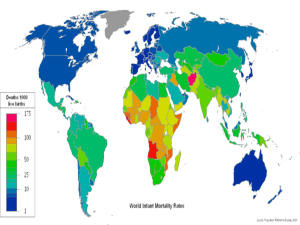
Ethics Approval Form 2015-16
advertisement

Conservatoires UK (CUK) Ethical Approval Form for staff and student questionnaire research with adults only Type of project: Title of project: Name of researcher(s): Name of supervisors(s): Date: STAFF / POSTGRADUATE / UNDERGRADUATE (delete as appropriate) [Click and type here] [Click and type here] [Click and type here] (for student research) [Click and type here] Mark with in box YES 1 Will you describe the purpose of the research at the outset, so that respondents are informed as to what to expect and how long the questionnaire will take to complete? 2 Will you tell respondents that their participation is voluntary? 3 Will you tell respondents that by completing and submitting their responses they are giving their informed consent? 4 Will you tell respondents that they have the option of omitting certain questions if they do not want to answer them? 5 Will you tell respondents that they can stop completing the questionnaire at any point for any reason? 6 Will you tell respondents that their data will be treated with full confidentiality and that, if published, it will not be identifiable as theirs? 7 Will you thank respondents at the end of the questionnaire? 8 Will you provide contact details for the researchers? NO N/A If you have marked No to any of Q1-8, but have marked Box A overleaf, please give an explanation on a separate sheet. (Note: N/A = not applicable). Mark with in box YES 9 Will your questionnaire involve deliberately misleading participants in any way? 10 Is there any realistic risk of any participants experiencing either physical or psychological distress or discomfort? If Yes, give details on a separate sheet and state what you will tell them to do if they should experience any problems (e.g. who they can contact for help). NO N/A If you have marked Yes to Q9 or 10 you should normally mark Box B overleaf; if not, please give a full explanation on a separate sheet. Mark with in box 11 Do respondents fall into any of the following special groups? If so, please refer to BPS or BERA guidelines, and mark Box B overleaf. You should ensure that you have DBS clearance. YES NO N/A School children (under 18 years of age) People with learning or communication difficulties Patients People in custody If you are conducting research with children, parental consent must be given. Please use the Ethical Approval Form for Research involving the participation of other people and follow the guidance notes attached. There is an obligation on the lead researcher to bring to the attention of the CUK Research Ethics Committee any issues with ethical implications not clearly covered by the above checklist. PLEASE MARK EITHER BOX A OR BOX B BELOW AND PROVIDE THE DETAILS REQUIRED IN SUPPORT OF YOUR APPLICATION. THEN PRINT AND SIGN THE FORM. Mark with A. I consider that this project has no significant ethical implications to be brought before the CUK Research Ethics Committee. Give a brief description of the purpose of the research, the questionnaire itself and the respondents who will complete the questionnaire (max. 150 words). [Click and type here] OR Mark with B. I consider that this project may have ethical implications that should be brought before the CUK Research Ethics Committee, and/or it will be carried out with children, vulnerable young people or vulnerable adults. Please provide all the further information listed below in a separate attachment. 1. 2. 3. 4. 5. 6. Title of project Purpose of project and its academic rationale Brief description of questionnaire Respondents: recruitment methods, number, age, sex, exclusion/inclusion criteria The information you will provide at the start of the questionnaire A clear and concise statement of the ethical considerations raised by the project and how you intend to deal with them Please discuss with the CUK REC representative at your institution (normally Dean of Research or equivalent) which guidelines are more appropriate for your research and then tick the box to indicate those you have read (): I have read and am familiar with either the BPS Code of Human Research Ethics (http://www.bps.org.uk/sites/default/files/documents/code_of_human_research_ethics.pdf) or the BERA Ethical Guidelines (http://www.bera.ac.uk/wp-content/uploads/2014/02/BERA-Ethical-Guidelines2011.pdf) and (if appropriate) I have discussed them with the other researchers involved in the project. If you are undertaking on-line questionnaire research, please tick the box to indicate that you have read the BPS Report of the Working Party on Conducting Research on the Internet (http://www.bps.org.uk/system/files/Public%20files/inf206-guidelines-for-internet-mediated-research.pdf) Signed: ………………………………...... Print name: ………………………………… Date: ……………………… Print name: ………………………………… Date: ……………………… Print name: ………………………………… Date: ……………………… (UG or PG researcher, if applicable) Signed: ………………………………...... (Lead Researcher or Supervisor, if applicable) Signed: ………………………………...... (CUK REC representative at your institution) E-mail address of CUK REC representative at your institution: ………………………………… Summary Your application should consist of: 1. Your signed Ethics Approval Form (no more than the equivalent of two sides of A4) with attachments if appropriate (e.g. questionnaire(s) to be used in the research) 2. Introductory paragraph (no more than one side). An electronic copy of the submission, formatted into one file, should be sent to Dr Liliana Araujo: liliana.araujo@rcm.ac.uk, with the subject Application to CUK REC Deadlines in 2015-2016 are as follows: Friday 23 October 2015 Friday 4 December 2015 Friday 5 February 2016 Friday 18 March 2016 Friday 20 May 2016 Friday 1 July 2016 Friday 26 August 2016 There should be no need for “extraordinary” applications to be made at other times, but they will be considered exceptionally, with good reason. Please turn to next page for notes on completing the form and guidance on providing information for respondents. Notes on completing the form PLEASE DELETE THESE NOTES PAGES ONCE YOU HAVE COMPLETED YOUR FORM. DO NOT INCLUDE THEM WITH YOUR APPLICATION. NOTE: THIS FORM SHOULD BE COMPLETED ELECTRONICALLY AND MUST NOT GO OVER TWO SIDES. Research involving participants is hence defined broadly to include research that: i. ii. iii. Directly involves people in the research activities, through their physical participation. Physical participation may signify invasive (e.g. surgery) and/or non-invasive research (e.g. interviews, questionnaires, surveys, observational research) and may mean the active or passive involvement of a person; Indirectly involves people in the research activities, through their provision of or access to personal data and/or tissue; Involves people on behalf of others (e.g. parents/legal guardians of children and the psychologically and/or physically impaired, and supervisors of people under controlled environments [e.g. prisoners, pupils]). Participants in questionnaire research are more commonly known as respondents. Some people participating in research may be more particularly vulnerable to harm than others. Particularly vulnerable groups include: i. ii. iii. iv. v. Infants and children under the age of eighteen; People with physiological and/or psychological impairments and/or learning difficulties; People dependent on the protection or under the control or influence of others (e.g. children, pupils, people in care, young offenders, prisoners, employees/fellow staff, students); Relatives of sick people (e.g. parents of sick children); People who may have only a basic or elementary knowledge of the English language. Sensitive personal data consist of information as to: i. ii. iii. iv. v. vi. vii. viii. The racial or ethnic origin of the participant, His or her political opinions, His or her religious beliefs or other beliefs of a similar nature, Whether he or she is a member of a trade union (within the meaning of the Trade Union and Labour Relations (Consolidation) Act 1992), His or her physical or mental health or condition, His or her sexual life, The commission or alleged commission by him or her of any offence, or Any proceedings for any offence committed or alleged to have been committed by him or her, the disposal of such proceedings or the sentence of any court in such proceedings. After participating in research, participants must be debriefed in a positive manner regardless of how successfully you think they have completed the task. Questionnaire respondents must be thanked. APPLICANTS MUST READ THE CODE OF HUMAN RESEARCH ETHICS PUBLISHED BY THE BRITISH PSYCHOLOGICAL SOCIETY OR THE ETHICAL GUIDELINES FOR EDUCATIONAL RESEARCH PUBLISHED BY THE BRITISH EDUCATIONAL RESEARCH ASSOCIATION AND THE BPS GUIDEILNES FOR INTERNET-MEDIATED RESEASRCH IF APPROPRIATE BEFORE SIGNING THE FORM. These documents can be found at: http://www.bps.org.uk/sites/default/files/documents/code_of_human_research_ethics.pdf http://www.bera.ac.uk/wp-content/uploads/2014/02/BERA-Ethical-Guidelines-2011.pdf http://www.bps.org.uk/system/files/Public%20files/inf206-guidelines-for-internet-mediated-research.pdf Guidance on providing information for respondents The introductory paragraph at the beginning of a questionnaire should be designed to assist respondents to make an informed choice as whether or not they want to complete the questionnaire, and should they take part, it should be made clear that they are free to stop at any time. Researchers should take all steps necessary to ensure that respondents understand the process in which they are to be engaged, including why their responses are necessary, how they will be used and how and to whom they will be reported. Research project title Is the title self-explanatory? If not, a simplified title should be included. Introductory paragraph The researchers should be named, the aim of the research should be provided and the respondents who are being recruited should be described. Respondents should be invited to contact the researcher if they have any questions. Informed consent You should explain that you will infer from respondents’ submission of the completed questionnaire, either electronically or by post, that they have given their consent to take part in the research. Confidentiality and anonymity You should explain that respondents’ contact details will be kept separate from the data they provide for the purposes of the research. Thus all information that is collected about them during the course of the research will be kept strictly confidential. Any information about them that is disseminated will have their name and address removed so that they cannot be identified by it. Here is an example of an introductory paragraph from a real on-line questionnaire, including references to informed consent, confidentiality and anonymity: Thank you for considering taking part in our research. We are interested in the strategies student performers use when learning a new piece for a performance. Consent Your participation in this research is voluntary, and you may withdraw from the study at any time if you wish. By submitting a completed questionnaire, however, you are giving your informed consent to participate in the study. You do not have to answer any question that you do not wish to answer. What will we do with your data? The data you provide will be anonymous (separated from your name) and confidential (not disclosed to anyone else). We may publish reports based on our findings, but you will not be identifiable from the data included. Who should I contact if I have queries about the research? If you have any queries or concerns about the research, you can contact Jane Ginsborg (jane.ginsborg@rncm.ac.uk) or Helen Prior (helen.prior@kcl.ac.uk). The project has been reviewed and approved by the Research Ethics Committees of the RNCM and King’s College London. Dissemination At the end of the questionnaire, you should explain how and to whom the results of the research will be disseminated. Thanks Finally, you should thank respondents for completing and submitting the questionnaire.