI (4', 6-diamidino-2-phenylindole) staining and a Live/Dead Assay using confocal microscopy and, AlamarBlue® Assay. Our preliminary results have shown decreased surface contact angle and increased surface roughness with NaOH-treated PCL fibers. Ongoing study is focused on the RGD modification and cellular response to the NaOH and RGD treated fibers as compared to buffer -treated controls. We anticipate increased fibroblast adhesion, proliferation, and matrix formation on the treated PCL fibers. The results will have important implications for the use of PCL fibers as scaffold materials in regenerating a variety of tissues. LAWRENCE TECHNOLOGICAL UNIVERSITY, DECEMBER 2012 2 BACKGROUND Polycaprolactone (PCL) is a synthetic aliphatic polymer. It has excellent biocompatibility in a sense that when it degrades, it produces non-toxic byproducts but has poor biocompatibility in a sense that it has poor cell-material interaction and thus cells will adhere on its surface. It also has low cost and easy processability, making it useful for a number of biomedical applications such as drug delivery systems and other tissue engineering applications. Electrospun PCL fibers are especially attractive as a scaffold material because they mimic the fibrous structure of the extracellular matrix (ECM) of many tissues. A scaffold used for tissue engineering can be considered an artificial extracellular matrix since it provides a surface for the cells to grow on. However, electrospun PCL has a hydrophobic surface due to the lack of functional groups and thus will not promote cell adhesion. Preliminary studies have shown that the water contact angle, which is a measure of hydrophilicity, of unmodified PCL fibers is about 82○. This is considered fairly hydrophobic and literature has shown that cell like hydrophilic surfaces, which is water contact angle of 50○ or less. It is now generally accepted in biomaterial community that both very hydrophilic and hydrophobic surfaces are not ideal for cell adhesion. Rather, a moderate hydrophilicity enables the polymer to adsorb proteins [9]. Surface modification of PCL is necessary for its use in tissue engineering due to these challenges. Various approaches have been proposed for modifying synthetic hydrophobic biomaterial polymer surfaces. One of the methods is Sodium Hydroxide (NaOH) treatment which causes the base hydrolysis of esters bonds in PCL fibers creating carboxylate ions (Figure 1). This creation of functional group will increase hydrophilicity of the fibers as well as provide means to covalently attach a bioactive compound to improve the cell-material interaction. NaOH treatment also creates nanoroughness which results in increase of surface area which will result in enhanced cell adhesion. . Literature has shown that nanotopography is beneficial for enhanced cellular attachment [2]. LAWRENCE TECHNOLOGICAL UNIVERSITY, DECEMBER 2012 3 Fig.1 Reaction of the base hydrolysis of esters Another approach is to immobilize RGD peptide on biomaterial surface. Immobilization is essential as it will create a strong anchoring or RGD peptide on surface which ultimately leads to enhanced cell adhesion [6]. RGD peptide is a common motif present in the ligands of extracellular matrix proteins like fibronectin. Immobilizing this peptide onto the PCL fibers will result in the binding of cells to the surface via integrins, which are present on the cell surface. Integrins recognize the RGD motif and bind to it, thus attaching the cell to the surface. RGD will be immobilized on the PCL surface using 1-ethyl-3-(3dimethylaminopropyl) carbodiimide (EDC) chemistry. Other surface modification methods include polyethylene glycol (PEG) treatment, ionized gas (plasma) treatments, chemical treatments as well as various mechanical modification approaches to creating nanotopography [1] but these methods are not feasible for our project and therefore were not considered. RESEARCH PLAN The research objective of this project is to find an optimized set of parameters for each of the two modification methods to achieve optimum cell adhesion, proliferation, and matrix formation. In this project, we will be modifying PCL fibers via two step modification method in order to achieve a more controlled cell-scaffold interaction. The first step is involves NaOH treatment which nano scale roughness by making the individual fibers rougher. The second approach involves chemical immobilization of RGD on the PCL surface. The carboxylate ion produced in step one will be treated first with EDC and then with sulfo-NHS to create a semi-stable amine reactive ester. Further reaction of this NHS ester with the amine group of RGD peptide results in a stable amide bond, which is strong enough to covalently bond RGD on PCL surface. LAWRENCE TECHNOLOGICAL UNIVERSITY, DECEMBER 2012 The modified PCL fibers will be characterized for change in topography, surface chemistry, and hydrophilicity using SEM, FT-IR and XPS, and Goniometer respectively. We will also conduct a systematic study of cellular response to the modified fibers. Human periodontal ligament fibroblasts will be used to perform this study. The cell attachment, proliferation, and protein synthesis will be evaluated by DAPI staining and a Live/Dead Assay using confocal microscopy and cellular metabolism studies will be done using AlamarBlue® Assay. Fig. 2 Research plan flowchart METHODS Surface modification techniques and protocols: PCL Fibers were treated with 0.5M and 1M concentrations of NaOH for 1 and 3 hour time periods. Multiple samples for each trial were used. Trials were done to find an optimal NaOH concentration and time to be 1M NaOH and 3 hours. These parameters the best combination creating a contact angle of 46o, as shown in the figure below (Figure 3). We were expecting a contact angle of 50 o and our preliminary study provided good results. 4 LAWRENCE TECHNOLOGICAL UNIVERSITY, DECEMBER 2012 Fig. 3 PCL fiber treated w/ 1M NaOH for 3 hours We have investigated two methods of RGD immobilization. One involves the introduction of free amine groups by Aminolysis [3], and the other uses 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC) chemistry. The second method, though more complicated than the first, is a low-cost, highefficiency, and high-strength immobilization method which allows for the immobilization of RGD along with attachment of important homing or differentiation factors in future studies. The EDC method requires a carboxylate ion, as shown in the figure below (Figure 4). This means that the fiber must initially be treated with NaOH to create the carboxylates. The method then involves the reaction of carboxylate with EDC, creating an unstable reactive O-acylisourea ester. Sulfo-NHS is then added to stabilize the intermediate which converts the unstable O-acylisourea into an amine-reactive NHS ester. This ester will react with the amino end of the RGD peptide to form a stable covalent amide bond between the PCL fiber and the RGD peptide. Fig. 4 Reaction of the immobilization of RGD on PCL surface by EDC chemistry 5 LAWRENCE TECHNOLOGICAL UNIVERSITY, DECEMBER 2012 6 Surface characterization techniques: The following techniques enable the evaluation of changes in hydrophilicity, surface topography, and surface chemistry as a result of the two step modifications. Water contact angle measurement will be done on buffered control fibers and modified fibers to evaluate the hydrophilicity with help of a goniometer. Previous literature [9] states that a decrease in water contact angle to 50o is an optimal improvement in hydrophilicity. For our pilot study, the ImageJ program was used to find the water contact angle on high resolution photos of the PCL fibers. In January, a goniometer at GM will be used for more accurate contact angle measurement. SEM characterization provides detailed visual information regarding the morphology of the PCL fibers. The images will be analyzed for change in fiber diameter and roughness. An infrared spectrometer will be used to evaluate the modified film surface chemical composition by recognizing the functional groups that are present on the surface. On the IR spectrometer, the lens will be covered with the sample, and the spectrometer will pass infrared radiation through the sample at different wavelengths (2.5-15 µm). The computer composes a spectrum using the absorption of specific wavelength energies of the functional groups of the compound. We expect to see additional peaks in the functionalized PCL at 1650 and 1540 cm-1 indicating the presence of amide bonds. XPS determines the chemical composition of a surfaces top several nanometers. An electron energy analyzer determines the energy of emitted photoelectrons when the sample is exposed to a beam of x-rays. A photoelectron peak is then created with a unique binding energy and intensity that can quantify the functional groups on the polymer surface. Detection of nitrogen and carbons from C=O bonds means that RGD is present on the surface. XPS analysis will be done before and after treatment of PCL at Oakland University. Cell Culture and Seeding: The cells being used for this project are human periodontal ligament fibroblasts (HPDLFs) – ligament cells found in the jaw muscle. These cells were chosen for their hardiness and nearindestructability. In addition, fibroblasts are the main type of cell found on the ACL. They are responsible LAWRENCE TECHNOLOGICAL UNIVERSITY, DECEMBER 2012 7 for secretion of the ligament’s extracellular matrix; a measure of success for the scaffold would be if the fibroblasts seeded onto it also began secreting an extracellular matrix. The cells are first grown on a culture plate and later seeded onto the modified PCL fibers (a control groups will also be seeded onto an unmodified scaffold). Analysis will be done at one and two weeks for the extent of cell growth and proliferation. Cell characterization techniques: For quantifying cell growth and migration, confocal fluorescent microscopy and DAPI (4',6diamidino-2-phenylindole) will be used. DAPI is a fluorescent stain that binds to adenine and thymine molecules in DNA. As a result, the application of DAPI to a cell culture will cause the cells’ nuclei to glow in fluorescent light. A confocal microscope will be used for viewing seeded cells as the three dimensional nature of the images produced can lend to quantitatively measuring the depth of cell penetration into the scaffold. A Live/Dead Assay makes use of fluorescent dyes; viable cells are stained green and dead cells are stained red (Figure 5). With the use of a confocal microscope, the dyed cells can be studied in situ for overall viability, as well as distribution and density on the scaffold. The assay will also lend to a quantitative measure of cell concentration. An AlamarBlue® assay will be performed to study the metabolism and growth kinetics of the cellseeded scaffolds vs. controls. The assay consists of a reagent that, when taken up by metabolically active cells, forms a fluorescent product. The amount of metabolic activity can thus be correlated with the intensity of the fluorescence, which is measured via spectroscopy. The measured metabolic activity can also be correlated to cell concentrations; by comparing the results at the 1 week and 2 week analysis time points, a rough estimation of cell growth on the samples can be inferred. LAWRENCE TECHNOLOGICAL UNIVERSITY, DECEMBER 2012 8 Fig. 5 Live/Dead Assay of PCL fiber seeded with cells Controls: Unmodified buffer-treated PCL fibers will also be done with every test and characterization technique for comparison studies with the NaOH and RGD modified fibers. Surface characterization methods will be done on the buffer-treated fibers. They will also be seeded with cells and characterized with the same cell characterization assays done on the treated fibers. Design: 72 PCL fibers will be needed for this experiment. 24 buffer treated (controls), 24 NaOH modified, and 24 NaOH and RGD modified will be used. From each scaffold type, four will be used for surface characterization studies; two for the contact angle measurement and FT-IR, and the other two for XPS. This is necessary since these techniques are destructive. The other 20 will be used for cell seeding; 10 for DAPI staining and Live/Dead assay, and 10 for AlamarBlue® Assay. Since we are analyzing cell growth at 1 and 2 week analysis, 5 scaffolds will be used for week 1 and 5 for week 2. These fibers will be used to complete the datasheet below summarizing the final results (Figure 6). LAWRENCE TECHNOLOGICAL UNIVERSITY, DECEMBER 2012 SCAFFOLD ONLY Buffer treated Trial #1 Water contact angle #1 #2 Infrared Spectroscopy (IR) #1 #2 9 XPS #1 #2 Trial #2 Trial #3 NaOH #1 #2 #1 #2 #1 #2 #1 #2 #1 #2 #1 #2 Trial #1 Trial #2 Trial #3 NaOH + RGD Trial #1 Trial #2 Trial #3 CELLS ON SCAFFOLD Buffer treated Trial #1 Trial #2 Trial #3 Confocal Microscopy: DAPI/LiveDead/Alamar Blue Assay #1 #2 #3 #4 #5 NaOH Trial #1 Trial #2 Trial #3 #1 #2 #3 #4 #5 NaOH + RGD Trial #1 Trial #2 Trial #3 #1 #2 #3 #4 #5 Fig. 6 Table to be used in data collection and analysis. In January, contact angle measurements will be done using a Goniometer, as well as a pilot study for the RGD to create a protocol. Cell culture on the culture plate will be practiced to establish standard operating procedure since we have not performed these techniques before. In February, cell-seeding studied on a new set of fibers will be done and data will be collected from the surface characterization studies. In March, cell characterization studies will be performed with simultaneous data collection. In April, the data will be analyzed for statistical significance. We will also prepare posters for conferences and a manuscript to be submitted to a biomaterials journal. CHALLENGES The most difficult challenges that will be encountered will involve the technical intricacies of certain cell modification and characterization methods. Already, we have had some difficulties obtaining readable images for measuring the water contact angle. Also, no protocols exist for treating electrospun PCL fibers; we anticipate the need to modify existing protocols and evaluation assays to make things work for our study. LAWRENCE TECHNOLOGICAL UNIVERSITY, DECEMBER 2012 10 DESIRED OUTCOMES We expect to find an optimized set of parameters for NaOH and RGD peptide treatments to achieve the greatest cell adhesion, proliferation, and matrix formation. We aim to find the optimum concentration of NaOH and time of treatment needed for an effective modification which will keep the bulk properties of the PCL intact. We also expect to find ideal time and concentration of RGD required for optimum cell adhesion. We also expect to establish a complete set of protocols on surface modification, characterization and evaluation of cellular response. FUTURE DIRECTIONS / IMPLICATIONS We aim to create a set of methods for surface modification to enhance cell adhesion and proliferation. These results will have important implications for the use of these fibers for scaffold use in the regeneration of tissues such as ligaments, since PCL fibers have greater tensile strength than their film counterparts. Future studies, using these optimized modification protocols, may focus on the behavior of cells on PCL fibers undergoing mechanical stimulation. The modified PCL fibers also provide model surfaces for us to further investigate the behavior of other types of cells in response to nanotopography and RGD ligand. COST ANALYSIS The PCL fibers will be provided by the University of Michigan free of cost. Equipment, chemicals, and materials will be provided by Lawrence Tech. XPS will be done at Oakland University and the goniometer will be provided by GM next year. The major cost will be for the chemicals for modification and the various characterization assays. Specific costs for the chemicals are shown below: RGD peptide - $118/25 mg Sulfo NHS - $150/500 mg AlamarBlue® Assay kit - $164.82/25 mL The budget for this experiment is $2,000 which will be provided by Dr. Yawen Li. LAWRENCE TECHNOLOGICAL UNIVERSITY, DECEMBER 2012 ACKNOWLEGMENTS Faculty Advisor: Dr. Yawen Li. Technical Advisors: Dr. Julie Zweisler-Vollick and Tristan Maerz. Acknowledgments to Dr. Hsiao-Ping Moore (Lawrence Technological University), Dr. Kevin Baker (Beaumont Orthopedics Department), Sam Tuck, and Dr. Joseph Corey (University of Michigan). 11 LAWRENCE TECHNOLOGICAL UNIVERSITY, DECEMBER 2012 12 REFERENCES: [1] Goddard, J.M. (07/2007). "Polymer surface modification for the attachment of bioactive compounds". Progress in polymer science (0079-6700), 32 (7), p. 698. [2] Chen, Weiqiang (05/2012). "Nanotopography Influences Adhesion, Spreading, and Self-Renewal of Human Embryonic Stem Cells". ACS nano (1936-0851), 6 (5), p. 4094. [3] Zhu, Yabin (11/2002). "Surface Modification of Polycaprolactone Membrane via Aminolysis and Biomacromolecule Immobilization for Promoting Cytocompatibility of Human Endothelial Cells". Biomacromolecules (1525-7797), 3 (6), p. 1312. [4] Hartman, Olga (07/2010). "Biofunctionalization of electrospun PCL-based scaffolds with perlecan domain IV peptide to create a 3-D pharmacokinetic cancer model". Biomaterials (01429612),31 (21), p. 5700. [5] De Luca, A. C. (2012). "Immobilization of cell-binding peptides on poly-ε-caprolactone film surface to biomimic the peripheral nervous system". Journal of biomedical materials research. Part A(1549-3296), p. n. [6] Hersel, Ulrich (11/2003). "RGD modified polymers: biomaterials for stimulated cell adhesion and beyond". Biomaterials (0142-9612), 24 (24), p. 4385. [7] Zhang, Huina (09/2009). "The interaction between bone marrow stromal cells and RGD-modified three-dimensional porous polycaprolactone scaffolds". Biomaterials (0142-9612), 30 (25), p.4063. [8] Zhu, Yang (2013). "Aminolysis-based surface modification of polyesters for biomedical applications". RSC advances (2046-2069). [9] Ma, Zuwei, Zhengwei Mao, and Changyou Gao. "Surface Modification and Property Analysis of Biomedical Polymers Used for Tissue Engineering." Colloids and Surfaces B: Biointerfaces 60.2 (2007): 137-57. Print.