emi412025-sup-0012-ts2
advertisement

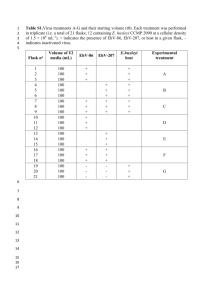
Supplemental Table 2. Primers and PCR conditions used in this study. Application Target Primers Sequence (5’-3’) Anneal temp (oC)/#cycles /extension (s) at 72oC MISA1 Round 1 spacer_pmoC606f GGGGYCAYRCBTTCTGGT (enrichment) spacer_pmoA191r CCARAARTCCCARTCNC Round 2 (bulk spacer_pmoC626f RCBTTCTGGTKBATGGAAGA amplification) spacer_pmoA189r CCARAARTCCCARTCNCC Group C GroupC_spacer1f AGGTAATGAAGCGCATGTCC 52/20/60” length divergent GroupC_spacer2f GCTGTTCAAGCAGCTTAGCC 52/5/60” 48/30/60” CuMMO-encoding GroupC_moBr GCTTTAKCAAASCGTTCAC GroupO_spacer1f ATTTACCCCGGGAGGTATTG 52/20/60” GroupO_spacer2f TTACCCCGGGAGGTATTGAT 52/5/60” 48/30/60” GroupO_moBr GGCCAAAGCGCTAATACACT GroupW_spacer1f GCTTTGTAATCCTCGGATGG 52/20/60” GroupW_spacer2f CAGACAACCAACGCGAGATA 52/5/60” 48/30/60” GroupW_moBr TCGATCGTACCGATCAGTGT GroupZ_spacer1f CACCATTACATTGGGGCTTT 52/20/60” GroupZ_spacer2f TCCATATTCACGCCATGTTAAC 52/5/60” 48/30/60” GroupZ_moAr AACGCAATACAACCCACCAT Degenerate moB156r KHYYKYTCRCCATGYGC CuMMOB moB172r CGCATRCGYATRAANGGYTC reverse primers moB175r TRCGCATRCGYAGRAANGG moB180r TACCABTGAAYSGTRCGCATMC moB185r TCRTACCABTGAAYSGTRCGCAT Recovery of full- gene 52/20/30” 52/5/30” 48/ 20/30” sequences22 Group O Group W Group Z Recovery of 16S gene Round 1 T1methph29F1 TTGAACGCTGGCGGYATGCT candidates, Groups (enrichment) T1methph1292R1 AGGACCGGCTTTBTGGGATT Round 2 (bulk T1methph44F2 ATGCTTAACACATGCAAGT amplification T1methph1279R2A TGGGATTTGCTGACTTTCGC from 5% of T1methph1279R2B TGGGACTTGCTCCACCTCGC enrichment) T1methph1279R2C TGGGATTAGCTTACTCTCGC T1methph1279R2D TGGGATTTGCTGACTCTCGC T1methph1279R2E TGGGATTGGCTCACTTTCGC OPU1 and 52/20/30” OPU33 52/8/60” 48/25/60” Recovery of marine pxm gene sequences4 Amplification pxmA_cons_f TMTTTGCCTAYCACTACTGGAAYT from sediment pxmA_cons_r GTGCCGCGCTCRATGATGCG Amplification pxmA550f CCSASYAAYTGGCCSMTYTT from water pxmC310r AGGWRYYRTCCTGYTCGG Group OPU1 pmoA_OPU1qPCR_242f TTACCCCGATCATGCTGGTT pmoA5 pmoA_OPU1qPCR_312R GATTCTGAAGTGTTCCCAAACGA Taqman probe TTCCCAGCCGCTGTTCAGGCA Group OPU3 pmoA_OPU3qPCR_492f TTGCACCTTTACATYTACCTGTTGA pmoA pmoA_OPU3qPCR_612r ACCTTTTTCTACCATTCYGATRTACTC Taqman probe ACAACGGCATGATGTTTACTGTTGCTGATTTA Group OPU1 16S_OPU1qPCR_f CAATGCCGCGTGTGTGAA 16S rRNA gene 16S_OPU1qPCR_r CCTCTCTTCCTCCCGACTGAA candidate Taqman probe AGGCCTGCGGGTTGTAAAGCA Group OPU3 16S_OPU3qPCR_f AGCACTTTCAATTGGGAGGAAA 16S rRNA gene 16S_OPU3qPCR_r GCCGGTGCTTCTTCTAAAGGT candidate Taqman probe MAGCTGGGTTAATAGCCCYGCTCTTGACA Pelagic pxmA wc_pxmA_qPCR_f CCGATTGAAGCAGGCAAC sequence wc_pxmA_qPCR_r TGCCAGCACGGATATATTGG Taqman probe AGCTAACCTTGGCTGACATGTTCGGCT pmoA_GroupC_qPCR_f CCGCGTTTCAGGCGATATT pmoA_GroupC_qPCR_r CAGGCGGTCGCACCAA Taqman probe TGGTACCTGTTTCGGCTGCC 52/35/30” 52/5/60” 48/ 30/60” column qPCR5,6 Group C pmoA 1 60/40 60/40 60/40 60/40 57/40 57/40 MISA, which targets the non-coding region between pmoC and pmoA among ammonia-oxidizing and methane-oxidizing proteobacteria, was performed as described previously (Tavormina et al., 2010) with a single primer modification to the reverse primer in the enrichment round, to reduce nonspecific products in the final bulk amplicon fraction. The modified primer was validated in parallel with the published assay against ten cultured ammonia-oxidizing and methane-oxidizing cultured strains as previously described, and provided results indistinguishable from the original assay. The primers used in this study have been optimized to the detection of proteobacterial Cu-MMO-encoding organisms, and thus the detection of verrucomicrobial, Gram-positive, or Archaeal Cu-MMO-encoding gene sequences was beyond the scope of this study. For amplification of fluorescent PCR product, pmoC_spacer626f includes a fluorescent tag (Well-Red D1, Sigma Proligo). To construct clone libraries, non-fluorescent MISA fragments were cloned into the pSmart-GC-Kan system (Lucigen, Middleton, WI) following the manufacturer’s guidelines. This system suppresses expression of insert DNA, and increases cloning efficiency of the MISA fragment. For each library, more than 48 colonies were selected for screening. Clones containing inserts of the correct size (between 70 and 95% of each library; the remainder of the libraries represented cloning of primer-dimers) were digested with RsaI and separately with HaeIII to assess restriction patterns. Clones demonstrating unique RFLP patterns were sequenced. 2 To recover full-length pmoA sequence from Cu-MMO variants, a well-conserved region in pmoB was targeted for primer design of 6 degenerate reverse primers. For each individual Cu-MMO sequence targeted, genotype-specific forward primers were designed from MISA fragment sequence. A 20-cycle enrichment round was performed using appropriate environmental template DNA, one of the genotype-specific forward primers and each of the degenerate reverse primers in separate reactions. This enrichment round was followed by qPCR amplification with internal genotype-specific primers on each of the enrichment products, to determine the most efficient reverse primers for each target. From this information, nested PCR was performed on the enrichment PCR product, to recover and sequence a full-length Cu-MMO-A gene fragment. To validate the recovered full-length sequence, specific reverse primers were designed from the recovered sequence, and paired with specific forward primers, to amplify the Cu-MMO target from environmental DNA in a single 35-cycle round of PCR, and the resulting amplicon was sequenced directly. 3 To identify candidate 16S rRNA gene sequences from groups OPU1 and OPU3, representative methane-oxidizing 16S rRNA gene sequences (accession numbers: DQ852349, AF150804, NR_043450, NR_025039, NR_043562, X72771, L20840, NR_025132, U77533) were aligned, and two conserved regions at positions 29-62 and 1259-1292 (M. capsulatus strain Bath numbering) were targeted for primer design. Reverse primers for the bulk amplification of 16S rRNA gene targets were combined into an equimolar cocktail. This strategy targets 16S rRNA gene sequences from gammaproteobacterial methanotrophs, but may also amplify non-methanotrophic 16 rRNA gene sequences from organisms including Kangiella and Beggiatoa. Amplicons were generated from environmental DNA from above Mound 12 (AT15-44 CTD1, 700 meters below sea level, [mbsl]) as well as from a previouslycharacterized bathypelagic DNA sample from the Santa Monica Basin that harbors abundant OPU1 and OPU3 microorganisms (SMB mound above clambed, (Tavormina et al., 2010). Amplicons were cloned and analyzed as described previously (Tavormina et al., 2008). 4 To recover pxm genes from the marine environment, water column (AT15-59 CTD11, Jaco Scarp 1714 mbsl) and sediment (Md. Quepos, 0-3 cm) samples were targeted with consensus primers pxmA_cons_f and pxmA_cons_r (Table S2). The sediment sample yielded an amplicon whose sequence affiliated (78% DNA identity, 92% protein similarity) with terrestrial soil and freshwater pxmA sequences. Alignment of this fragment with existing pxmA sequences allowed the design of a new degenerate consensus primer, pxmA550f, which when paired with degenerate consensus primer pxmC310r, resulted in the amplification of a ~2-kb pxmABC-encoding fragment from marine water column DNA (AT15-59, cast CTD11, 1714 mbsl). 5 Quantitative PCR assays (Taqman qPCR) for the OPU1 and OPU3 pmoA clades, OPU1 and OPU3 candidate 16S rRNA gene sequences, marine water column pxmA sequence, and for Group C pmoA, were developed using Primer Express v2.0 (Applied Biosystems, Foster City, CA, USA), based on all available related sequences (>96% identity), including sequences recovered in this study. The assays in this study may not reliably enumerate these clades in environments where significant divergent gene evolution has occurred. Quantitative PCR was performed on a 7300 real-time PCR system (Applied Biosystems) using a two-step PCR (94oC, 15 s and 57oC or 60oC, 60 s) for 40 cycles. Standard curve analysis on linearized control templates over 5 orders of magnitude indicated an amplification efficiency >95% for all assays. Signal from nontarget CuMMO-encoding sequences was below the level of detection, indicating that each assay is specific for its designated target. For the 16 water samples collected in AT15-59 CTD11, qPCR was conducted in triplicate. Deviation in estimated abundance between these triplicate measurements was typically between 10 and 30% for individual samples. For all other casts and dives (~100 environmental samples), single qPCR assays were conducted for all targets. qPCR amplicons for pmoA gene targets of groups OPU1, OPU3, and Group C, and 16S rRNA gene targets, were sequenced bi-directionally to validate the identity of amplified sequences. 6 Sequencing throughout this study was performed using a commercial service (Big Dye chemistry, Laragen, Culver City, California) and all sequences have been deposited in Genbank (Accession numbers JX569083-JX569145).