Aleesa Muir Research Plan 1 November 2012
advertisement

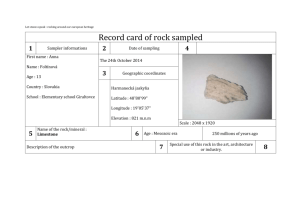
Muir 1 Aleesa Muir Research Plan 1 November 2012 Question Are different types of rocks more susceptible to weathering from acid rain (3.8 – 4.3 pH) as compared to “normal” rain (5.1 – 5.6 pH)? Hypothesis If comparing the susceptibility of weathering with different types of rocks to acid rain, then the weathering of the rock will increase for all of the rocks. Research indicates that acid rain, compared to “normal” rain, accelerates the chemical weathering process; however, minerals in the rock and the formation of the rock influence how susceptible the rock is to deteriorating. Metamorphic rocks will be the least susceptible to weathering because they are formed by heat and pressure, therefore making them more compact. Sedimentary and igneous rocks will vary in susceptibility to weathering based on the formation of the rock. The cementation between the layers in a sedimentary rock will determine the compaction of the rock and how the rock reacts with acid. The rate at which an igneous rock cools will determine if the rock will be more porous or compact affecting its rate of weathering. Variables Independent: Type of rocks Dependent: Weathering/deterioration of rocks Control: Rock with “normal” rain (5.1 – 5.6 pH) Constants Amount of raining time; volume of rock; method of measuring weathering of rock; amount of acid rain; temperature of environment; method of pouring acid rain; equipment set-up; distance the rain is above the rock; speed of the acid rain; range of acidity level of rain (acidic 3.8 – 4.3 pH and normal 5.1 – 5.6 pH). Materials List - Equipment 1 – Lattice plastic (240.0cm×120.7cm×3.2 mm) 2 – Tub (75.7 L) 2 – 8-Port Manifold (adjustable flow) 12 – Spray Heads (adjustable flow) 12 – Drip Line (15.2×0.6id cm) 2 – Fountain Pump (378.5 – 586.7 Lph) 2 – PVC Pipe (12.7×2.5 id cm) 2 – PVC Pipe (10.2-14.6 cm) 4 – PVC Pipe threaded (7.6×1.3 cm) 2 – PVC Pipe threaded (15.2×1.3id cm) 4 – PVC Pipe threaded/barbed (1.3-2.5 cm) 2 – T-Joint PVC threaded/barbed (1.3 cm) 2 – PVC Plug barbed (5.1×1.3id cm) 2 – PVC Elbow threaded/barbed (2.5-1.3 cm) 4 – PVC Elbow threaded/barbed (1.9-1.3 cm) 1 – Wood Dowel (91.4×0.64cm) 22 – Zip Strips 4 – Vinyl Tubing (5.1×1.3id cm) 2 – Vinyl Tubing (10.2×1.3id cm) 2 – Vinyl Tubing (2.5×1.3id cm) 2 – Vinyl Tubing (5.1×1.9id cm) Muir 2 Materials List - Experiment 1 – Electronic pH tester (Vernier LabPro) 1 – Pipette 190.0 L – Distilled Water 8 – Colorado Moss Rock (100 cmᶟ each) 8 – Siloam Stone Rock (100 cmᶟ each) 8 – Mountain Granite Rock (100 cmᶟ each) 8 – Table Mountain Rock (100 cmᶟ each) 8 – Lava Moss Rock (100 cmᶟ each) 8 – Pioneer Granite Rock (100 cmᶟ each) 1 – Spoon (plastic) 1.0 L – Sulfuric Acid (0.18 M 1.0 L) 0.5 L – Sulfuric Acid (0.002 M 0.5 L) 1 – Calculator (TI-84 Plus) 1 – Glass container with lid (1.0 L) 1 – Glass container with lid (0.5 L) 2 – Chemical Aprons 2 – Chemical goggles 2 – Chemical gloves (resistant) 1 – Measuring cup (2,000 mL) 1 – Electronic balance (±0.1 gram) 1 – Glass cup (small) 1 – Towel (bath) 1 – Aluminum baking pan (28×22×6cm) 1 – Water dispenser (500 mL) Procedure - Equipment 1. Cut tubes. 2 – Vinyl tubing (1.3id cm) [5.1cm long] 1 – Vinyl tubing (1.3id cm) [10.2cm long] 1 – Vinyl tubing (1.3id cm) [2.5cm long] 1 – Vinyl tubing (1.9id cm) [5.1cm long] 6 – Drip line tubing (0.6id cm) [15.2cm long] 2. Connect tubes and pipes to form tube system. 3. Adjust tubes and pipes to desired positions. 4. Connect pump to tube system. Zip strip tubes to pipes for tight and secure fit. 5. Connect spray heads to 15.2cm×0.6cm drip line tubes. 6. Connect 6 drip line tubes to 8-port manifold. Place caps over 2 unused drip line ports. 7. Place tube system and pump into tub centering the vertical PVC pipe (15.2×1.3id cm) in middle of tub. 8. Measure the top of the vertical PVC pipe to the inside wall of the tub in order to get the lattice radius. 9. Draw circumference on lattice using the radius measurements. Use a pencil, marker, and a string to draw a circle. Make sure there is a hole in the center for vertical pipe to fit through. 10. Cut lattice circle. 11. Cut the 2.5cm PVC pipe 12.7cm long and slide the PVC pipe over the 15.2×1.3id cm vertical PVC pipe (lattice will rest on this PVC pipe). Muir 3 12. Place lattice flat over the tube system (in the tub). Align the end of the vertical pipe with the center hole of the lattice. Place the pump cord threw a slot on the side of the lattice and out of the tub. 13. Attach 8-port manifold (on top side over lattice) onto vertical pipe (threaded), securing the lattice in between PVC pipe (12.7×2.5cm) and 8-port manifold. 14. Cut the 10.2cm PVC pipe (15.2 cm long) for drip lines to go over the rocks. Drill and cut 6 slots 1.3cm down from one end of the PVC (spacing the holes equal distance apart around the PVC). 15. Place the 15.2×10.2cm PVC pipe over the manifold so it is resting on the lattice. 16. Attach the 15.2×10.2cm PVC pipe to lattice using zip strips. 17. Position the 6 drip line tubes in the 1.3cm slots on the PVC pipe (1 drip line per slot). 18. Pour water in the tub so the pump is fully submerged. 19. Plug pump into outlet to make water flow and drip line adjustments. 20. Repeat steps 1 – 19 to assemble a second rain system. Procedure - Experiment 1. Ensure all rocks have an approximate volume of 100 cmᶟ using Archimedes principle. 2. Identify the current pH level of the distilled water. Fill each of the two tubs with approximately 22.7 L of distilled water. 3. Create a rainwater solution using sulfuric acid and distilled water. Normal (control) rain: 5.1 – 5.6 pH Acid rain: 3.8 – 4.3 pH Record the pH levels of each type of rain solution in the data table. 4. Set up two rain systems (see diagram 1). 5. Measure the initial mass (g) of each rock type used in trial. Record the mass (g) of each rock in the data table. 6. Verify the rain system is operating as designed. 7. Place each rock type on a predetermined station number identified within the rain system. 8. Initiate the rain systems by plugging in each pump. Make adjustments (rock or water) to ensure each rock is receiving optimal water coverage. Allow the rain system to run for seven (7) consecutive days. Verify three (3) or more times per day to insure that the rain system is still operating correctly. 9. Record the pH of rain in data table one time each day for the 7 days. Add sulfuric acid to each tub to achieve the correct range of pH levels. Normal (control) rain: 5.1 – 5.6 pH Acid rain: 3.8 – 4.3 pH 10. Rinse each rock with distilled water after the conclusion of the 7 days (168 hours.) 11. Allow the rocks to dry for 3 days (72 hours.) 12. Identify the ending mass (g) of each rock. Record the mass (g) of each rock in data table. 13. Repeat steps 2 - 12 three more times (for a total of 4 trials.) Based on the previous trial rock location, move each type of rock counter clock wise to a different station number after each trial. Muir 4 Operational Definition Measure the mass (g) of the rocks before the experiment; place rocks in respective positions; begin acid rain (process) for 7 consecutive days; stop acid rain; remove rocks; rinse rocks with distilled water; allow rocks to dry for 3 consecutive days; measure the mass (g) of the rock after 3 consecutive days of drying; find the percent lost of mass (g). Form 3 – Hazardous Chemicals, Activities, and Devices Safety concerns: o Sulfuric Acid o Electrical Equipment Risks: o Sulfuric Acid: Highly toxic; skin and eye damage; heat generated o Electrical Equipment: Electrocution Precautions: Add acid to water; avoid contact with eyes, skin, and clothing; wear goggles, glove, and apron; always have adult supervision; wash hands prior to the experiment and after; electrical equipment is in good condition; wipe up spills immediately; neutralize with baking soda if needed Disposal: When finished with the acid, wash down the sink with large quantities of water. Safety Information: "Sulfuric Acid." Flinn Scientific Inc. 2002. Web. 18 November 2010 Works Cited “Acid Rain.” U*X*L Complete Life Science Resource. 2009. Gale Science in Context. U*X*L. Web. 14 September 2010. Cloos, Mark. “Rock.” World Book Online. World Book Encyclopedia. 2010. Web. 12 July 2010. Crawford, Maria Luisa. “Metamorphic Rock.” World Book Online. World Book Encyclopedia. 2010. Web. 12 July 2010. Crawford, Maria Luisa. “Sedimentary Rock.” World Book Online. World Book Encyclopedia. 2010. Web. 12 July 2010. Hames, Willis. “Igneous Rock.” World Book Online. 2010. World Book Encyclopedia. Web. 12 July 2010. Hames, Willis. “Mineral.” World Book Online. World Book Encyclopedia. 2010. Web. 12 July 2010. “How Rocks Change.” Annenberg Media. 2010. Web. 14 September 2010. “Human Influences on Weather and Climate.” U*X*L Encyclopedia of Weather and Natural Disasters. 2008. Gale Science in Context. U*X*L. Web. 14 September 2010. “Minerals.” Earth Sciences. 2008. Gale Science in Context. Gale. Web. 14 September 2010. “Minerals.” World of Biology. 2006. Gale Science in Context. Gale. Web. 14 September 2010. Oxlade, Chris. Acids & Bases. Chicago: Heinemann Library. 2002. Print. “Picture: The water cycle.” World Book Online. World Book Encyclopedia. 2010. October 13 2010. “Rocks and Rock Cycle.” World of Earth Science. 2010. Gale Science in Context. Gale. Web. 14 September 2010. “Rocks.” Earth Sciences. 2008. Gale Science in Context. Gale. Web. 14 September 2010. “Rocks.” U*X*L Encyclopedia of Science. 2007. Gale Science in Context. U*X*L. Web. 19 September 2010. “The Rock Cycle Diagram.” Annenberg Media. 2010. Web. 14 September 2010. “Types of Rocks.” Annenberg Media. 2010. Web. 14 September 2010. “The Water Cycle, a Quick Summary.” U.S. Geological Survey. 2010. 13 October 2010. “Weathering.” Environmental Encyclopedia. 2009. Gale Science in Context. Gale. Web. 14 September 2010. “Weathering.” The Gale Encyclopedia of Science. 2008. Gale Science in Context. Gale. Web. 14 September 2010.