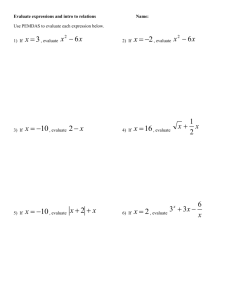AS Chemistry topic checklist
advertisement

Weak acids Strong acids, Kw and strong bases Acids and bases Know the Bronsted-Lowry definition of an acid and a base and write equations to show the dissociation of each in water Write full and ionic equations for the reactions of acids with metals, carbonates, bases and alkalis Explain the difference between strong and weak in terms of dissociation, and represent this with equations Identify acidic and basic conjugate pairs Know that pH is a measure of the H+ concentration in solution Calculate pH using pH = -log10[H+(aq)] for strong monoprotic and diprotic acids CARE: the ‘-‘ means negative, not subtract Calculate concentration using [H+(aq)] = inverse log –pH for strong monoprotic and diprotic acids Write equations for the partial dissociation of water Write an expression for the equilibrium constant of water, giving units Write an expression for the ionic product of water, Kw, stating units, and know its value at 298K CARE: this changes with temperature Calculate the pH of a strong base by rearranging the expression for Kw to find the [H+(aq)] then using pH = -log10[H+(aq)] Calculate the pH change of a solution when acid and alkali are added to one another CARE: the volume of solution will change Write equations for the partial dissociation of weak acids Write an expression for the acid dissociation constant, Ka, giving units Rearrange Ka to find any of the values in the expression Know that in a weak acid, [H+(aq)] [A-(aq)] are equal i.e [H+(aq)]2 Calculate the pH of a weak acid using Ka to find the [H+(aq)] CARE: remember to √ - see above, then using pH = -log10[H+(aq)] Calculate the concentration of a weak acid by using [H+(aq)] = inverse log –pH and substituting this into the expression for Ka Know why pKa values may be given instead of Ka values Convert Ka to pKa using pKa = -log10Ka Convert pKa to Ka using Ka = inverse log –pKa CARE: always use Ka in pH calculations Exam ques Revised ): S: Covered ACIDS, BASES AND pH (: A2 Chemistry – Unit 4 pH curves Buffers Be able to sketch pH curves for any combination of strong/weak acid with strong/weak base CARE: read carefully which is being added so the curve is drawn the correct way round Understand what is meant by the equivalence point Use the equivalence range to select a suitable indicator Know the pH ranges in which methyl orange and phenolphthalein operate, and the associate colour changes Use a pH curve to carry out titration calculations Interpret a pH curve with two equivalence points as a result of a diprotic acid Define what a buffer is Describe how to make an acid and alkali buffer Write equations to show the formation of an acid and alkali buffer, explaining relative concentrations of species referring to partial or full dissociation. Explain how buffers resists changes in pH when small amounts of acid or alkali are added, referring to the position of equilibrium Write equations for the buffer action described above Calculate the pH of buffers using the expression for Ka to find [H+(aq)] then using pH = -log10[H+(aq)] Calculate the pH of buffers after the addition of acid or alkali (CARE: recalculate moles, volume and molarity) Discuss some uses of buffers e.g. shampoo, washing powder, controlling blood pH Optional: Write equations for the buffer which keeps our blood pH constant and explain the importance of this