Note packet
advertisement

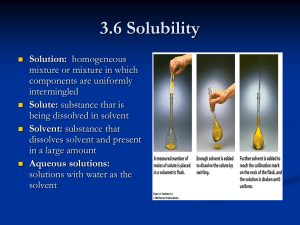
SOLUTIONS UNIT Name:_________________________________________________Period:____Date:____ I. MIXTURES: 1) ____________________ = a blend of two or more kinds of matter, each of which retains its own identity and properties can be physically separated (filtration, evaporation, decanting, magnetism, etc) a) ___________________________________= a mixture that is uniform in composition throughout Ex: Food coloring and water b) ___________________________________= a mixture that is NOT uniform in composition throughout Ex: Oil and water A. Types of Mixtures: 1) ______________________________= a homogeneous mixture 2) ______________________________= a mixture in which the particles are so large that they settle out unless the mixture is constantly stirred or agitated Heterogeneous mixture Ex: Sand and water 3) ____________________________= a mixture consisting of particles that are intermediate in size between those in solutions and those in suspensions Heterogeneous mixture Ex: Milk, mayonnaise, smog, butter, whipped cream 1 a.) What property of a colloid helps to prevent colloid particles from settling out of a mixture? ___________________________________= the random continuous motions of colloidal particles b.) _____________________________________= visible pattern caused by the reflection of light from suspended particles in a colloid (or from suspended particles in a suspension if the particles have not settled out) Ex: visibility of a headlight beam on a foggy night B. Comparison of solution, colloids, and suspensions Solutions Colloids Suspensions Heterogeneous Heterogeneous Particle size: 1-1000 nm, Particle size: over 1000 nm, dispersed; can be aggregates or suspended; can be large particles large molecules or aggregates _______ separate on standing ________ separate on standing Particles settle out Cannot be separated by Cannot be separated by filtration filtration ___________ scatter light Scatter light (Tyndall effect) _________________ Particle size: 0.01-1 nm; can be atoms, ions, molecules _____ be separated by filtration ______ scatter light, but are not transparent C. Determining if a mixture is a true solution, a colloid, or a suspension: 1) If particles settle or can be filtered out = ___________________ 2) If particles DO NOT settle or filter out shine a beam of light (Tyndall effect) through the mixture If the Tyndall effect is observed = ____________________________ 2) If the Tyndall effect is NOT observed = _________________________ THE NATURE OF SOLUTIONS: 1) ______________ = the substance that does the dissolving in a solution a) Typically present in the greatest amount b) Typically a liquid c) ____________________ is the most common or “universal” solvent B/c water molecules are __________________________ 2 The hydrogen side of each water (H2O) molecule carries a slight positive electric charge, while the oxygen side carries a slight negative electric charge. Water can dissociate ionic compounds into their positive and negative ions The positive part of an ionic compound is attracted to the oxygen side of water while the negative portion of the compound is attracted to the hydrogen side of water. Water won't dissolve or won't dissolve well. If the attraction is high between the opposite-charged ions in a compound, then the solubility will be ________________. o Ex: hydroxides exhibit low solubility in water. o Ex: nonpolar molecules don't dissolve very well in water (fats and waxes) 2) ________________________ = substance being dissolved in a solution a) Typically present in the least amount b) Typically a solid A.9 Possible Solution Combinations: Solvent Gas Solute Common Example Gas Diver’s tank Gas Liquid Gas Solid Liquid Gas Liquid Liquid Liquid Solid Solid Gas Solid Liquid Solid Solid Moth ball Vinegar o NOT all solutions are liquids/solids! o Solutions are formed in ALL 3 states! Gas stove lighter Sterling Silver (Ag + Cu) C. Solvation: 1) ___________________________________ = the process of dissolving a.) First- solute particles are surrounded by solvent particles b.) Then- solute particles are separated and pulled into solution 2) ____________________________________= separation of an ionic solid into aqueous ions 3 Ex: NaCl + H2O – the Na ion and Cl ion become hydrated and gradually move away from the crystal into solution. Each ion in the solution acts as though it were present________________: So there is only a solution containing Na+ and Cl- ions uniformly mixed with H2O particles NaCl(s) Na+(aq) + Cl–(aq) 3) ____________________________= breaking apart of some polar molecules into aqueous ions Ex: HNO3(aq) + H2O(l) H3O+(aq) + NO3–(aq) 4) _________________________________________ =molecules stay intact Ex: C6H12O6(s) C6H12O6(aq) D. Factors Affecting the Rate of Dissolving (Increases Solution Rate): 1) ____________________: increases surface area exposed to solvent 2) ____________________: allows solvent continual contact with solute 3) ____________________: increases kinetic energy; increases mixing 3) Electrolytes and Nonelectrolytes A. Electrolytes and Nonelectrolytes 1) ________________________ = a substance that dissolves in water to give a solution that conducts electric current 2) _________________________ = a substance that dissolves in water to give a solution that does NOT conduct an electric current 3) Solutions of electrolytes can conduct electric current: a) The positive ions and the negative ions ___________________________(separate) in solution. The ___________________ions can move a charge from one point in the solution to another point 4) Solutions of nonelectrolytes CANNOT conduct electric current: a) When a nonelectrolyte dissolves in water there are NO _________________________________ in solution. b) Ex: Solute exists as molecules 5) Weak Electrolytes a) Only a portion of dissolved molecules ionize. 6) Solid ___________________ compounds cannot conduct electric current: a) Ions are present but they are NOT ___________________. 4 4) SOLUBILITY: 1) _________________________= quantity of solute that will dissolve in specific amount of solvent at a certain temperature. (Pressure must also be specified for gases). a) Ex: 204 g of sugar will dissolve in 100 g of water at 20C a) soluble and insoluble are relative terms b) solubility should NOT be confused with the rate at which a substance dissolves 2) _______________________ = a stable solution in which the maximum amount of solute has been dissolved. a) Visual evidence: a quantity of undissolved solute remains in contact with the solution 3) __________________________________ = state where the solute is dissolving at the same rate that the solute is coming out of solution (crystallizing). a) Opposing processes of the dissolving and crystallizing of a solute occur at equal rates. b) solute + solvent solution 4) _________________________________ = a solution that contains less solute than a saturated solution under existing conditions 5) __________________________________= a solution that temporarily contains more than the saturation amount of solute than the solvent can hold. Unstable – if disturbed, the excess solute will crystallize out of solution B. 3 FACTORS EFFECTING SOLUBILITY: 1) Nature of solute and solvent a) “____________________________” = rule of thumb for predicting whether or not one substance dissolves in another “Alikeness” depends on: o Intermolecular forces o Type of bonding o Polarity or nonpolarity of molecules: ________________ solutes tend to dissolve in polar solvents but not in nonpolar solvents b) Solvent-Solute Combinations: Solvent Type Solute Type Polar Polar Polar Nonpolar Nonpolar Polar Nonpolar Nonpolar Is Solution Likely? “Like Dissolves Like” 5 2) __________________________: a) Pressure has __________________ on the solubility of liquids or solids in liquid solvents. b) The solubility of a _________ in a ______________ INCREASES when pressure __________________. It is a direct relationship. 3) ______________________________: a) The solubility of a gas in a liquid solvent DECREASES with an ______________ in temperature. b) The solubility of a solid in a liquid solvent MOST OFTEN increases with an increase in temperature. However, solubility changes vary widely with temperature changes sometimes decreasing with temperature increases. 5) SOLUBILITY GRAPH: A. Solubility Curve 1) ______________________ = any point on the line or ABOVE the line 2) ___________________________ = any point BELOW the line B. Solubility Curve Problems 1) What is the solubility of the following solutes in water? a) NaCl at 60ºC = ____________ b) KCl at 40ºC = _____________ c) KNO3 at 20ºC = ____________ 2) Are the following solutions saturated or unsaturated? Each solution contains 100 g of H20. a) 31.2 g of KCl at 30ºC = _______________ b) 106g KNO3 at 60ºC = _______________ c) 40 g NaCl at 10ºC = _______________ d) 150 g KNO3 at 90ºC = _______________ 6 3) For each of the following solutions, explain how much of the solute will dissolve and how much will remain undissolved at the bottom of the test tube? a) 180 g of KNO3 in 100 g of water at 80ºC ______________________________ ______________________________ b) 180 g of KNO3 in 100 g of water at 20ºC _______________________________ _______________________________ c) 60 g of NaCl in 100 g of water at 60ºC ________________________________ ________________________________ 4) A saturated solution of KNO3 is formed from one hundred grams of water. If the saturated solution is cooled from 90°C to 30°C, how many grams of precipitate are formed? _______________________________ 5) A saturated solution of KCl is formed from one hundred grams of water. If the saturated solution is cooled from 90°C to 40°C, how many grams of precipitate are formed? __________________________________ 6) CONCENTRATION: MOLARITY & MOLALITY 1) ______________________________= The amount of solute in a solution. a) Describing Concentration: • % by mass - medicated creams • % by volume - rubbing alcohol • ppm, ppb - water contaminants • molarity - used by chemists • molality - used by chemists A. Molarity: 1) ___________________________= unit of concentration of a solution expressed in moles of solute per liters of solution. • Formula: M = ___MOLES of solute______ LITERS of solution Ex: 2M HCl o What does that mean? Mol M= 2M HCl = L 2 mol HCl 1L 7 B. Molarity Calculations: 1) To solve for Molarity when given grams of solute, you will use gram/mole conversion and the equation for Molarity. Use the following steps: a) Change grams of solute to moles. To do this you need to use the following conversion: Grams of solute 1 mole of solute = moles of solute Molar mass of solute* *make sure formulas are correct; Use ion list! b) Change the volume of solution to liters (L) by moving the decimal 3 places to the LEFT. Remember that 1000 mL = 1 L. Water will most often be the solvent in a solution. c) Substitute the information into the molarity equation and solve. 2) Ex: What is the molarity of a solution composed of 22.4g of sodium chloride dissolved in enough water to make 500mL of solution? 22.4 g of NaCl 1 mole_NaCl = 0.383 moles NaCl 58.443 g NaCl 0.383 moles NaCl = 0.766 M NaCl Note: You need to change mL of solution to L. So move decimal 3 places to the left! 0.500 L solution 3) Use the Molarity equation to solve for grams of solute. You will need to solve the Molarity equation for moles of solute. Then convert moles of solute to grams of solute. 4) Ex: How many grams of lithium bromide are present in 300mL of a 0.4M lithium bromide solution? a) Substitute the information and solve for moles by cross multiplying or multiplying by the reciprocal: “X” moles LiBr___ = 0.4M LiBr 0.300 L solution X = 0.12 moles of LiBr are needed b) Convert moles to grams using the following conversion: Moles of solute molar mass of solute = grams of solute 1 mole of solute The substitution would look like: 0.12 moles LiBr 86.845 g LiBr = 10. g LiBr 1 mol LiBr 8 C. Dilution Calculations: 1) Sometimes solutions of lower concentrations are made from existing solutions. 2) Moles of solute remain the same. 3) Formula: M1V1 = M2V2 M1 is the original molarity concentration V1 is the volume of the original solution (in L) M2 is the new concentration V2 is the amount of the new solution needed (in L) 4) Ex 1: What volume of 15.8M HNO3 is required to make 250 mL of a 6.0M solution? GIVEN: WORK: M1 V1 = M2 V2 M1 = 15.8M V1 = ? (15.8M) V1 = (6.0M)(250mL) M2 = 6.0M V2 = 250 mL V1 = 95 mL of 15.8M HNO3 D. Molality: 1) _____________________________(m) = a unit of concentration of a solution expressed in moles of solute per kilogram of solvent. a) Ex: A solution that contains 1 mol of solute, ammonia (NH3), dissolved in exactly 1 kg solvent, is a “one molal” solution. b) Formula: m = _MOLES of solute kg of solvent c) Note: 1 kg water = 1 L water E. Calculations: Solving for Molality: 1) If needed convert: grams of solute moles of solute mL of solvent kg of solvent (move decimal 3 places to the left!) 2) Solve for molality m = moles of solute kg of solvent 3) Ex: Find the molality of a solution containing 75 g of MgCl2 in 250 mL of water. 9 F. Calculations: Using the Molality equation to solve for grams of solute: 1) Use Molality equation and solve for moles 2) Then convert moles of solute to grams of solute 3) Ex: How many grams of NaCl are required to make a 1.54m solution using 0.500 kg of water? COLLIGATIVE PROPERTIES: A. Colligative Property: 1) ___________________________________= A property that depends on the NUMBER of solute particles rather than the type of particle. B. Examples of colligative properties: 1) ______________________________________ LOWERING of a solution a) Ex: We have two beakers. One contains H2O, the other contains a salt solution (NaCl) in H2O. Both beakers are placed into a sealed chamber: DAY 3: If we leave it for a couple of days and then come back and take a look, this is what it might look like. DAY 1 b) WHAT HAPPENED?: Na+ and Cl- ions are non-volatile ions. In other words, they will not leave and go into the vapor phase. Those H2O molecules with enough kinetic energy will leave the surface of the solutions and enter into the vapor phase. The fact that Na+ and Cl- ions are dissolved in H2O indicates that there is an attractive interaction between the solutes and the H2O. 10 The interaction of the Na+ and Cl- ions for H2O will act to hinder the ability of the solvated H2O molecules to leave and go into the vapor phase (Na+ and Cl- ions are nonvolatile). This hindrance of the H2O molecules to enter the vapor phase will reduce the vapor pressure of the H2O in the salt-containing solution. The two beakers are in the same sealed container, thus, the vapor pressure above the solutions is identical. The rate of H2O entering the solutions by collisions from the vapor state will be identical The rate of H2O leaving the liquid phase and entering the vapor phase is slower for the NaCl-containing solution 2) _______________________________________ = If two solutions, with different solute concentrations, are separated by a semi-permeable membrane, there can be a net flow of solvent across the membrane. 3) _________________________________________= The solution will begin to freeze at a temperature BELOW that of the pure solvent. 4) _________________________________________ = the solution will begin to boil at a temperature ABOVE that of the pure solvent. C. How are solutions different than pure liquids? 1) One of the ways in which they are different, is that when you add a solute to a liquid _________ the freezing point and boiling point of the solution ________________________. a) ____________________________________ – In a solution, the solute particles interfere with the attractive forces among the solvent particles. This prevents the solvent from entering the solid state at its normal freezing point b) ____________________________________- When the temperature of a solution containing a nonvolatile solute is raised to the boiling point of the pure solvent, the resulting vapor pressure is still less than the atmospheric pressure and the solution will not boil. Thus, the solution must be heated to a higher temperature to supply the additional KE needed to raise the vapor pressure to atmospheric pressure. 2) Water is the liquid we will be dealing with most often a) The freezing point of pure water is _______. b) The normal boiling point of water _________. c) But if you make a solution using water as the _____________________, the freezing point of that solution will _________ be 0°C nor will the boiling point be 100°C! D. Freezing Point Depression & Boiling Point Elevation: 1) ____________________________________________(TFP) – solutions will freeze at LOWER temperature than the pure solvent a) The more solute dissolved, the _________________the effect. 11 b) Ex: ethylene glycol (antifreeze) protects against freezing of the water in the cooling system by lowering the freezing point to about -40°C c) Ex: making of homemade ice cream- The ice cream mix is put into a metal container which is surrounded by crushed ice. Then salt is put on the ice to lower its melting point. This gives a temperature gradient across the metal container into the saltwater-ice solution which is lower than 0°C. The heat transfer out of the ice cream mix allows it to freeze. 2) _______________________________________________(TBP) – solutions will boil at HIGHER temperatures than the pure solvent a) The boiling point of pure water is 100°C, but that boiling point can be elevated by the addition of a solute such as a salt. b) The more solute dissolved, the _____________________the effect. E. Calculating Freezing Point Depression & Boiling Point Elevation: 1) Solution concentrations are given in molality (m) 2) Colligative properties are ______________ proportional to the molal concentration of a solute 3) Account for particle molality for ELECTROLYTES Ex: NaCl = 2 ions (Na+ & Cl-) Ex: MgCl2 = 3 ions (Mg+2 and 2 Cl-) 4) A change in the concentration (m), changes the freezing point and boiling point of a solution a) You will need to figure out the change in temperature for the normal freezing point (FP) or boiling point (BP) and adjust them with the following steps: ∆T = mK ∆T = change in temperature m = moles solute/kg solvent (MOLALITY) K = constant You will mostly use the constants for water: Kfp = 1.853 ˚C/m Kbp = 0.515˚C/m Other values can be found below: FREEZING Point Depression Constants BOILING Point Elevation Constants Compound Freezing Point (oC) Kfp (oC/m) Compound Boiling Point (oC) Kbp(oC/m) water 0.000 1.853 water 100.000 0.515 acetic acid 16.660 3.90 ethyl ether 34.550 1.824 benzene 5.530 5.12 carbon disulfide 46.230 2.35 p-xylene 13.260 4.3 benzene 80.100 2.53 naphthalene 80.290 6.94 carbon tetrachloride 76.750 4.48 cyclohexane 6.540 20.0 camphor 207.420 5.611 12 4) Steps solving for ∆T: a) Solve for molality (see steps from previous problems) m = moles of solute kg of solvent b) Find your K constant from the chart listed in your notes. Be sure to find the correct K constant for what you are solving for-- either FP or BP!! c) Solve for ∆T ∆T = mK d) If solving for: Freezing point: Take the normal freezing point of the solvent and SUBTRACT the ∆T value o Round final answer to 3 significant figures! Boiling point: Take the normal boiling point of the solvent and ADD the ∆T value o Round final answer to 6 significant figures! 5) Ex #1: At what temperature will a solution that is composed of 32.8 g of glucose (C6H12O6) in 225 g of water boil? 6) Ex #2: Find the freezing point of a saturated solution of NaCl containing 28.0 g NaCl in 115 g water. 13 Solubility Curve Worksheet 1) Look at the graph below. In general, how does temperature affect solubility? ________________________________________________________________________________ 2) Which compound is LEAST soluble at 10 °C? ______________________ 3) How many grams of KCl can be dissolved in 100g of water at 80°C? ______________________ 4) How many grams of NaCl can be dissolved in 100g of water at 90°C? ______________________ 5) At 40°C, how much KNO3 can be dissolved in 100g of water? ___________ 6) Which compound shows the least amount of change in solubility from 0°C-100°C? ________________ 7) At 30°C, 90g of NaNO3 is dissolved in 100g of water. Is this solution saturated or unsaturated? _____________ 8) At 60°C, 72g of NH4Cl is dissolved in 100g of water. Is this solution saturated or unsaturated? _____________ 9) A saturated solution of KClO3 is formed from one hundred grams of water. If the saturated solution is cooled from 90°C to 50°C, how many grams of precipitate are formed?_______________ 10) A saturated solution of NH4Cl is formed from one hundred grams of water. If the saturated solution is cooled from 80°C to 40°C, how many grams of precipitate are formed?_______________ 11) Which compounds show a decrease in solubility from 0°C-100°C? _____________________________ 12) Which compound is the most soluble at 10°C? _____________________________ 13) Which compound (besides Ce2(SO4)3) is the least soluble at 50°C?________________________ 14) For each of the following solutions, explain how much of the solute will dissolve and how much will remain undissolved at the bottom of the test tube? a) 120 g of KCl in 100 g of water at 80ºC _______________________________ _______________________________ b) 130 g of NaNO3 in 100 g of water at 50ºC _______________________________ _______________________________ 14 Molarity Worksheet 1) What is the molarity of a solution that is composed of the following?: a) 30g of calcium chlorate dissolved in enough water to make 370mL of solution b) 450g of potassium iodide dissolved in enough water to make 4L of solution c) 25g of hydrochloric acid dissolved in enough water to make 600. mL of solution 15 d) 0.750g of aluminum sulfate dissolved in enough water to make 75.0 mL of solution e) 35.5g of silver nitrate dissolved in enough water to make 125mL of solution 2) How many grams of each of the following solute are needed to make the solution: a) 215 mLof 0.400M iron III nitrate b) 565 mL of 3.00M potassium iodide 16 c) 355 mL of 0.250M chromium II permanganate d) 400.0 mL of 6.00M zinc sulfate e) 800.0 mL of 1.00M magnesium acetate 17 Dilutions Worksheet 1) What formula is used when solutions of lower concentrations are made from existing solutions? 2) What volume of 5.30M ammonium phosphate is required to make 200.mL of a 4.25M solution? 3) What volume of 2.50M mercury I chromate is required to make 225mL of a 0.200M solution? 4) What volume of 0.5M copper II cyanide is required to make 25mL of a 0.10M solution? 5) What volume of 1.50M tin II fluoride is required to make 250. mL of a 0.875M solution? 6) What volume of 2.55M manganese dichromate is required to make 750. mL of a 0.150M solution? 18 Molality (m) Worksheet 1) Determine the molality of each of the following solutions: a) 199g of nickel II bromide in 500. g H2O b) 4.5 mol Mg(NO3)2 in 2.5 kg H2O c) 0.625 mol K2SO4 in 850. g H2O d) 92.3g of potassium fluoride in 137g H2O 19 e) 85.2g tin II bromide in 142g H2O 2) How many grams of KCl are required to make a 0.525m solution using 250. g of water? 3) How many moles of each solute would be required to prepare each of the following solutions? a) 0.45 m CaSO4 in 2.5 kg H2O b) 3.25 m Ba(NO3)2 in 750. g H2O 20 4) Determine how many grams of solute is required to make each of the following solutions: a) A 4.50 m solution of H2SO4 in 1.25 kg H2O b) A 3.50 m solution of MgCl2 in 0.450 kg H2O 5) A solution is prepared by dissolving 17.1 g of sucrose (C12H22O11), in 275 g of H2O. What is the molality of that solution? 21 6) How many kilograms of H2O must be added to 75.5 g of Ca(NO3)2 to form a 0.500 m solution? 7) How many grams of glucose (C6H12O6), must be added to 750. g of H2O to make a 1.25 m solution? 22 Freezing Point Depression & Boiling Point Elevation Worksheet: Directions: Round final answer to 3 significant figures for freezing point depression problems and round final answer to 6 significant figures for boiling point elevation! 1) If 68.4g of sucrose C12H22O11 are dissolved in 500. g of water, what will be the boiling and freezing points of the solution? 2) Calculate the freezing point of a solution containing 5.75 g of sugar C12H22O11 in 59.5 g of water. 3) Calculate the freezing point of solution that contains 32.7 g of sodium acetate dissolved in 250. g of water. 23 4) Calculate the freezing point, if 46.5 g of glycerol, C3H5(OH)3, are dissolved in 500. g of water. 5) Calculate the boiling point of 30.8 g of silver nitrate in 500. g water. 6) Determine the boiling point of 16.3 g of calcium chloride dissolved in 250. g water. 7) Determine the boiling point of 23.9 g of copper (II) sulfate in 300. g water 24 Review Molarity, Dilutions, Molality, Freezing Point Depression & Boiling Point Elevation: 1) What is the molarity of a solution that is composed of the following?: a) 26g of calcium chloride dissolved in enough water to make 425mL of solution. b) 329g of calcium iodide dissolved in enough water to make 2.5L of solution. 2) How many grams of each of the following solute are needed to make the following solutions?: a) 500. mL of 3.40M iron (III) nitrate. b) 700. mL of 0.880M lithium sulfate 25 3) What volume of 6.25M ammonium chloride is required to make 500. mL of a 3.92M solution? 4) What volume of 5.50M sodium dichromate is required to make 725mL of a 0.600M solution? 5) Determine the molality of each of the following solutions: a) 254 g of NaCl in 575 g water b) 9.65 mol MgCl2 in 675 kg water 26 6) Determine the freezing point and boiling point of a solution containing 98.5 g of NaCl in 100. g water. 7) Determine the freezing point and boiling point of a solution containing 425 g of CaCl 2 in 550. g water. 27 SOLUTIONS CONCEPT REVIEW QUESTIONS: 1) Explain why solutions are classified as mixtures instead of compounds. 2) What are the three different types of mixtures? 3) Differentiate between a homogeneous mixture and a heterogeneous mixture. 4) Classify each of the following as a heterogeneous mixture or a homogeneous mixture. a) Salad __________________________________ b) Tap water _______________________________ c) Muddy water ____________________________ 5) What another name for a solution? ______________________________ 6) What is the difference between a solute and a solvent? 7) What is the Tyndall Effect? Give a common example of this effect. 8) In what type of mixture is to easiest to separate the component substance? Why? 9) How does a solution behave differently from a suspension when a beam of light is shined through it? 28 10) What property of a colloid helps to prevent colloid particles from settling out of a mixture? 11) Given an unknown mixture consisting of two substances, explain how a scientist could use lab techniques to determine whether the mixture is a true solution, a colloid, or a suspension. 12) What is considered to be the “universal” solvent? ___________________ a) Explain why. 13) What is the solute in a brass alloy containing 75% copper and 25% zinc? ____________________ a) How do you know? 14) Not all solutions are solids dissolved in liquids. Provide two examples of other types of solutions. 15) Suppose you were making a salt water solution. Sodium ions and chloride ions are separating from one another. a) What is this called? _____________________________________________ 16) Suppose you were making a salt water solution. Sodium ions and chloride ions are each being surrounded by water molecules. b) What is this called? _______________________________________ 17) Describe how stirring increases the rate of dissolving of a solute. 18) Describe how heating increases the rate of dissolving of a solute. 29 19) Describe how grinding a solute increases the rate of dissolving of a solute. 20) Define solubility. 21) What are 3 factors that affect solubility? a) ________________________________________ b) ________________________________________ c) ________________________________________ 22) What happens to the solubility of a gas in a liquid solvent as temperature increase? 23) Explain the rule, “Like Dissolves Like”. 24) State whether each of the following will conduct an electric current. Also, explain why each does or does not conduct an electric current. a) salt (NaCl) water b) sugar (C12H22O11) water c) solid NaCl 30 25) When does solubility equilibrium occur? 26) What are the differences between a saturated solution, unsaturated solution and a supersaturated solution? 27) How could you tell by looking at a solution that it was saturated? 28) Differentiate between molarity and molality. 29) What does 5M HNO3 mean? 30) What is the molarity formula? 31) What is the molality formula? 32) What formula is used when solutions of lower concentrations are made from existing solutions? 31 33) Define colligative property. 34) Provide 4 examples of colligative properties. 35) Explain how the boiling point and freezing point of solutions are different than pure liquids? Use the solubility cure below to answer the following questions: 36) Which salt is LEAST soluble at 20 °C? ______________________ 37) How many grams of KBr can be dissolved in 100g of water at 60°C? ______________________ 38) How many grams of NaCl can be dissolved in 100g of water at 100°C? ______________________ 39) At 40°C, 180g of NaClO3 is dissolved in 100g of water. Is this solution saturated or unsaturated? _____________ 40) At 70°C, 70g of KBr is dissolved in 100g of water. Is this solution saturated or unsaturated? _____________ 41) A saturated solution of NaClO3 is formed from one hundred grams of water. If the saturated solution is cooled from 80°C to 60°C, how many grams of precipitate are formed?_______________ 42) How much of the solute will dissolve and how much will remain undissolved at the bottom of the test tube? d) 160 g of KNO3 in 100 g of water at 50ºC ____________________________ ____________________________ 32 SOLUTIONS TEST STUDY GUIDE Chapter 13 (pg 513-547) Read over your notes and rework your class/homework assignments. Study your lab reports. You will do “Mole-tastic” on this test if you can do the following things. Mixtures Explain the difference between mixtures and compounds? Explain the difference between mixtures (solutions, colloids, suspensions). What can be done experimentally to differentiate between the 3 types of mixtures? Examples of each type of mixture Define the Tyndall Effect. Nature of Solutions Explain the difference between solute and solvent? Explain why is water considered the “universal solvent”? Describe at a molecular level the dissolving of an ionic compound in water. Explain the 3 factors that can increase the solution rate. Electrolytes & Non-electrolytes Differentiate between electrolytes and non-electrolytes. What properties do electrolytes have that are not present in non-electrolytes. Solubility Define solubility. Explain the difference between a saturated, unsaturated and a supersaturated solution. Explain the general rule of thumb, “like dissolves like”. Explain the 3 factors that affect solubility. Solubility Graph Be able to interpret a solubility curve Concentrations Explain the difference between Molarity and Molality Be able to solve Molarity problems For Molarity For grams of solute Be able to calculate how to dilute a solution Be able to solve Molality problems For Molality For moles of solute For grams of solute Colligative Properties Define colligative property. Describe examples of colligative properties. How are solutions different than pure liquids? Explain what happens to boiling and freezing points Calculate freezing point and boiling point of solutions. 33