appendix
advertisement

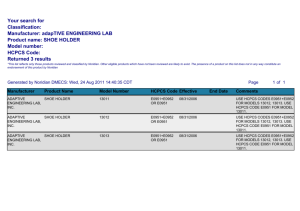
Online Appendix for the following JACC article TITLE: Human Cardiac Progenitor Cells Engineered With Pim-I Kinase Enhance Myocardial Repair AUTHORS: Sadia Mohsin, PHD, Mohsin Khan, PHD, Haruhiro Toko, MD, PHD, Brandi Bailey, PHD, Christopher T. Cottage, MS, Kathleen Wallach, BS, Divya Nag, Andrew Lee, BS, Sailay Siddiqi, MD, Feng Lan, PHD, Kimberlee M. Fischer, PHD, Natalie Gude, PHD, Pearl Quijada, MS, Daniele Avitabile, PhD, Silvia Truffa, BS, Brett Collins, BS, Walter Dembitsky, MD, Joseph C. Wu, MD, PHD, Mark A. Sussman, PHD APPENDIX Table S1 Primary and Secondary Antibodies Application Antibody Dilution Amplify Company ICC GFP 1:200 No Molecular Probes ICC MEF2C 1:200 No Aviva Systems Biology ICC GATA6 1:200 No Santa Cruz ICC VWF 1:200 No Invitrogen IHC α-sarcomeric actin 1:100 No Sigma IHC GFP 1:300 Yes Rockland IHC vWF 1:100 No Invitrogen IHC SM22 1:100 No ABCAM IHC c-kit 1:50 Yes ABCAM FACS c-kit 1:100 No Biolegend FACS CD45 1:100 No Biolegend FACS CD31 1:100 No Biolegend FACS CD34 1:100 No Biolegend FACS Glycophorin 1:100 No Biolegend FACS GFP 1:200 No Rockand Western blot GFP 1:500 No Invitrogen Western blot Pim-1 (C20,E16) 1:500 No Santa Cruz Western blot B tubulin 1:1000 No Santa Cruz Western blot P21 1:100 No ABCAM Western blot Phospho p21 1:100 No Santa Cruz Table S2 Primers for quantitative RT-PCR Primer Name Forward/Reverse Sequence vWF Forward GGATTTGCATGGATGAGGATGGGA vWF Reverse TTGACCCGATGACTCTTCAGCAAG Mef2c Forward AGATCTCCTGTTGACAGCTTGA Mef2c Reverse GTGGAATTCGTTCCGGTGAT GATA6 Forward CCAGGAAACGAAAACCTAAGAACA GATA6 Reverse TTGGAGTCATGGGAATGGAATT Table S3 P values for EF and FS for echocardiography 20 weeks after transplantation Table S4 P values for MRI and Echocardiography data 8 weeks after transplantation SUPPLEMENTAL METHODS Generation of Lentiviral vectors and transduction of hCPCs hCPCe and hCPCeP are transduced with lentivirus for eGFP and Pim-1 and eGFP. Lentivirus were made as described earlier (1) and lentiviral constructs are described in online supplementary fig 1C. Lentivirus for Fluc-RFP(-HSVttk) (Fig 6 A) is collected from the supernatant of transfected 293FT cells and concentrated as previously described (1). hCPCe –Luc and hCPCeP-Luc were transduced at a MOI of 10. Human Cardiac Stem Cell Isolation hCPCS are isolated from heart tissue obtained from patients undergoing surgery for Left Ventricular Assist Device (LVAD) implantation. The study protocol is IRB exempt by NIH guidelines for human subjects research exemptions (45 CFR 46.101) defined as: "Research involving the collection or study of existing data, documents, records, pathological specimens, or diagnostic specimens, if these sources are publicly available or if the information is recorded by the investigator in such a manner that the subjects cannot be identified, directly, or though identifiers linked to the subjects." The only information collected with the tissue sample is the age and sex of the patient. The human cardiac progenitor cells (hCPCs) in our study are obtained from a male patient at 68 years of age. Normally, tissue samples are procured from the left ventricular wall core plug removed to insert the LVAD pump that would otherwise be discarded. Tissue is minced in small pieces and digested in collagenase at 150U mg/ml (Worthington Bio Corp, Lakewood, NJ) for 2 hours at 37°C. The cells are then passed through 100µm and 50µm filters (BD biosciences CA), centrifuged at 1200rpm for 5 min, with resuspension of the pellet in hCPCs media consisting of Hams F12 (Fisher), 10% FBS, 1% PSG, 5mU/ml human erythropoietin (Sigma), 10ng/ml basic FGF (Peprotech). Following overnight incubation at 37°C the cells are collected and passed through 100µm and 50µm filters and centrifuged at 1200rpm for 5 min. The pellet is incubated with c-kit labeled beads (miltenyi biotec, CA) and sorting is performed according to manufacturer’s protocol. The cells are subsequently seeded and expanded clonally. hCPCs were successfully isolated from 29 LVAD samples out of 41 total samples for a success rate of 70%. Transduction and differentiation of hCPC Six hCPCs isolates from distinct patient samples are infected with bicistronic lentivirus expressing eGFP alone (Lv-eGFP) or in combination with Pim-1 (Lv-eGFP+Pim-1; Supplement Fig 1C). For lentiviral infection, hCPC are plated in 6-well dish (50,000 cells/ well), serially transduced with lentivirus (multiplicity of infection (MOI) of 20 followed by MOI of 10 two days after first infection) to obtain hCPC expressing enhanced green fluorescent protein (eGFP) together with Pim-1 (hCPCeP) or eGFP alone (hCPCe). Engineered hCPCs are expanded and analyzed using flow cytometry to validate high percentage eGFP expression. All six cell lines successfully transduced to over express Pim-1 and GFP and the cell line having the highest Pim-1 expression was chosen for use in this study. Additional studies incorporate modification of hCPCs with a firefly luciferase (Luc) reporter construct to generate hCPCe Luc or hCPCeP Luc for bioluminescence Imaging (BLI) as described in online supplementary methods. For differentiation hCPCs are treated with dexamethasone 10-8 mol/L for 7 days as previously described (1) Karyotype Analysis Karyotype analyses of hCPCs are performed at the Cytogenetics Laboratory, Department of Pathology, Stanford Hospital and Clinics. Cells are treated with 0.1 mg/mL colcemid for induction of mitotic arrest with subsequent harvest by trypsin dispersal, hypotonic shock, and fixation with 3:1 methanol: acetic acid. For each cell line, 20 metaphase spreads are analyzed using standard G-banding method. MTT, CyQuant, Trypan Blue Exclusion and Trap Assay MTT and CyQuant assay of hCPCs, hCPCe and hCPCeP involves plating 2000 cells/ well in quadruplicate in a 96-well plate and incubation with MTT reagent (Sigma, St Louis, Mo) or CyQuant reagent (Invitrogen) as described by our group (1). Pharmacologic inhibition of Pim-1 kinase by quercetagetin (10µmol/L; Calbiochem, San Diego, Ca) is performed as previously reported (1). Telomerase activity is assessed by TRAP assay using quantitative PCR according to the manufacturer’s protocol (TRAPeze RT; Chemicon S7710). Real time RT- PCR Total RNA is isolated from hCPCe and hCPCeP before and after dexamethasone treatment by using Quick-RNA MiniPrep (Zymo Research). cDNA is prepared using iScript cDNA Synthesis Kit (Bio Rad). Real time PCR is performed on samples in triplicate using iQ SYBER Green (Bio Rad) according to manufacturer’s protocol. The primer sequences used for experiments are listed in Table S2. Protein Isolation and Western blot Analysis hCPCe and hCPCeP are plated in 6-well dishes (50,000 cells/ well). The samples were harvested in 50µl of sample buffer, boiled and sonicated. Protein lysates are run on 412% NuPage Novex Bis Tris gel (Invitrogen), transferred on polyvinylidene fluoride (PVDF) membrane, blocked in 8% skim milk in Tris-buffered saline Tween-20 (TBST) solution for 1 hour at room temperature and then incubated with primary antibodies overnight at 4°C (Table S1). Membrane is incubated with secondary antibodies for 1 hour at room temperature after several washes with TBST solution. Fluorescence signal is detected and quantified using Typhoon 9400 fluorescent scanner together with Image Quant 5.0 software (Amersham Biosciences). Myocardial Infarction, Echocardiography, Hemodynamics Surgery is performed on SCID mice including sham=15, vehicle=20, hCPCe=20 and hCPCeP=20 that are anesthetized by isoflurane inhalation, intubated and ventilated. Thoracotomy reveals the left anterior descending (LAD) artery for ligation. Cells are injected into 5 separate sites (100,000 cells) at the border zone area after infarction. Infarction size is standardized by echocardiography using Vevo 770 imaging system (Visualsonics, ON, Canada) using a parasternal short axis view by M mode recorded 7 days after surgery. Lack of anterior wall motion with at least 40% decrease in ejection fraction and fractional shortening is the minimal inclusion criteria for experimental groups. Echocardiographic assessment for ejection fraction at 1 week after infarction is 33.9% by the Stanford group (Wu) versus 15% for the San Diego group (Sussman). These measurements suggest initial infarction injury is not comparable between the two sets of experiments, although within each separate study the loss of contractile function is consistent throughout the time course of the study. Closed chest hemodynamic assessment is performed on mice anesthetized with 3% Chloral hydrate, 300mg/Kg prior to insertion of micro-tip pressure transducer (FT111B, Scisense) into the right carotid artery and advanced into left ventricle. The catheter is connected to A/D converter (FV892A, Scisence) for data acquisition. Echocardiographic measurement of cardiac function Animals receiving LAD artery ligation are scanned on days 2, 28, and 56 using a Siemens-Acuson Sequoia C512 system equipped with a multi frequency (8–14 MHz) 15L8 transducer. Fractional shortening (FS) is acquired using standard M-mode image acquisitions of left ventricular short axis images as previously described (2). BLI of cell survival and localization Severe Combined Immunodeficiency Disease (SCID) mice (8 weeks of age; Charles Rivers, MA) are anesthetized by inhaled isoflurane (2%), intubated, and ventilated. A left thoracotomy is performed, followed by ligation of the left anterior descending (LAD) artery for 30 minutes after which 1x106 hCPCe Luc or hCPCeP Luc are injected intramyocardially into 3 sites near the peri-infarct zone in a total volume of 20 µl as previously described (2). Survival and engraftment of hCPC in vivo is assessed by BLI performed on animals receiving 375 mg/kg body weight d-luciferin reporter probe on days 2, 4, 7, 14, 21, 28, 35, 42, 49, and 56 using an IVIS Spectrum (Xenogen, Alameda, CA). Living Image 4.0 (Caliper Life Sciences, Hopkinton, MA) software is used to quantify signal intensity in units of photons/sec/cm2/sr as previously described (2). Magnetic resonance imaging (MRI) MRI is performed using a Signa 3.0T Excite HD scanner (GE Healthcare Systems, Milwaukee, WI; http://www.gehealthcare.com/euen/mri/index.html) with a Mayo Clinic T/R MRI coil (Mayo Clinic Medical Devices, Rochester, MN). Briefly, mice are anesthetized with 2% isoflurane and placed in the supine position for imaging. A small animal electrocardiogram and respiratory gating system (Small Animal Instruments, Stony Brook, New York) are used to acquire images as previously described (3). Following localization, gradient recalled echo (GRE) and fast spoiled GRE (FSPGR) sequences are used to acquire sequential short-axis slices from apex to base of the mouse heart every 1.5 mm. For each sequence, 20 cine frames encompassing 1 cardiac cycle are obtained with the following sequence parameters: TR = 10 ms, TE = 4.6 ms, number of excitations (NEX) = 2, field of view (FOV) = 60 × 60 mm, matrix = 256 × 256, flip angle (FA) = 30°, and slice thickness 1.5 mm. A contouring program, OsiriX (OsiriX Foundation, Geneva, Switzerland) is used to trace the endocardial border of the LV myocardium for each slice of the heart over the entire cardiac cycle to determine ejection fraction (EF), end-diastolic (EDV) and end-systolic volumes (ESV). Histology and Staining Hearts are retroperfused through the abdominal aorta with heparin (Sigma) followed by relaxation buffer to arrest in diastole followed by fixation in formalin for 24 hours. Hearts are processed, embedded in paraffin, and 5µm sections are cut. Slides are deparaffinized in xylene for 5min for three changes, followed by 3 changes in 100% ethanol for 3 minutes each, 95% ethanol 2 changes for 3 minutes, 70% ethanol for 3 minutes and in deionized water 3 changes for 3 minutes. After deparafinization, the sections are boiled 10mmol/L citrate buffer (pH 6.0) followed by quenching with 3%H2O2/1XTN for 20 min (if required) otherwise proceed to blocking with Tris-NaCl-Blocking buffer (TNB; Perkin Elmer catalog FP1020) for 30 minutes. Primary antibodies are applied overnight at 4°C in TNB buffer. The next day secondary antibodies are applied for 2 hour at room temperature after washing with TN buffer. When amplification is required, slides are incubated with streptavidin horseradish peroxidase-conjugated antibody for 30 minutes at room temperature and signal is developed using tyramide substrate diluted 1:50 v/v in amplification diluent (Perkin-Elmer) for 10 minutes. Slides are washed in 1XTN for 5 minutes 3 changes before staining for DNA. The slides are mounted in Vectashield mounting medium (Vector Labs) and scanned using Leica TCS SP2 confocal laserscanning microscope. A List of Primary and secondary antibodies used for immunostaining is provided in Table S1. Telomere Length Measurements Telomere length is analyzed by quantitative in situ hybridization (Q-FISH) and confocal microscopy. PNA probe was purchased from DAKO (K5325) and measurements were done according to manufacturer’s protocol. Briefly, slides were deparaffinized and rehydrated, followed by antigen retrieval using 10mM Citrate buffer pH 6.0. Slides were subsequently washed with PBS and treated for 9 minutes with Proteinase K (Dako, S3020). Following another PBS wash slides were dehydrated with a series of cold ethanol then allowed to dry. PNA probe was placed on the section and denatured at 80C for 10 minutes and hybridized at 37C overnight. Slides were washed two times in Wash Buffer at 65C and counterstained with Sytox blue. Telomere signal was acquired in each nucleus using Leica software and divided by Sytox blue signal to account for differences in nuclear size. Telomerase Activity Telomerase activity is assessed by quantitative PCR according to manufacturer protocol (TRAPeze RT, Chemicon S7710). Briefly, hCPCs are harvested in CHAPS buffer and centrifuged at 4°C. 1 μg of lysate is obtained then incubated in a solution containing reverse transcriptase reaction mix and Taq polymerase at 30°C for 30 minutes. Control cells provided by the manufacturer are used as positive control and serial dilutions of control template TSR8 provided by the manufacturer is employed for quantitation. CHAPS buffer in the absence of protein lysates and heat-inactivated lysates are used as negative control. PCR cycling conditions and data analysis are performed according to manufacturer protocol. In Situ Hybridization Human Alu repeats are identified using in situ hybridization on paraffin embedded experimental hearts and human left ventricular assist device plugs as a positive control. Deparaffinization and antigen retrieval is performed as described previously. Following antigen retrieval and subsequent PBS washes slides are treated with Digest All 3 Pepsin (00-3009, Invitrogen) at 37C for 5 minutes. Slides are then washed and dehydrated and allowed to completely dry. Biogenex Alu probe is added to the slide and placed at 80C for 10 minutes to facilitate denaturation. The slides are then incubated at 37C overnight. The following day the slides are washed three times with 50% formamide/ 2X SSC at 37C, then 3 times with 2X SSC. Endogenous peroxidase activity is quenched with 3% peroxide for 20 minutes, and then slides are treated with anti-fluorescein HRP (Roche) for 30 minutes at room temperature. Following a PBS wash, slides are treated with Tyramide FITC (Perkin Elmer, NEL 701001KT) according to manufacturer protocol, washed and counterstained with Topro-3. Animal Studies Institutional Animal Care and Use Committee approved all animal studies. Statistics All data are expressed as mean +/- SEM. Student’s t-test is used when comparing two groups, one-way or two- way repeated measures. ANOVA with post Bonferroni test is used to compare more than two groups. Kolmogorov Smirnov test is used for TRAP assay. P value less than 0.05 is considered as statistically significant. Statistical Analysis is performed using Graph Pad Prism v 5.0 software. References: 1. Fischer KM, Cottage CT, Wu W, et al. Enhancement of myocardial regeneration through genetic engineering of cardiac progenitor cells expressing Pim-1 kinase. Circulation 2009;120:2077– 87. 2. Cao F, Wagner RA, Wilson KD, et al. Transcriptional and functional profiling of human embryonic stem cell-derived cardiomyocytes. PLoS One 2008;3:e3474. 3. Yue P, Arai T, Terashima M, et al. Magnetic resonance imaging of progressive cardiomyopathic changes in the db/db mouse. Am J Physiol Heart Circ Physiol 2007;292:H2106–18. Supplementary Figure 1 Human CPCs overexpressing Pim-1 A) Human CPCs in culture, B) characterization of human CPCs by FACS analysis, C) Schematic diagram of lentiviral vectors for eGFP and eGFP Pim-1, D) percentage of GFP in hCPCe and hCPCeP, E) immunoblot analysis of human CPCs for overexpression of Pim-1 and GFP lentiviral transduction, F) karyotype analysis of hCPCe and hCPCeP. Supplementary Figure 2 Colocalization of human mitochondrial marker and Alu with GFP staining: Immunohistochemistry for human mitochondrial marker (white), GFP (green), Desmin (red) and nuclei (blue) respectively for A) vehicle B) remote zone, C) infarct zone of hCPCeP injected mice 12 weeks after transplantation. Scale bar = 50μm. FISH analysis for Alu (green), sarcomeric actin (red) and nuclei (blue) for D) human heart sample E-F) infarct zone of mice transplanted with hCPCeP after 12 weeks G) Human heart sample no probe (control) H-I ) Infarct zone of mice with hCPCeP 20 week after transplantation. Scale Bars: A-C = 50μm, D, G-I = 40μm and E-F = 10μm. Supplementary Figure 3 Pim-1 expression remains increased in hCPCeP at 12 weeks post- transplantation: Confocal microscopy images show immunolabeling for Pim-1 in hCPCe (A) or hCPCeP (B). Pim-1 (white), GFP (green), α-sarcomeric actin (red) and nuclei (blue). Scale bar = 40μm