EFFECTIVENESS, META-ANALYSIS AND QUALITY
advertisement

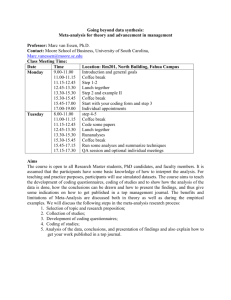
EFFECTIVENESS, META-ANALYSIS AND QUALITY-ADJUSTED LIFE YEARS Seminar work of Pharmacoeconomics Collection of most important questions from the seminar work Prepared by: Ognen Jakasanovski, Faculty of Pharmacy, University of Skopje Rosa López Pazos, Faculty of Pharmacy, University of Santiago de Compostela Luis Marin, Faculty of Pharmacy, University of Grenada Raquel Alonso Rubín, Faculty of Pharmacy, University of Santiago de Compostela Nicolas Jose Mallen, Faculty of Pharmacy, University of San Jorge Mariona Armengol Saborit, Faculty of Pharmacy, University of Barcelona Mentor: Prof. Mitja Kos, PhD Head of Department of Social Pharmacy Faculty of Pharmacy, University of Ljubljana Ljubljana, 25.05.2015 1. What is a randomized clinical trial? Randomized clinical trials are scientific investigations that examine and evaluate the safety and efficacy of new drugs on human volunteers 2. How do we classify clinical trials? Phases, Trial design, number of centers and others. 3. Which errors we can find on the clinical trial? - Bias/ systematic error - Confounding - Random error 4. How is a clinical trial developed? - Objectives and endpoints - Study design - Patient’s population - Sample size calculation - Conduct of the trial - Interim monitoring - Final data analysis -Trial reporting 5. What should be asked of patients before the beginning of a clinical trial? The patients need to sign an informed consent. 6. What is the sample size calculation used for? The sample size calculation is used for minimize random error. 7. How can we analyze outcomes? All endpoints can analyse with the log-rank test, stratified and displayed on Kaplan–Meier plots. 8. Why does the Sampling error occur? The sampling error is produced because the sample might yet show a chance false result compared to the overall population. 9. What is a systematic review? A systematic review is a summary of the best available research studies on a specific question. This is done by identifying, selecting and synthesizing the results of several studies relevant to that question. They have replaced traditional narrative reviews. Nowadays the implementation of them in practice is becoming mandatory for all healthcare professionals. 10. Which are the differences between a traditional and a systematic review? Traditional reviews differ from a systematic review in that they are not led via a peer-reviewed protocol and so it is not often possible to replicate the findings. The traditional way is rarely explicit about how studies are selected, assessed and integrated. The lack of rigor in traditional reviews is another difference. In traditional reviews may be different conclusions, but in a systematic review there is a common conclusion between reviewers (peer-reviewed). 11. In which situations is needed to do a systematic review? Needed to propose a future research plan Required by authors who want to research in primary healthcare Required by postgraduate theses Needed whenever there is a question with different findings 12. Are these sentences true or false? Reviewers must search only for published articles in a systematic review (False) It is necessary to perform a sensitivity test to carry out a good systematic review (True) Reviewers have to exchange their data and then aggregate them (True) 13. What is a meta-analysis? Meta-analysis can be defined as a statistical research technique which combines the findings from previous research. It is a method for systematically combining qualitative and quantitative data from selected independent studies to develop a single conclusion. 14. Which are the advantages of a meta-analysis? Overcoming bias. The danger of a narrative reviews is that they could introduce bias. If a meta-analysis is carried out on a rigorous way, it can overcome these dangers. Precision. These researches have more power to detect clinical significant effects. Transparency. A good meta-analysis should allow readers to determine for themselves the reasonableness of the decisions taken. 15. Which is the difference between a systematic review and a meta-analysis? Meta-analysis is based on systematic review methodology: impartial summary of the existing research. Besides they include a quantitative approach, it is a statistical part. If the systematic review provide a quantitative estimation of benefit is termed a meta-analysis. 16. What is a QALY? The QALY is a measure of a person’s length of life weighted by a valuation of their healthrelated quality of life. It is expressed by this equation: QALY= UxT being the T the life years gained and the U the quality of those years. 17. Which type of cost-outcome analysis is QALY used in? Cost-utility analysis 18. Quality of a person is valued in a range between 0 and 1, what state represents each number? Quality could be valued between 0 and 1. In this scale, the zero value is the death stage and one is the perfect health. In the middle of this scale, there are different statuses of health. 19. What is the null hypothesis in a clinical trial? 20. Which are the requirements to choose an endpoint? 21. Limitations of death or time to death as an endpoint 22. Which are surrogate endpoins? 23. When are very used surrogate endpoints? 24. Which are the measures used in health economic endopoints? 25. When are useful surrogate endpoints? 26. What are health utilities and what kind of data is generated? 27. Describe de importance of using patients for the assessment of levels of health. 28. Explain the Standard Gamble (SG) method. 29. Differences between SG, TTO and VAS 30. What does Rating scale mean? -Is a set of categories designed to give information about quantitative and qualitative. 31. Why Standard gamble is really important? -The method has been used extensively in the field of analysis decisions. 32. What is TTO? -Is a tool used in health economics to help determine the quality of life of a patient or group. 33. How is EQ-5D classify ? -Mobility, Self-care, Usual activities, Pain/Disconfort, Anxiety/depression. 34. How are Health Utilities Index divided? -Divided in two systems: ·HUI2 & HUI3; the scale to measure them are between 0 and 1 scale. In both cases are based on standard gamble utilities measured on the general public, and the scores are on the conventional dead-healthy 0-1 scale. 35. Which details of the meta-analysis are commonly displayed above the graph? -Review: Title/research question of the meta-analysis -Comparison: Intervention VS control group -Outcome: Primary outcome measure and presented in the graph 36. What is a meta-analysis of binary measure? -A research study for Cancer, Infart… This kind of method measure Risk “RR” (Relative Risk) and Odds “OR” (Odds Ratio). To evaluate the effect you need to compare Risk in intervention group and in control group. 37. What is IC? -In called Interval Confident, Quantifies the stimation´s precision. Isn´t the same value a risk of 10% stimated in 10 patients than in 1000 patients. 38. What is Heterogeneity test, and what does it measure? -Is values in I^2 and the range are between 0% and 100%. It measures the variability between studies; how comparable studies in meta-analysis are. 39. What is the QALYs equation? QALY= UxT being the T the life years gained and the U the quality of that years. 40. What is the test for assessing health condition? The EQ-5D test 41. List the two main approaches of post-randomization data analysis of clinical trials. - Per-protocol (PP) analysis Intention-to-treat (ITT) analysis 42. What is the main principle behind the per protocol analysis? The per-protocol analysis excludes subjects who were not fully compliant with study protocol. It is also called efficacy, explanatory analysis, or analysis by treatment administered. By focusing only on the fully compliant subjects, one can determine the maximal efficacy of a treatment using the PP analysis. 43. Explain why PP analysis is often considered to undermine randomization. Excluding non-adherent participants from the analysis leaves those who may be destined to have a better outcome and presents a problem for the desired unbiased comparison between treatment done by randomization. 44. What is intention-to-treat analysis? What is the main rationale for conducting this analysis? An intention-to-treat (ITT) analysis is a specific clinical trial design and conduct strategy for generating the results of an RCT. It is an approach of comparing all trial subjects according to the treatment groups in they were initially randomized, regardless of treatment compliance, or any treatment subsequently received. The rationale for using the ITT method is that, even though not all patients will fully adhere to the therapy, the non-compliant subjects would probably be equally arranged between the different treatment groups. 45. What are the advantages of conducting an intention-to-treat (ITT) analysis? - Preserving the strengths of randomization and minimizing bias; Depicting real-life situations better than the per-protocol (PP) method; Ability to perform interim analysis; Providing practical, pragmatic information about a particular treatment option. 46. What is the main limitation of the intention-to-treat (ITT) analysis? This type of analysis does not determine the maximum potential efficiency of a treatment as a PP method would. Therefore, in some studies, the ITT method might not show a statistically significant benefit, or might show the benefit to be smaller than that generated by a PP analysis. 47. What are the recommendations for reporting clinical trials with intention-to-treat (ITT) analyses? • Specify that ITT analysis has been carried out, explicitly describing the handling of deviations from randomized allocation and missing response. • Report deviations from randomized allocation and missing response. • Discuss the potential effect of missing response. • Base conclusions on the results of ITT analysis. 48. What is a cost-utility analysis? Cost-utility analysis (CUA) is a form of evaluation that focuses particular attention on the quality of the health outcome produced or forgone by health programmes or treatments. It has many similarities to cost-effectiveness analysis (CEA), because they have similar underlying principles of conducting. 49. What is the main difference between the cost-effectiveness and cost-utility analyses? In the CEA analysis, the incremental cost of a programme from a particular viewpoint is compared to the incremental health effects of the programme, with the health effects being natural units related to the objective of the programme (blood pressure improvement in mm Hg, cases found, cases of disease averted, seizures prevented, blood HDL reduced etc.). The results are usually expressed as cost per unit of effect. On the other hand, CUA compares the incremental cost of a programme from a particular viewpoint (same as CEA) with the incremental health improvement attributable to the programme, where the health improvement is measured in quality-adjusted life-years (QALYs) gained, or possibly an alternative, like disability-adjusted life-years (DALYs) gained. The results are expressed as cost per QALYs gained. 50. List the most important advantages of the cost-utility type of analysis. - - It is a more suitable type of analysis when the health-related quality of life is an important outcome. It can be used in cases where the programme tested affects both mortaility and morbidity, and there is a necessity of a common unit of outcome combining both effects (e.g. cancer chemotherapy, which may improve longevity and long-term quality of life, but decreases quality of life during the treatment). It enables comparability and transferability of results, and allows for the programme to be tested and compared with other programmes.