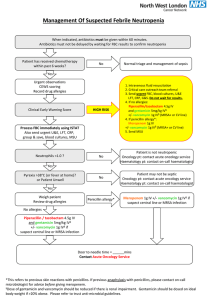
calves 76
advertisement

ΠΑΡΑΡΤΗΜΑ 1: ΠΕΡΙΛΗΨΗ ΤΩΝ ΧΑΡΑΚΤΗΡΙΣΤΙΚΩΝ ΤΟΥ ΠΡΟΪΟΝΤΟΣ 1. Name of the veterinary medicinal product: Gentamycin 5% ,50 mg/ml, solution for injection for cattle, dogs, and cats. Gentamicin as Gentamicin sulphate. 2. Qualitative and quantitative composition: 1 ml solution for injection contains: Active substance(s): Gentamicin As Gentamicin sulphate Excipients(s): Methyl parahydroxybenzoate Propyl parahydroxybenzoate Sodium metabisulphite 50.0 mg 85.0 mg For a full list of Excipients, see section 6.1. 3. Pharmaceutical form: Solution for injection 4. Clinical particulars: 4.1 Target specie(s): Cattle, dog, cat. 4.2 Indications for use, specifying the target species: For treatment of the following diseases caused by Gentamicin-sensitive pathogens: Cattle: infections of the uterus. Calf: infections of the respiratory tract (bronchi, lung), infections of the gastrointestinal tract (enteritis caused by E. coli and Salmonella spp.), septicaemia, joint infections. Dog, cats: infections of the respiratory tract, the kidneys and urinary tract as well as the uterus, the gastrointestinal, inflammations of the auditory meatus, tonsillitis (not in cats). 4.3 Contraindications: Do not use in case of: - Bacterial resistance against Gentamicin, kanamycin, streptomycin dihydrostreptomycin and paromomycin. - Renal dysfunction. - Auditory or equilibral dysfunction. - Known hypersensitivity against Gentamicin or against other aminoglycoside antibiotics or any of the Excipients. 1/6 Do not administer concomitantly with bacteriostatic acting antibiotics, highly active diuretics and potential nephrotoxic medicines. Do not use concomitantly intravenously or intraabdominally when muscle relaxans are administered. Do not use in dehydrated animals because of risk of an acute kidney failure. 4.4 Special warnings for each target species: Not known. 4.5 Special precautions for use: Special precautions for use in animals: Because of the low therapeutic index of Gentamicin, dosage should be done with respect to the exact body weight. Generally, the administration of Gentamycin 5% should be done with respect of an antibiogram. Because of the risk of incidence of neuromuscular blockade by intravenous administration, Gentamycin 5% has to be injected very slowly. Administration of Gentamycin 5% requires strong indication. In dehydrated animals, fluid balance has to be corrected before treatment. Special precautions to be taken by the person administering the veterinary medicinal product to animals: Avoid direct contact of the product with the skin or mucosa because of the risk of sensitisation. 4.6 Adverse reactions (frequency and seriousness): Disturbances of the auditory and equilibral function and of the kidneys may be observed after prolonged administration. The dysfunction of the kidneys may be characterised by proteinuria and an increase of nonprotein nitrogen content in the blood. In incidences of neuromuscular blockade, which may lead to cramps, respiratory distress and collapse, the drug must be withdrawn immediately. An antidote calcium and neostigmin may be injected. In allergic and anaphylactic reactions, the drug must be withdrawn immediately and a corresponding therapy with epinephrine (adrenalin), antihistaminics and/or glucocorticoids must be implemented. Cats are especially sensitive to gentamicin. 4.7 Use during pregnancy, lactation or lay: In mice (s.c. 10 mg gentamicin/kg b.w.) and rats (i.m. 50 mg gentamicin/kg b.w.) foetotoxic effects have been demonstrated. Therefore, administration during pregnancy requires strong indication. 2/6 4.8 Interaction with other medicinal products and other forms of interaction: Gentamicin increases neuromuscular blockades. Do not use Gentafromm concomitantly with ototoxic and neurotoxic drugs. The combination therapy with suitable antibiotics (ß-lactam antibiotics) may induce synergistic effects. Synergistic effects with acylamino penicillins have been observed in Pseudomonas aeruginosa and synergistic effects with cephalosporines have been observed in Klebsiella pneumonia. 4.9 Amounts to be administered and administration route: By intramuscular, subcutaneous, and slow intravenous application.. Target species Dosage Gentamicin Eq. to gentamicin Gentamycin 5% sulphate Cattle 4 ml/50 kg b.w. 6.80 mg/kg b.w. 4.0 mg/kg b.w. Calf in the first few months of life Initial treatment 2 ml/25 kg b.w. 6.80 mg/kg b.w. 4.0 mg/kg b.w. after the second 1 ml/25kg b.w. 3.40 mg/kg b.w. 2.0 mg/kg b.w. treatment Each twice daily at intervals of 12 hours for 3 – 5 days Dogs older than 2 0.44 ml/5 kg b.w. 7.48 mg/kg b.w. 4.4 mg/kg b.w. weeks Dogs younger than 2 weeks Initial treatment 0.088 ml/1 kg b.w. 7.48 mg/kg b.w. 4.4 mg/kg b.w. after the 2nd 0.044 ml/1 kg b.w. 3.74 mg/kg b.w. 2.2 mg/kg b.w. treatment Each twice daily at intervals of 12 hours, after the 2nd day once daily at intervals of 24 hours for 3 – 10 days Cats older than 2 0.3 ml/5 kg b.w. 5.10 mg/kg b.w. 3.0 mg/kg b.w. weeks Cats younger than 2 weeks Initial treatment 0.03 ml/0.5 kg b.w. 5.10 mg/kg b.w. 3.0 mg/kg b.w. nd after the 2 0.015 ml/0.5 kg b.w. 2.55 mg/kg b.w. 1.5 mg/kg b.w. treatment Each twice daily at intervals of 12 hours for 3 – 10 days If after 3 days of treatment no therapeutical effects is obvious, diagnosis must be verified, and if necessary, therapy must be changed. If in case of proven sensitivity of the pathogens a longer duration of therapy than usual should be necessary, a regular control of the renal function must be done due to the danger of possible renal damage. 4.10 Overdose (symptoms, emergency procedures, antidotes), if necessary: Overdose and fast intravenous administration may induce neuromuscular blockade which may lead to cramps, respiratory distress and collapse. The drug must be withdrawn immediately and the in the paragraph “adverse reactions” mentioned counteractions are to be fulfilled. In allergic and anaphylactic reactions, the drug must be withdrawn 3/6 immediately and a corresponding therapy with epinephrine (adrenaline), antihistaminics and/or glucocorticoids must be implemented. Due to the oto- and nephrotoxic properties of gentamicin, overdosage will lead to corresponding symptoms. An immediate withdrawal of the drug is necessary. 4.11 Withdrawal period(s): Cattle, calf (edible tissues): Cattle (milk): 95 days 6 days 5. Pharmacological particulars ATCvet Code: QJ01GB03/gentamicin Pharmacotherapeutic group: aminoglycoside 5.1 Pharmacodynamic particulars: Gentamicin is an aminoglycosid antibiotic, gained from cultures of Micromonospora purpurea, resp. Micromonospora echinospora. It acts bactericidal due to inhibition of bacterial protein synthesis of proliferating germs by binding to the ribosomal structures and is effective against Gram-positive and Gram-negative pathogens (Proteus and Pseudomonas spp., E. coli) and certain fungi and mycoplasmas. During translation from DVA to RNA failure occurs and also formation of peptid conjunctions is disturbed. Pathogens are classified as susceptible if values of MIC ranging between 1 – 4 μg / ml. Development of resistance against gentamicin occurs slowly and is plasmid mediated. Resistance against gentamicin is accompanied by resistance against other aminoglycosides like neomycin, kanamycin, streptomycin, and dihydrostreptomycin, whereas vice versa resistant germs are still sensitive to gentamicin. 5.2 Pharmacokinetic particulars: Following parenteral administration (i.m. or s.c.), gentamicin is almost completely absorbed from the injection site. The absorption after subcutaneous application in cats turned out with 76%, whereas after intramuscular injection, it turned out with 76%. After administration of therapeutic doses (3-5 mg/kg b.w.), within 30 to 90 minutes maximal levels in the blood are reached. According to the animal species peak levels may reach more than 20 μg / ml (in cats). Due to its polar molecular structure in the physiological pH range, gentamicin hardly crosses biological membranes. Its distribution compartment is therefore restricted to the extra cellular space. The gentamicin concentrations obtained in tissue remain low and do not reach values as obtained in the blood, with the exception of the renal cortex and the inner ear, where an undesired accumulation of gentamicin due to an 4/6 active transport is reached. The effect is supported in case of disturbed renal function. The volume of distribution differs in the animal species between 0.2 and 0.5 ml/kg. The distribution increases significantly when the membranes show an increased permeability due to inflammation. The binding of gentamicin to plasma proteins reaches about 20 to 30%, depending on animal species. Elimination of gentamicin after parenteral administration occurs almost entirely by glomerular filtration with 24 hours. Elimination is characterised by an initial phase followed by a time determining terminal phase (t½ß). Elimination half-life is about 1.5 hours in cattle, dogs, and cats, in calves about 3 hours. 6. Pharmaceutical particulars 6.1 List of Excipients: Methyl parahydroxybenzoate, propyl parahydroxybenzoate, sodium metabisulphite, water for injections. 6.2 Incompatibilities: In additionally other drugs are administered, application should be done separately to avoid any risk of interactions (inactivation) e.g. know for mixtures with carbenicillin. 6.3 Shelf-life: Shelf-life Of the finished product as packaged for sale: 2 years Of the finished product after first opening the container: 42 days 6.4 Special precautions for storage: Store below 25°C! Protect from light! 6.5 Nature and composition of immediate packaging: Glass vial (brown coloured, hydrolytic resistance of type II) containing 100 ml solution for injection. 6.6 Special precautions for the disposal of unused veterinary medicinal product of waste materials derived from the use of such products: Any unused veterinary medicinal product or waste material derived from such veterinary medicinal products should be disposed of in accordance with local requirements. 7. Marketing authorisation holder: Demetrios Christoforou Ltd Tseriou Avenue 169 2048,Strovolos,Nicosia 5/6 8. Marketing authorisation number:11959 9. Date of first authorisation/Renewal of the authorisation: 12/10/1998 10. Date of revision of the text: 29/7/2010 11. Prohibition of sale, supply and/or use: Not applicable 12. Legal status: On prescription 6/6