Organic Chemistry Study Guide
advertisement

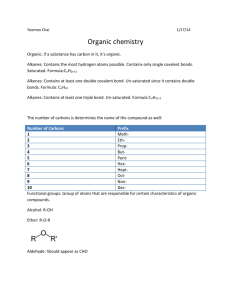
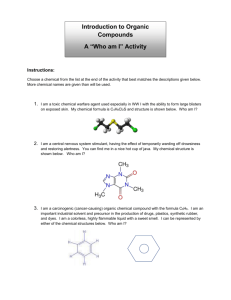
Study Guide Organic Chemistry Please start by reading the summary…page 5 Introduction and Classification of Organic Compounds, and page 4 Aliphatic Hydrocarbons There are 2 main branches of Organic Chemistry- which is the study of hydrocarbonscompounds composed primarily of carbon and hydrogen. Aliphatic, or “straight chain” compounds which exist as 2 types Saturated- all single bonds between carbon atoms and the maximum number of hydrogen present, and have a general formula of CnH2n+2, where n is the number of carbon and 2n+2 the number of hydrogen. This class of compounds is known as alkanes, and will end their name with the suffix –ane. Unsaturated- one or more double or triple bonds between carbon atoms and less than the maximum number of hydrogen atoms present Double bonded hydrocarbons are known as alkenes and will end their name with the suffix – ene. A hydrocarbon with only one double bond and all other single bonds will have a general formula of CnH2n While triple bonded hydrocarbons are called alkynes and end their name with the suffix –yne. A hydrocarbon with only 1 triple bond and all other single bonds will have a general formula of C2H2n-2 Alicyclic, or hydrocarbons with “ring structures” which also exist as 2 main types. Cyclic Hydrocarbons- ring structures with only single bonds between the carbon atoms and have a general formula of CnH2n This class of hydrocarbon will have a prefix of cyclomeaning ring. Aromatic hydrocarbons- a ring structure that possess at least 1 double bond between the carbon atoms in the ring. And typically have very low boiling points. The Naming of organic compounds is very ordered…We have already discussed the suffixes, -ane, -ene, -yne and now you need to know about the roots. The root name for an organic compound comes form the number of carbon atoms present. It is simply a method of counting…Look at the chart on the following page. Therefore a saturated hydrocarbon CnH2n+2 where n=4 would be….C4H10 and its root would be but (that is with a long U, like the city Butte, Montana) so we have a root, and from before, we have a suffix (-ane). Put them all together…butane. Yes, the stuff inside a lighter. If we follow this pattern, a hydrocarbon with one double bond and n=3 would be C3H6 and have a name of propene. And so on and so forth…And of course a hydrocarbon with a triple bond and n=12 would be dodecyne, C12 H10 If the double or triple bond does not occur on the first carbon, care must be taken to describe the location of the bond. We do this by using a number in front of the name. See Aliphatic Hydrocarbons page3 Sometimes a hydrocarbon will be “substituted” - that is to say that something other than hydrogen is stuck on it. It can be a carbon chain or a functional group. See Introduction and Classification of Organic Compounds pages 2-3 Now go back and reread the first 2 sections!!! At this point you should be able to complete the practice activity on page 5 of Introduction and Classification of Organic Compound as well as the homework#1 Next read the summary for Cyclic Hydrocarbons on page 7 Now don’t forget those silly ring structures… they are the ones with the prefix cyclo- and general formula CnH2n and yes, I know that is the same general formula for the – enes Oh well… If you have an alicyclic structure where n=5, you need to use the prefix, cycloand its root would be pent and the suffix is ane, (remember alicyclic are single bonded.) Put them all together, and you get…cyclopentane. See page 2 Cyclic Hydrocarbons Simple aromatics, which are monocyclic- one ring; are named using these same rules. See pages 3 and 4 Cyclic Hydrocarbons Many aromatic compounds have more than 1 double bond, or are polycyclichave more than 1 ring, and therefore names become extremely complicated. This is one reason they have their own names, and as you use them you simply become familiar with them. See pages 5 and 6 Cyclic Hydrocarbons Just for fun, why don’t you try Google and look up caffeine. When you see the structure, you will be glad we call it caffeine. Now go back and reread section 3!!! At this point you should be able to complete the practice activity on page 7 of Cyclic Hydrocarbons. Then, read the summary for Organic Surfactants, Emulsifiers, Solvents, and Polymers page4 Surfactants are chemicals that reduce the surface tension of a liquid. Surfactants make very good detergents and stain removers. You should already know that surface tension is caused by the attractive forces among the liquid molecules. Surfactants are usually very large compounds and are occasionally called amphiphilic-one end of the compound is hydrophilic-highly attracted to water or polar molecules while the other end is hydrophobic- (also known as lipohpilic) highly resistant to water or attracted to non-polar molecules. This makes surfactants soluble (dissolve) in both polar and nonpolar compounds. Micelles- surfactants with the same 2 ends that the amphiphilics have, but are specifically tear drop shaped. There are 4 divisions of surfactants which come about primarily due to the strange shapes of the very large molecules. Anionic- mostly negatively charged, cationic-mostly positively charged, non-ionic- relatively no charge, and zwitterionic- or having both positive and negative charges on the molecule. Emulsifiers-are chemicals used to stabilize a liquid that is a mixture of both polar and non-polar substances, and prevent separation of the mixture into 2 layers. Most emulsifications also add texture and quality of thickness to the liquid. See page 2 Organic Surfactants, Emulsifiers, Solvents, and Polymers. A solution is a special kind of mixture that has two parts. There is the solvent- the part that dissolves things, usually a liquid, but always the substance present in greatest amount. And the solute– the part being dissolved, usually a solid, but always the substance in least amount So for example if you put salt in water, the water is the solvent and the salt is the solute. Another significant characteristic of solvents and solutes is the “Like dissolves Like” rule; which means, polar substances dissolve other polar substances (salt and water) and non-polar substances dissolve other non-polar substances (oil and gasoline). With organic solutions there are additional considerations. Some organic solvents are protic- easily release hydrogen ions while others are aprotic- do not release hydrogen ions. This comes about because hydrocarbons have so many hydrogen atoms to begin with. Because of their relatively low boiling points, organic solvents pose a physical threat. They are generally very prone to flash fires- flammable (burns easily) and are volatile- evaporates easily, as well and many are known carcinogens- cancer causing agent. Polymers are very, very, very, very, looooong chains of monomers-single carbon units or a group (not specifically a single atom).This is similar to a paperclip chain where each paperclip would be a monomer and the chain is the polymer. There are 2 main classes of polymers, SyntheticPolymers- which are man made. Like plastics, and nylon. Bakelite- one of the earliest produced polymers, one use was production of costume jewelry and it is considered valuable now to collectors. Thermoplastic- are usually developed from 2 or more organic compounds that are manipulated with high heat. Biopolymers- naturally occurring polymers, such as proteins (your hair happens to be one) Now go back and reread Section 4!!! And complete the Research Activity on page 5 of Organic Surfactants, Emulsifiers, Solvents, and Polymers You should now be able to complete Kevlar Research Activity, Organic Chemistry HW#2, Virtual Lab: Hydrophobic and Hydrophilic Compounds Gizmo, and Lab - Hydrophobic and Hydrophilic Compounds